Summative meds
1/66
Earn XP
Description and Tags
Flashcards for revising for CYP medicine management summative exam
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
Paracetamol pharmacokinetics
A Majority is absorbed from the GI tract, mainly the small intestines
D Rapidly and evenly throughout most tissues and fluids
M Predominantly by the liver
E In urine (by kidneys)
Half life ~2 hours
Paracetamol pharmacodynamics
Paracetamol’s exact mechanism of action has not been fully established. It’s usually categorised alongside NSAIDs due to its ability to inhibit the cyclooxygenase (COX) pathways. It’s thought to exert central actions which ultimately lead to the alleviation of pain symptoms. One theory is that it increases the pain threshold by inhibiting COX-1 and COX-2, which are involved in prostaglandin (PG) synthesis. Prostaglandins are responsible for eliciting pain sensations.
It has no anti-inflammatory affects. Paracetamol does not inhibit cyclooxygenase in peripheral tissues and, therefore, has no peripheral anti-inflammatory effects. The antipyretic actions of paracetamol are likely attributed to direct action on heat-regulating centres in the brain, resulting in peripheral vasodilation, sweating, and loss of body heat.
Ibuprofen pharmacokinetics
A In the upper GI tract
D Around the body in the blood and serum
M By the liver into inactive metabolites
E Excreted as metabolites and a small amount unmetabolised, by the liver. Almost completely in urine (>95%)
Half life ~ 2 hours
Ibuprofen pharmacodynamics
Ibuprofen has multiple actions in different inflammatory pathways involved in acute and chronic inflammation. The main effects reported in ibuprofen are related to the control of pain, fever and acute inflammation by the inhibition of the synthesis of prostanoids by COX-1 and COX-2.
It’s a non-selective COX inhibitor and hence, it inhibits the activity of both COX-1 and COX-2. The inhibition of COX-2 activity decreases the synthesis of prostaglandins involved in mediating inflammation, pain, fever, and swelling while the inhibition of COX-1 is thought to cause some of the side effects of ibuprofen including GI ulceration.
The exact mechanism of action of ibuprofen is unknown. However, ibuprofen is considered as an NSAID and thus it is a non-selective inhibitor of cyclooxygenase, which is an enzyme involved in prostaglandin (mediators of pain and fever) and thromboxane (stimulators of blood clotting) synthesis.
Sodium Valproate pharmacokinetics
A rapidly from the GI tract
D primarily distributed within the extracellular plasma
M extensively (90%) in the liver, producing over 220 metabolites
E urine
Half life in children 6-9 hours, in adults ~15 hours
Sodium Valproate pharmacodynamics
Thought to increase cortical inhibition contributing to control of neural synchrony and may have a neuroprotective effect preventing damage and neural degeneration. Exact mechanisms unknown.
Known to inhibit the inhibitor of GABA (increases succinic semialdehyde), which increases GABAergic neurotransmission (an inhibitory neurotransmitter). GABA is an inhibitory neurotransmitter (primary neutrotransmitter for the CNS) which reduces neuronal excitability and slows down neuronal activity
Midazolam pharmacokinetics
A rapidly absorbed dependent on route. buccal and nasal mucosa rapid. IM rapid. poorly orally/GI tract
D high volume of distribution, almost 97% protein bound
M primarily in the liver and gut into hydroxylated metabolites
E in urine
Half life ~2 hours (1-2 in children)
Midazolam pharmacodynamics
Short-acting benzodiazepine CNS depressant. Enhances the inhibitory action of GABA neurotransmitters (gamma-aminobutyric acid).
When binding to the GABA receptors, it increases the frequency of chloride channels opening – produces a sedating effect, relaxes skeletal muscles and reduces the (overactive) electrical signals in the brain (causing the epileptic seizures). The receptors are found in the heart and skeletal muscles, but mainly in the CNS
Benzylpenicillin pharmacokinetics
A - benpen is poorly absorbed orally, easily destroyed by stomach acid (best absorbed by small intestine). Therefore given as IV or IM. Slow absorption. Penicillins generally are absorbed in the GI tract at varying rates and extents.
D - widely distributed, into body tissues and fluids. Poor penetration into BBB/CSF except when meninges inflamed.
M - mostly by liver into metabolites such as peniclloic acid
E - rapidly in urine (and a small amount in bile). Most of it excreted unchanged in therapeutic concentrations.
