Chapter 7 - Enzyme mechanisms
1/65
Earn XP
Description and Tags
Biochemistry chapter 7
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
66 Terms
Ribozymes are enzymes composed of
RNA
Most enzymes are
Proteins
Lock and key model
A biological concept explaining that a specific substrate fits into a specific enzyme's active site, similar to a key fitting into a lock.
induced-fit model
<span>A theory that suggests enzymes undergo a conformational change to better fit the substrate upon binding, enhancing the enzyme's activity and specificity.</span>
conformational selection
A model of enzyme interactions where the enzyme exists in multiple conformations and the substrate selectively binds to one preferred conformation.
Enzymes usually bind substrates with high specificity
due to their unique active site shapes and the presence of specific amino acid residues that interact with the substrate.
Substrate binding to the active site is often associated with structural changes in the enzyme
Induced fit modelsuggests that the enzyme changes shape to better bind the substrate.
Enzyme activity is highly regulated in cells
this is because it is necessary to maximizes energy balance between catabolic and anabolic pathways
The two primary modes of enzyme regulation are _______ and ________
Bioavailibility, activity
Bioavailbility
the amount of enzyme present in the cell as a result of regulated gene expression and protein turnover.
Enzymes alter the rate of reactions without
changing the ratio of substrates and products at equilibrium. They simply decrease the time it takes to reach equilibrium.
Catalase
an enzyme that decomposes hydrogen peroxide (H2O2)
Transition state theory
Transition state theory explains the process of bond formation and breaking during a chemical reaction. It focuses on the high-energy, unstable states (transition states) that exist momentarily as reactants transform into products. This theory helps in understanding the activation energy needed to initiate a reaction and the pathway taken by molecules as they progress from reactants to products.
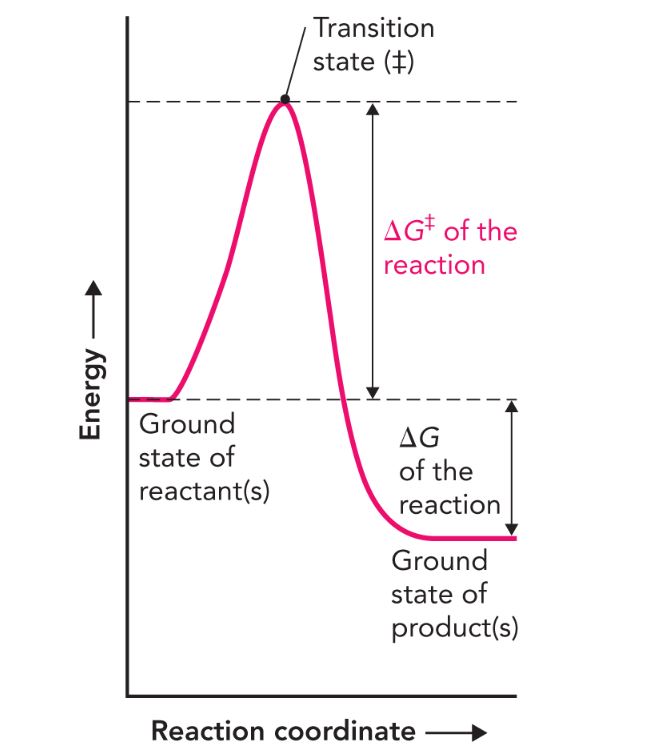
What is this called? What does it do?
Reaction coordinate diagram. It measures the change in free energy between the ground states of products and reactants.
Activation energy
The energy required to reach the transition state (Delta G)
How does catalase work?
It provides a highly reactive Fe3+ porphyrin ring with a protected environment, which promotes product formation as a function of the reduced transition state energy barrier.
Cofactors
Small molecules that aid in the catalytic reaction within the active site.
Include inorganic ions such as Fe2+, Cu2+ and Mg2+
Holoenzyme
An enzyme with a bound cofactor
Apoenzyme
Enzyme without cofactor
Coenzymes
Enzyme cofactors that require organic components (Non protein)
Prosthetic groups
coenzymes that are permanentyl associated with enzymes
Common cofactors used in catalysis are
typically trace elemnts found in the body
What is a heme
A prosthetic group found in enzymes and proteins that has a central Iron inside a porphyrin ring that is critical in forming reactions.
NAD+/NADH
Typically found in a redox reaction where NAD+ is the oxidized form and NADH is the reduced form.
