AP Bio Unit 1 - Chemistry of Life by Kori
1/131
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
132 Terms
Hypothesis
An explanation to a question, tested by an experiment or continuous observation.
Theory
Summarizes a group of hypotheses.
Inductive reasoning
A process of reasoning going from specific to broad.
Deductive reasoning
A process of reasoning going from broad to specific.
Experimental group
A group that is tested with a changed variable.
Scientific Law
Statement of fact usually as a mathematical formula.
Control Group
Expected results of an experiment.
Constants
All the factors that stay the same in an experiment.
Mean
Average of the data set.
Median
Middle number in a range of data points.
Mode
Value that appears most often in a data set.
Independent variable
One factor that is changed or manipulated.
Dependent variable
Factor which is measured.
Null hypothesis
No difference between two groups of data and experimental observations due to chance.
Alternative hypothesis
Opposing theory to the null hypothesis.
Central tendencies
Statistical measures that describe data trends.
Variability
Measure of how far a data set diverges from central tendency.
Range
Difference between largest and smallest values.
Standard deviation
Measure of how spread out the data is from the mean.
Standard error of the mean
Determines precision and confidence in mean value.
Solute
Product that the solvent dissolves.
Solution
Product of solute and solvent.
Solvent
Dissolving agent in a solution.
Valence electrons
Outermost electrons in outmost valence shells.
Molecule
Atoms bonded together.
Acid
Molecule that can donate or accept a proton.
Base
Molecule that can accept hydrogen ions in water.
Functional groups
Chemical groups attached to carbon skeleton that participate in chemical reactions.
Organic chemistry
Study of compounds with covalently bonded carbon.
Organic compounds
Compounds that contain Carbon and Hydrogen.
Hydrocarbons
Organic molecules of only carbon and hydrogen.
Dehydration reaction
Bonds two monomers with the loss of H2O.
Hydrolysis
Breaks bonds by adding H2O.
Polymers
Chain-like macromolecules of similar or identical repeating units.
Monomers
Repeating units that make up polymers.
Disaccharide
Two monosaccharides joined by covalent bonds.
Monosaccharide
Simple sugars formed with multiple units of CH2O.
Polysaccharide
Polymer with many sugars joined via dehydration reactions.
Carbohydrate
Sugars and polymers of sugars containing a carbonyl group and many hydroxyl groups made of Carbon, Hydrogen, and Oxygen.
Proteins
Molecules consisting of polypeptides folded into a 3D shape.
Amino Acids
Molecules that have an amino group and a carboxyl group.
Polypeptide
Many amino acids linked by peptide bonds.
Cellulose
Polysaccharide that is the structural component of the cell wall.
Glycogen
Polysaccharide of glucose stored in the body as energy storage.
Hydrophobic
Repel water.
Hydrophilic
Attracts water.
Glycosidic linkage
Covalent bonding connecting a carbohydrate to another group.
Starch
Polysaccharide of glucose stored as energy in plants.
Primary Structure
Linear chain of amino acids determined via genes.
Secondary Structure
Coils up and folds due to hydrogen bonding within the backbone of the polypeptide.
Tertiary Structure
3D folding due to interactions between the side chains of the amino acids.
Quaternary Structure
Association of two or more polypeptides only found in some proteins.
Lipid
Class of small, nonpolar molecules that do not include true polymers.
Fats
Composed of glycerol and fatty acids.
Fatty Acid
Long carbon chains with a carboxyl group at the end.
Phospholipid
Component of cell membranes, two fatty acids attached to glycerol and phosphate.
Denature
Unfolding of a protein and modifying the structure.
Steroid
Molecules that regulate cellular functions like reproduction and development.
Antiparallel
DNA strands run in opposite directions parallel to one another 3’-5’.
DNA
Deoxyribonucleic acid, carries genetic information for the development and functioning of an organism.
RNA
Ribonucleic acid, a nucleic acid used to produce proteins.
Nucleotide
Compound of a nitrogenous base, pentose, and a phosphate group
Larger range
Greater variability.
Smaller range
Less variability.
Low standard deviation
Data is closer to the mean, IV causes changes.
High standard deviation
Data is farther from the mean, other factors cause changes.
What is the difference between inductive and deductive reasoning?
Inductive is specific to broad and deductive is broad to specific
What is the difference between a theory and a law?
A theory summarizes a group of hypothesis while laws state scientific facts, usually a math equation
How are hypotheses formulated?