Half life ~1- 2 hours (older = faster)
Benzylpenicillin pharmacodynamics (penicillin)
Causes the inhibition of cell wall synthesis (bactericidal).
Are beta lactam antibiotics
Penicillins are mainly effective against gram positive (benpen is) but some are gram negative
bacterial cell walls are made up of peptidoglycan made using the enzyme transpeptidase.
Transpeptidase is targeted by beta lactam antibiotics which trick it into binding to peniciliin instead of the cell wall, this stops the enzyme from working
without a cell wall the bacteria lyses
Ondansetron pharmacokinetics
A from GI tract
D widely distributed, around 80% binds to plasma proteins
M some limited first pass metabolism in liver. Later extensively metabolised into major urinary metabolites
E in urine (and faeces)
Half life ~3 hours
Ondansetron pharmacodynamics
Selective antagonist of the serotonin receptor subtype 5-HT3 – it’s an antiemetic. Suppresses vomiting reflex (particularly seen after chemotherapy and radiotherapy as they are associated with the release of 5-HT). the serotonin stimulates the vagal and splanchnic receptors that project to the medullary vomiting centre. Ondansetron stops this.
What are controlled drugs?
Substances regulated by law due to potential for misuse or harm. They include opioids, benzodiazepines and stimulants. CDs are essential for pain management, anesthesia and some MH conditions
What was in the misuse of drugs act 1971?
The act is a statutory framework for the manufacture supply and possession of controlled drugs in order to prevent misuse
Act sets out categories of drugs A, B, C based on harm
Enforced a penalty for use was dependent on the class of drug
What was in the misuse of drug regulations in 2001?
Governs the control and use of CDs, working alongside the Misuse of Drugs Act 1971, providing a more detailed framework for the practical management of CDs within healthcare and research
The regulations control the production, possession, supply, storage, and prescribing of controlled drugs
The regulations classify controlled drugs into five schedules, based on their potential for harm, medical use, and the need for regulatory oversight
What was in the controlled drugs regulations (2006, revised 2013)
In 2007, The Controlled Drugs (Supervision of Management and Use) Regulations (2006) were introduced as part of the Government’s response to the Shipman Inquiry.
The aim of these regulations was to strengthen the governance arrangements for the use and management of CDs.
As a consequence of passing the Health and Social Care Act 2012, the 2006 regulations were revised - The Controlled Drugs (Supervision of Management and Use) Regulations 2013 came into force
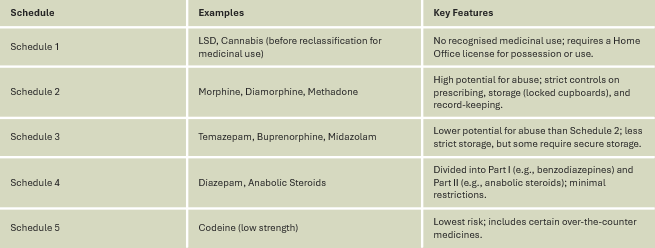
What are the schedules of controlled drugs - what does each mean and gives examples (there’s 5 - 2, 3, 3, 2, 1)
What are CD accountability officers (CDAOs)?
The Controlled Drugs (Supervision of Management and Use) Regulations (2013) require NHS Trusts to appoint CDAOs - Senior professionals responsible for overseeing the safe management and use of CDs within their organisation.
Their role involves:
Developing and implementing systems for monitoring the prescribing, storage, administration, and disposal of CDs.
Overseeing the reporting and management of any incidents, discrepancies, or concerns related to CDs.
Investigating breaches or concerns, including loss, theft, or inappropriate prescribing
How to safely store CDs?
Controlled drugs, particularly those in Schedules 1, 2, and certain Schedule 3 drugs must be stored securely in a manner that limits access to authorised staff only.
CDs must be kept in locked cabinets or safes that meet British Standard security levels
Cabinets should be located in a secure area, out of public view, and inaccessible to unauthorised people.
Only authorised staff should have access to the storage area.
Keys must be kept under secure control or in a digital access system
How to record keep CDs?
A Controlled Drugs Register is a legal requirement for all Schedule 2 drugs and some other CDs as per local policies.
It ensures accountability and traceability of every transaction involving CDs.
Must be a bound book or electronic system that prevents retrospective alteration.
Entries must be made chronologically and at the time of administration. Each entry should be signed by the person responsible for administering and a second checker. CDRs are retained for 2 years after their last entry.