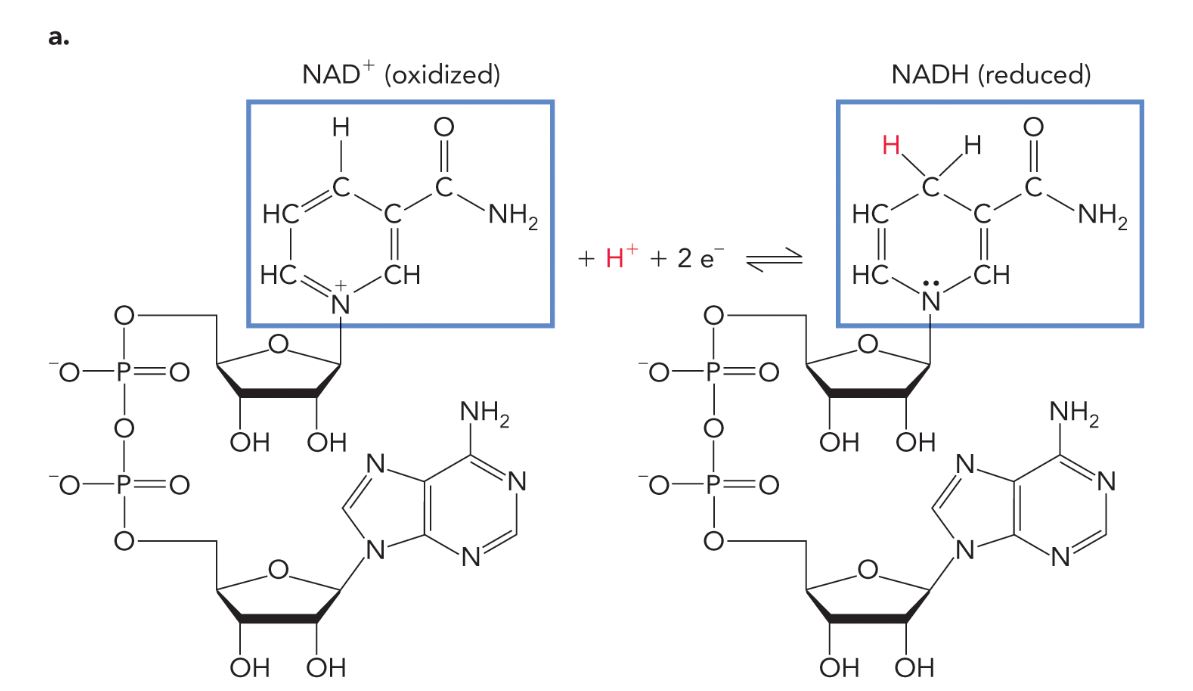
NAD+/NADH Redox reaction
The NAD+/NADH redox reaction is a metabolic process where NAD+ acts as an electron carrier, converting to NADH by accepting electrons during cellular respiration. This conversion is crucial for ATP production, as NADH donates electrons to the electron transport chain.
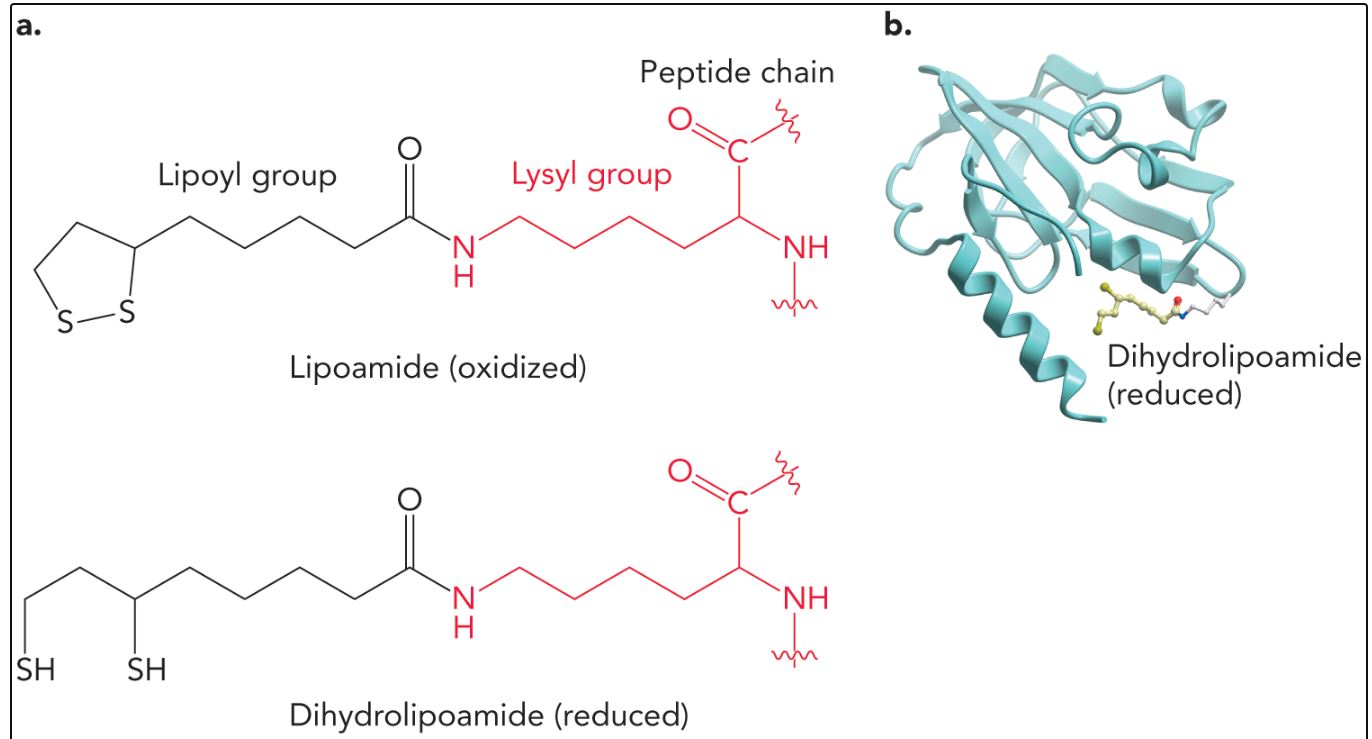
Lipoamide
a redox-active cofactor derived from lipoic acid, playing a crucial role in enzyme-catalyzed reactions. It facilitates the transfer of acyl groups and electrons, thus participating in various metabolic pathways, including the pyruvate dehydrogenase complex and the alpha-ketoglutarate dehydrogenase complex. Lipoamide acts as a bridge in the transfer of acetyl and other acyl groups, contributing to ATP production and cellular respiration.
Most enzymes end in
“-ase”
Oxidoreductase
Participates in oxidation-reduction reactions and trasnfers either H or O atoms.
Transferase
Type of reaction: transfers functional groups
Hydrolase
