Polar Bonds
0.0(0)
Card Sorting
1/6
Last updated 3:12 PM on 3/6/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
1
New cards
Polar Covalent Bond
Uneven sharing of electrons eg. HCL (hangs around chlorine more).
2
New cards
Delta &
use to indicate small difference positive pole and negative pole &+ and &-
3
New cards
Dipole
One end is positive other is negative
4
New cards
bonding continuum
helps us understand the type of bonding that occurs between two atoms. EN difference
5
New cards
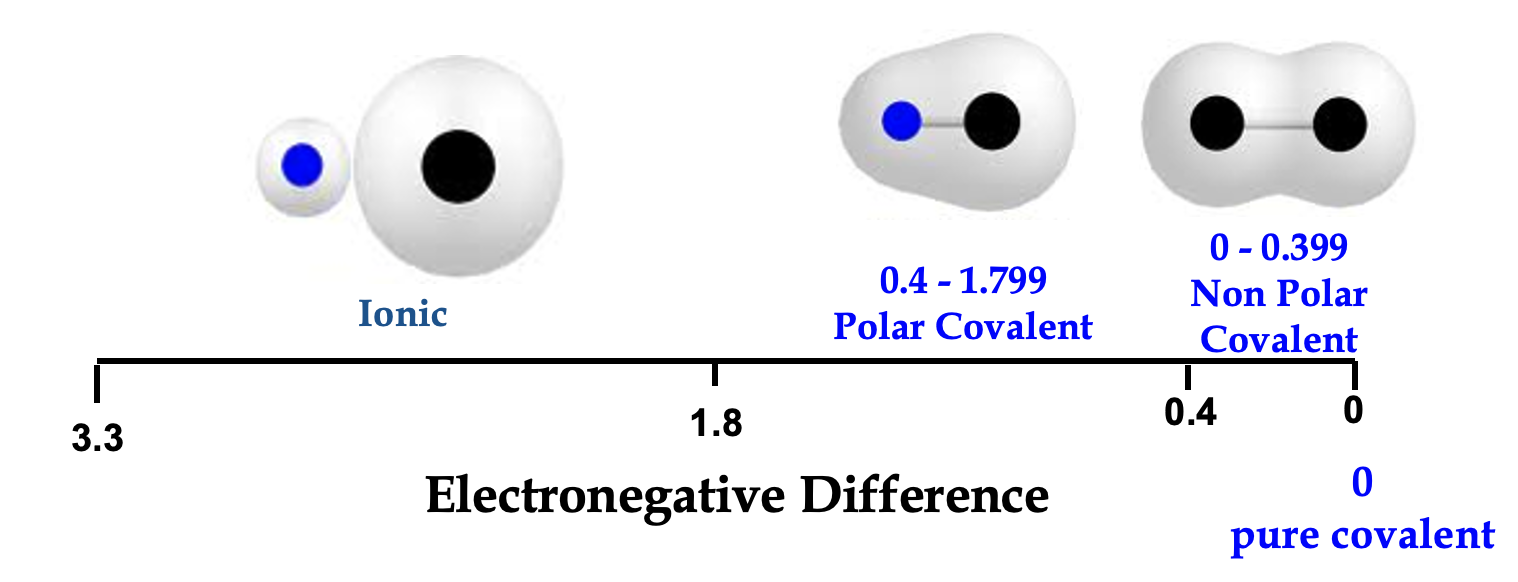
EN Difference
EN Diff> or = to 1.7 it is ionic. En Diff < 1.7 it is covalent. Polar Covalent, Non Polar Covalent/Pure Covalent
6
New cards
Polar Molecules
Need to have 1+ polar bonds and should not have symmetry.
7
New cards