Contents of the CDR:
- Date and time of use
- Name of the drug and its form
- Strength and quantity administered
- Name of the person the drug was administered too
- Amount remaining following administration
How to manage discrepancies in CDs stock?
Regular checks of stock against the CDR should be conducted
Discrepancies may occur due to errors in recording, loss, theft or diversion
Any discrepancies must be investigated and the findings documented
CDAOs should be informed
In cases or theft or diversion, the police and regulatory bodies should be informed
How should you dispose of CDs?
Expired CDs or those that are no longer required must be destroyed in a manner that prevents misuse. Schedule 2 drugs require destruction in the presence of an authorised witness.
Records of destruction must include:
drug name, form and quantity
date and method of destruction
signatures of those involved in the process
What do you need to remember when administering a CD?
Monitor for side effects e.g. resp rate after morphine
Take CD book, drug, and drug chart with you to the patient
Follow the Rs still
Still need a 2nd checker
Fill in CD book - document
Compliance vs adherance?
compliance - ‘‘the extent to which the patient’s behaviour matches the prescriber’s recommendations”
adherence - “the degree to which a person’s behaviour corresponds with the agreed recommendations from the HCP” (assumes an agreement between the patient and prescriber)
Direct methods of monitoring adherence?
assessment of blood levels
measurement of the control of the condition
rates of repeat prescriptions
measuring physiological parameters
Indirect methods of monitoring adherence?
taking an adherence history
reviewing the clinical response to treatment
self-report questionnaires
Issues with non adherence?
increase in morbidity
increase in medical complications
poor QOL
overuse of the healthcare system
leads to ethical debate
Factors that can lead to non adherance
development stage
family dysfunction/positive family functioning
transition
MH issues for the CYP
MH issues for the care giver
complex therapy
side effects
denial of illness
close friends
internal locus of control
treatment with immediate benefits
parents beliefs in seriousness of illness and efficacy of treatment
HCP empathy
What does NICE say about involving people in relation to adherence?
adapt communication style to meet an individuals needs
offer all patients the opportunity to be involved in DM of their medications
offer patients info that is relevant to their conditions and treatments in a way that is easy to understand
be aware of patients concerns over their medications
routinely assess non-adherence in a non-judgmental way
What is polypharmacy?
The use of multiple medications by a patient
Changed over time from 4-5 medications to upward of 10 by some authors
McCraken et al (2017) cites 9 or more medications
NG5 (2015)- the use of multiple medications by a person
More frequently associated with older adults or CYP with complex, long-term conditions that require complex treatment
Appropriate vs problematic pharmacy (Kings Fund, 2013)
Appropriate - Prescribing for an individual for complex conditions or for multiple conditions in circumstances where medicines use has been optimised and where the medicines are prescribed according to the best evidence
Problematic - The prescribing of multiple medicines inappropriately or where the intended benefit of the medicines are not realised
What are the benefits of polypharmacy?
helps to treat co-existing conditions
can reduce symptoms and improve daily functioning
can prevent the progression of certain diseases e.g. cardiovascular disease
What are the challenges of polypharmacy?
risk of adverse drug reactions (ADRs)
increased side effects
medication non-adherence
confusion over multiple prescriptions
higher healthcare costs
What method do we use in CYP for medications reviews and why?
Need and indication
Open questions
Tests and monitoring
Evidence and guidelines
Adverse events
Risk reduction or prevention
Simplification or switches
promotes shared DM and empowerment
What are the 7/8Rs of safe medication administration
Drug
Dose
Patient
Route
Time
Documentation
Refusal
(Reason/form)
What should happen if there is a drug error?
If still being administered and wrong dose/drug etc. stop it
Inform NIC and medical team
Document
Datix
Duty of Candour to explain to patient and family what has happened.
What is self-administration and why is it important?
When patients take responsibility for managing and administering their prescribed medication.
Promotes independence, encourages patient autonomy, and prepares patient for LT conditions
Benefits of SAM for the patient
gives increased understanding of their medication
improves adherence
enhances independence
allows patients to become familiar with new medication in a safe environment
Benefits of SAM for the healthcare system?
reduces nurse workload eventually
identifies potential for education
can support smoother transitions from hospital to home
reduces medication errors
improves long term health outcomes
? cost effective
Disadvantages of SAM for patients?
risk of non-adherence or misuse
patient may not want to self administer
might not be suitable for all medications
Disadvantages of SAM for healthcare systems?
clear policies and guidance need to be in place and adhered to
education, training as assessment of patients is time consuming
Legal and ethical considerations of SAM?
consent and capacity
ensure the patient has capacity
document discussions around consent/assent
accountability
nurses remain accountable for patient safety during the SAM process
clear communication and documentation are essential
professional standards
adhere to NMC code principles of promoting patient safety
What is level 1 of SAM?