Formation of two products by hydrolyzing (breaking chemical bonds using water) a substrate
Lyase
Cleavage of C-C, C-O, C-N and other bonds by means other than hydrolysis or oxidationI
Isomerase
Intramolecular rearrangements, transfer of groups within molecules
Ligase
Formation of C-C, C-O, C-S, C-N bonds using ATP cleavage
Enzymes increase the rate of reaction inside cells in three major ways
They stabilize the transition state, thus lowering the activation barrier
They provide an alternative path for product formation
They orient the substrates appropriately for the reaction to occur hence reducing entropy
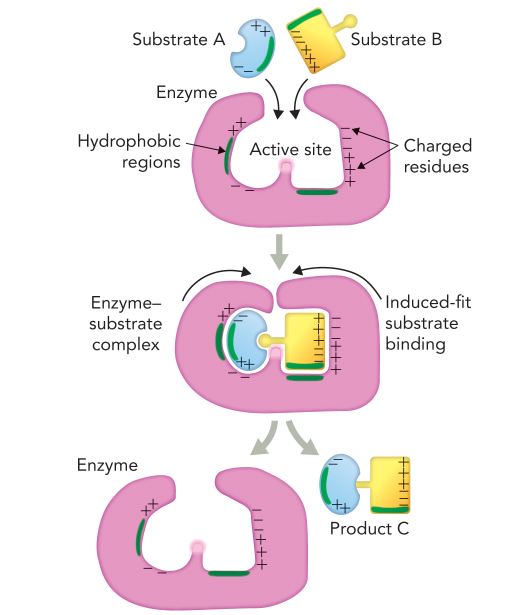
What is happenning here?
The enzyme active site produces an optimal environment that promotes product formation by deterring random collisions and coordinating the reactants to from a stable product and aligns the reactive groups correctly.
What are the three specific physical and chemical properties of enzyme active sites that contribute to their catalytic properties?
The sequestered microenviornment of the active site
Excludes excess solvent
Provides optimal orientation of the substrate relative to the reactive chemical group
binding interactions between the substrate and the enzyme that facilitate formation of the transition state
The presence of catalytic functional groups
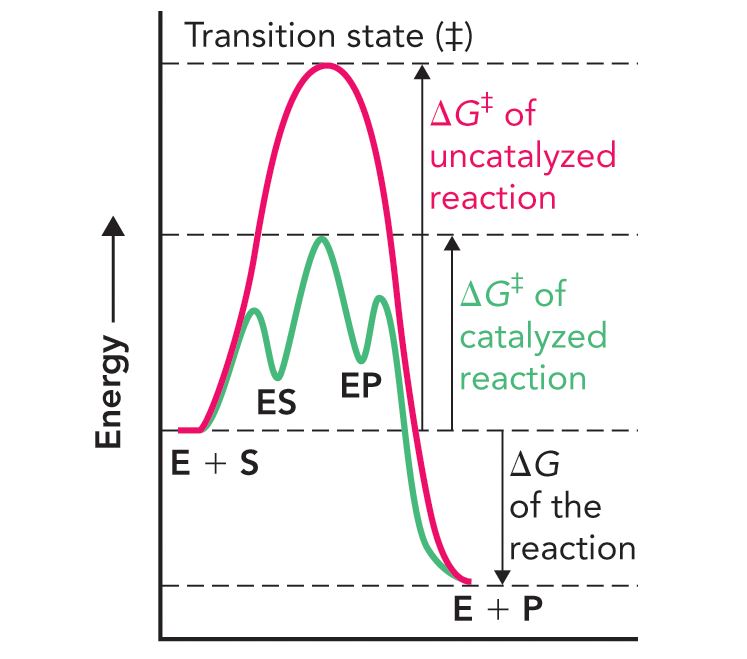
What is happenning here?
Substrate binding to the enzyme (E+S) leads to the formation of an enzyme-substrate complex (ES) followed by the conversion of the enzyme-bound substrate to an enzyme bound product (EP). Finally, the product is released from the enzyme (E+P).
Transition state analogs
A molecule that mimics the transition state of a chemical reaction but is actually stable causing no reaction to occur.
What are the three most common catalytic reaction mechanisms in the active enzyme site?
Acid base catalysis
Covalent catalysis
Metal-ion catalysis
Acid-base catalysis
The removal or addition of a proton. Can happen in specific acid-base catalysis or general acid-base catalysis.
Specific acid-base catalysis
Involves water to transfer protons
General acid base catalysis
Involves a functional group to transfer protons. (i.e water and zinc)
Covalent catalysis
Creates an unstable intermediate that promotes the catalytic reaction by forming a transient covalent bond between the substrate and the enzyme. This is done because the nucleophile group on the enzyme attacks the electrophilic group on the center of the substrate.
Metal ion catalysis
Metals are used to promote proper orientation of bound substrates and can aid in redox reactions
The three general categories of enzyme mediated reactions are:
Coenzyme-dependent redox reactions
Metabolite transformation reactions
Reversible covalent modification reactions
Coenzyme-dependent redox reactions
Redox reactions that involve coenzymes, allowing for the transfer of electrons and protons in biochemical reactions.
Include dehydrogenases
Involve NAD+/NADH, NADP+/NADPH, FAD/FADH2 and FMN/FMNH2
Metabolite Transformation reactions
The chemical transformation of metabolites to generate reactive intermediates
Three types of reactions are the most common in metabolic pathways:
Isomerization
Condensation
Hydrolysis/Dehydration
Isomerization
Reactions that do not change the molecular formula of the product compared to that of the substrate
Condensation
Combines two substrates to form a larger molecule with the loss of a smaller molecule
Hydrolysis or dehydration
involve the addition or removal of water. For many hydrolysis reactions, the substrate is cleaved when water is added.
Reversible covalent modifications
These are chemical modifications on proteins or other biomolecules that can be easily added or removed. Common modifications include phosphorylation, acetylation, methylation, and ubiquitination. These modifications play crucial roles in regulating protein function, including enzyme activity, protein stability, localization, and interactions with other molecules, allowing for dynamic control of biological processes
Chymotrypsin: A serine protease
Aids in digestion to cleave the peptide backbone of dietary proteins
Uses His, Asp, Ser to form a hydrogen bonded network required for catalysis
Ser is converted to a highly reactive nucleophile
Describe the two phases of the mechanism of chymotrypsin
In the first phase, and acyl-enzyme intermediate is formed which promotes cleavage of scissile peptide bond. The carboxyl terminal is then released.
In the second phase, the enzyme is regenerated after a series of steps that results in de-acylation and release of the amino-terminal polypeptide fragment.