Nurse led administration
nurse administers all meds to the patient
the patient is encouraged to observe and learn, fostering understanding of their medication regimen
this level is used when the patient lacks the capacity, confidence or knowledge to self administer safely
What is level 2 of SAM?
Supervised self-administration
the patient administers their own medication under direct supervision from a nurse
this level helps assess the patient’s ability to self-administer correctly and safely, while providing real-time support and education
suitable for patients transitioning to independence but requiring oversight
What is level 3 of SAM?
Independent self-administration
the patient takes full responsibility for managing and administering their own medication without supervision
the nursing team provides ongoing support as needed but does not actively intervene
this level is for patients who demonstrate full understanding, competence, and adherence to their medication regimen
Things to consider when initiating self-administration
CYPs developmental stage
age-appropriate communication
parental involvement when appropriate (family education)
health literacy and understanding
liase with school staff
consider safe storage
how it may affect them socially
regularly review adherence
remember positive reinforcement and empowerment
cultural and individual needs
legal and ethical considerations
Steps for successful SAM implementation?
conduct a thorough risk assessment
develop individualised plans with patient and family
provide training and resources e.g. leaflet and apps
monitor outcomes and address issues promptly
encouragement e.g. sticker charts
What is an advanced drug reaction, and what are the 2 types?
An adverse drug reaction (ADR) is a noxious and unintended response to a medicine, where a causal relationship between the medicine and the adverse event is either known or strongly suspected.
could be mild-severe or life-threatening
A = dose related, B = allergic reactions
ADRs vs side effects
ADRs: Harmful, unintended responses to a drug that occur at normal doses. They are typically classified based on their mechanism (e.g., Type A - dose-dependent, Type B - unpredictable).
ADRs can range from mild to life-threatening and may require medical intervention.
ADRs can be unpredictable, particularly Type B reactions (e.g., allergic reactions).
ADRs are more significant in regulatory reporting and patient safety as they can lead to drug withdrawals or warning
Side Effects: Unintended effects of a drug that occur at normal doses but are not necessarily harmful. They may be predictable.
Side effects are usually mild and do not always require stopping the drug
Side effects are often predictable and based on the drug’s pharmacology
Side effects are typically expected and listed in drug information leaflets
Why are children more vulnerable to ADRs?
Immature liver and kidney function affecting drug metabolism and excretion.
Differences in enzyme activity leading to variations in drug efficacy and toxicity.
Weight-based dosing challenges.
Limited clinical trials in paediatrics leading to gaps in drug safety knowledge
Common ADRs in children
allergic reactions e.g. anaphylaxis, rashes
GI effects e.g. nausea, vomiting and diarrhoea
CNS effects e.g. drowsiness, agitation, seizures
haematological effects e.g. thrombocytopenia, neutropenia
High risk medications for ADRs
antibiotics e.g. penicillin, cephalosporins - risk of hypersensitivity reactions
opioids and analgesics e.g. respiratory depression with morphine
anticonvulsants e.g. sodium valproate - risk of hepatotoxicity, Stevens-Johnson syndrome
chemotherapeutic agents e.g. risk of bone marrow suppression and organ toxicity
How to recognise and assess for ADRs?
regularly assess vital signs and symptoms for potential ADRs - early recognition is important
gain a through medication history to identify potential ADR risks including: current and past meds, allergies and previous ADRs, polypharmacy, any recent changes to meds
inform patients of any side effects and when to seek help
How to classify ADRs as mild moderate severe or delayed?
Mild = nausea, dizziness, headaches, mild rashes
Moderate = diarrhoea, significant drowsiness, hypotension, minor bleeding
Severe = anaphylaxis, severe rashes (e.g. Stevens-Johnson syndrome), organ toxicity (hepatoxicity, nephrotoxicity)
Delayed = conditions affecting the blood (e.g. anaemia, thrombocytopenia), long term organ damage, carcinogenic effects
Management of mild/moderate ADRs
immediate actions: stop the medication if necessary and put in place any interventions required e.g. antiemetics, antihistamines
remember A-E
communicate - inform prescriber, doctors and pharmacist
provide support, reassurance and education to the CYP and their family
close observation to look out for worsening symptoms
consider lowering the dose of medication or switching to an alternative
Pathophysiology of anaphylaxis
On first exposure the allergen stimulates B cells to produce IgE antibodies which bind to the surface of mast cells and basophils “priming” them
On subsequent exposure, the allergen binds to IgE antibodies on these, triggering a huge histamine release from these cells.