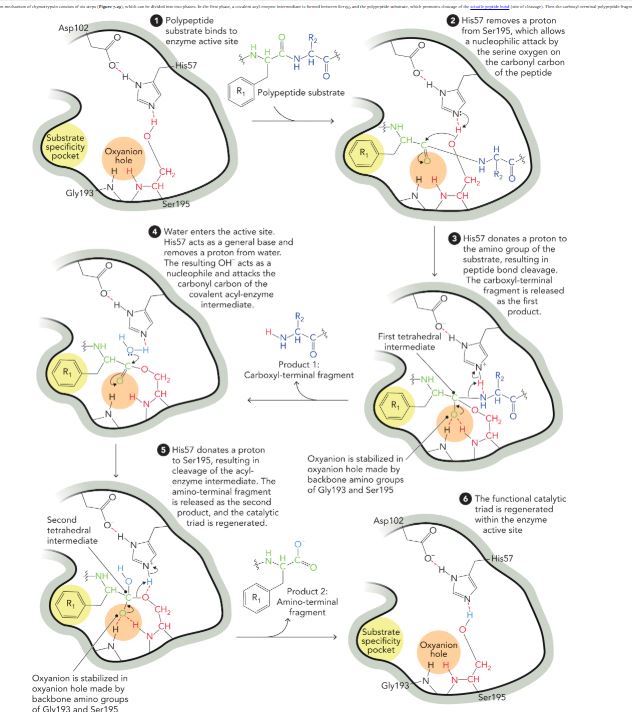
Chymotrypsin reaction mechanism
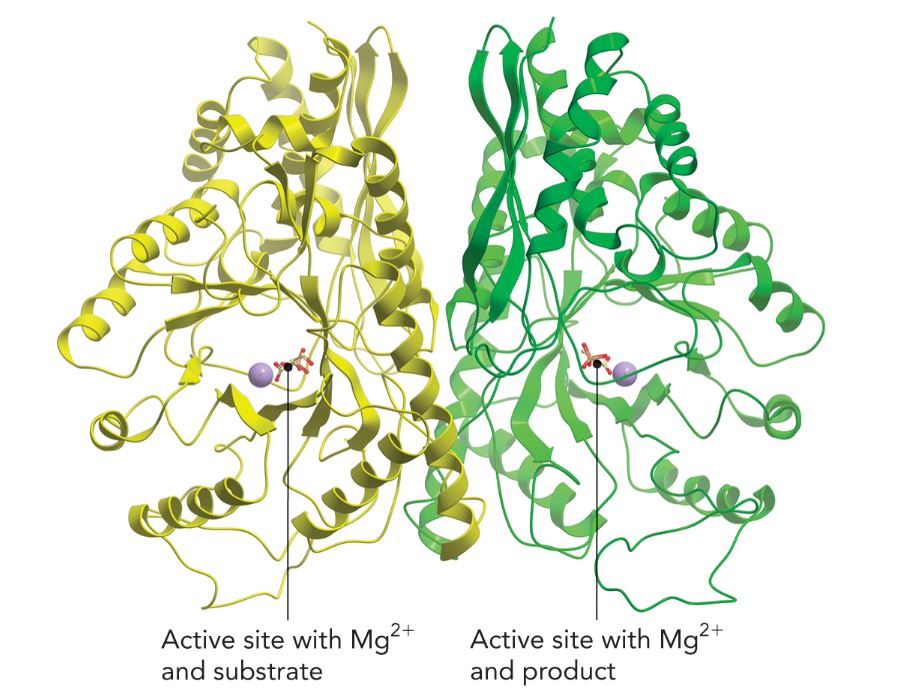
Enolase
An enzyme that contains two divalent metal ions which are required for a reaction used in gluconeogenesis.
Enzyme regulation is mediated by two primary mechanisms
Bioavailability with regard to the amount of enzym in different tissues and cellular compartments
control of catalytic efficiency through protein modification
Stimulatory enzyme activity
Enzyme activity that enhances or accelerates metabolic processes.
Inhibitory regulation of enzyme activity
Control mechanism that decreases enzyme activity and slows metabolic processes.
Allosteric regulation of catalytic activity
R state
Enzyme is active and is ready to be binded to ATP hence starting a reaction
T state
inactive enzyme- binding of CTP (feedback inhibition) stopping a reaction
Competitive inhibitor
When the inhibitory competes with a subtrate for the same space in the enzyme active site
Uncompetitive inhibition
The inhibitor binds only once the substrate is bound to the active site
Mixed inhibition
Mixed inhibition occurs when an inhibitor decreases the activity of an enzyme by binding to both the enzyme and the enzyme-substrate complex, resulting in reduced reaction rates.