Leads to vasodilation, increased vascular permeability and bronchoconstriction
Systemic effects of anaphylaxis (CVD, resp, GI, skin+mucous)
Cardiovascular - vasodilation (hypotension and shock due to fluid leakage into tissues), tachycardia and arrhythmias (due to compensatory adrenaline release)
Resp - bronchoconstriction (airway narrowing, wheezing and distress), mucosal oedema (swelling of airway leading to laryngeal obstruction)
GI - smooth muscle contraction (abdo pain, vomiting and diarrhoea)
Skin and mucous membranes - urticaria (hives) and angioedema (due to increase cap permeability)
Management of anaphylaxis
Immediate IM adrenaline
Oxygen and IV fluids to maintain O2 and BP
continuous monitoring for biphasic reactions
Meds such as antihistamines, corticosteroids, beta-2 agonists
crash bell, crash trolley, A-E, inform PA, Dr, pharmacist
document in notes, datex, and yellow card reporting
reassure CYP and family
What is the yellow card scheme?
national reporting system for ADRs run by MHRA
encourages HCPs, patients and caregivers to report ADRs to improve drug safety monitoring
What is covert administration?
The practice of administering medication to a patient without their knowledge, typically disguised in food or drink.
For children and young people under 16, this practice may be considered when the child refuses essential medication and lacks capacity to make informed decisions
Legal frameworks to consider for covert administration
For those over 16 years:
Mental Capacity Act (2005)- Ensures decisions are made in the patient's best interests if they lack capacity.
For those under 16 years:
Gillick competence- a child/young person can consent to their treatment if they have sufficient understanding and intelligence to appreciate what it involves. If not Gillick competent, decisions are made by parents/guardians in the child’s best interests.
The Children Act (1989)- requires actions to prioritise the child’s welfare as the paramount concern.
NMC Code (2018)- Uphold professional accountability and ethical standards
Ethical considerations for covert administration
Balancing beneficence and non-maleficence.
Respect for developing autonomy- children/young people should have the opportunity to engage the child in discussions about their treatment.
Parental responsibility- parents (or legal guardians) can provide consent for a child who lacks capacity
Best interests- consider the short- and long-term benefits for the child’s health and wellbeing
DM for covert administration in over and under 16s
Over 16s
assess capacity
convene a best interests meeting
document decisions
Under 16s
assess competence
consult parents/guardians if the child is not competent or refuses medication
engage the MDT
document decisions and discussions and strategies attempted
Challenges of covert administration in practice
Navigating ethical dilemmas and ensuring the best interest's principle is upheld.
Conflicts between parents wishes and healthcare professionals recommendations
The emotional impact of refusal and covert administration on the child/young person, family and healthcare professionals
Cultural/religious beliefs may influence decisions
Medication efficacy needs to be considered
Staff may need training to administer medication correctly
Hospitals, schools and home settings needs to align their approaches to avoid errors
Strategies to overcome challenges of covert administration
promote shared DM - use appropriate communication and work with parents
ensure legal compliance - follow MCA or Gillick to assess capacity, seek support from safeguarding if parental disagreements or concerns, keep reviewing and document
clinical and safety concerns - consult pharmacist, implement clear protocols, minimise deception, support wellbeing
practical challenges - standardised training, use behavioural strategies e.g. rewards
MDT oversight - involve everyone in DM, review to assess if needed
What to remember when doing covert administration?
Should only be considered when all other methods have been explored and have failed.
Ensure the decision aligns with the child/young person’s best interests
Follow pharmacist recommendations to avoid altering medication efficacy
Ensure all are aware of the agreed upon method to prevent errors
Monitor for ADRs
Regularly assess the need for covert administration of med
Step down plan from covert administration
Schedule periodic reviews to assess whether covert administration is still necessary
Work towards open administration- involve the CYP in medication discussions if appropriate
Develop a tailored step-down plan (small realistic goals)
Gradual exposure and reduced concealment
Encourage independence with medication
Gradually shift towards open exposure
Transition to full awareness
At all times, monitor for emotional and behavioural responses