From a question to be proven or disproven, stating a null hypotheses and alternate hypotheses
How are variables often graphed?
Dependent Y-axis, independent X-axis
When should a positive control be used?
To compare experimental exposure to a known, effective exposure
When should a negative control be used?
To compare the effectiveness of exposure to no exposure
Quantitative
Measured with instruments.
Qualitative
Measured with senses.
When should median be used?
When a data set has extreme values or abnormal distribution
When should mode be used?
When describing the distribution of data where mean and median are not appropriate
What is used to measure variability?
Range and standard deviation
Polar
Electrons not equally shared
Nonpolar
Electrons equally shared
What are two examples of covalent bonds?
Carbon dioxide, glucose
What are two examples of ionic bonds?
Sodium chloride, lithium fluoride
How does adhesion benefit living organisms?
Water can penetrate the cell wall of a plant
How does cohesion benefit living organisms?
Moving water up the xylem against gravity
How does surface tension benefit living organisms?
Organisms that float on water can do so
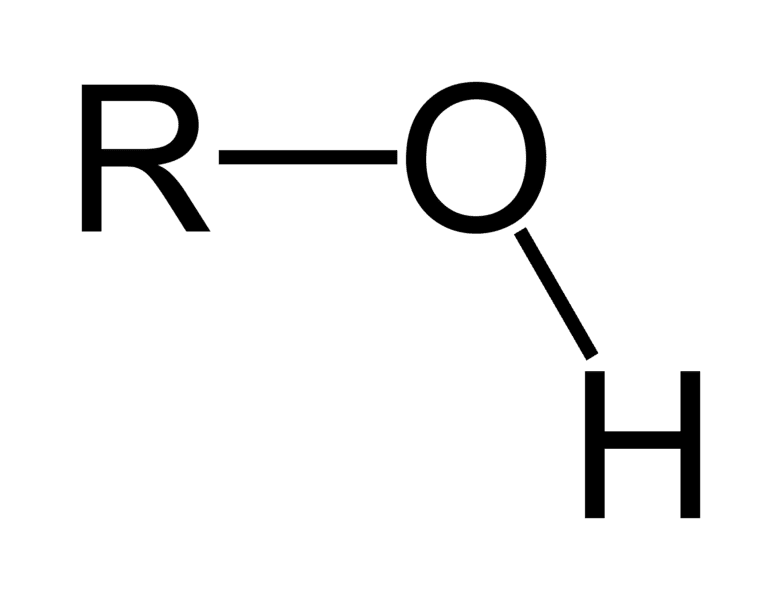
Hydroxyl Group
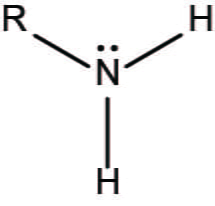
Amino Group
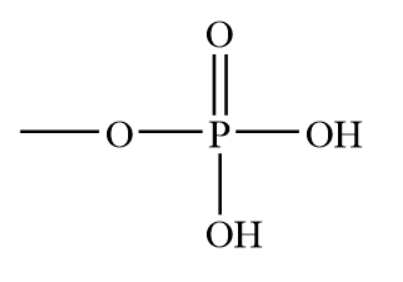
Phosphate Group
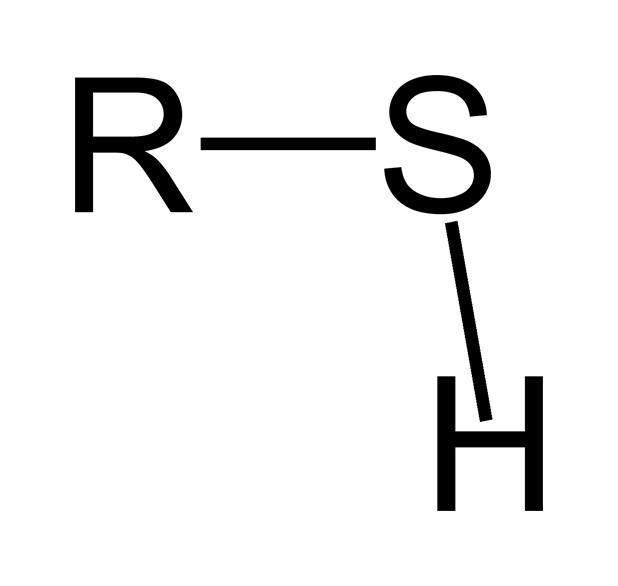
Sulfhydryl (Thiol) Group
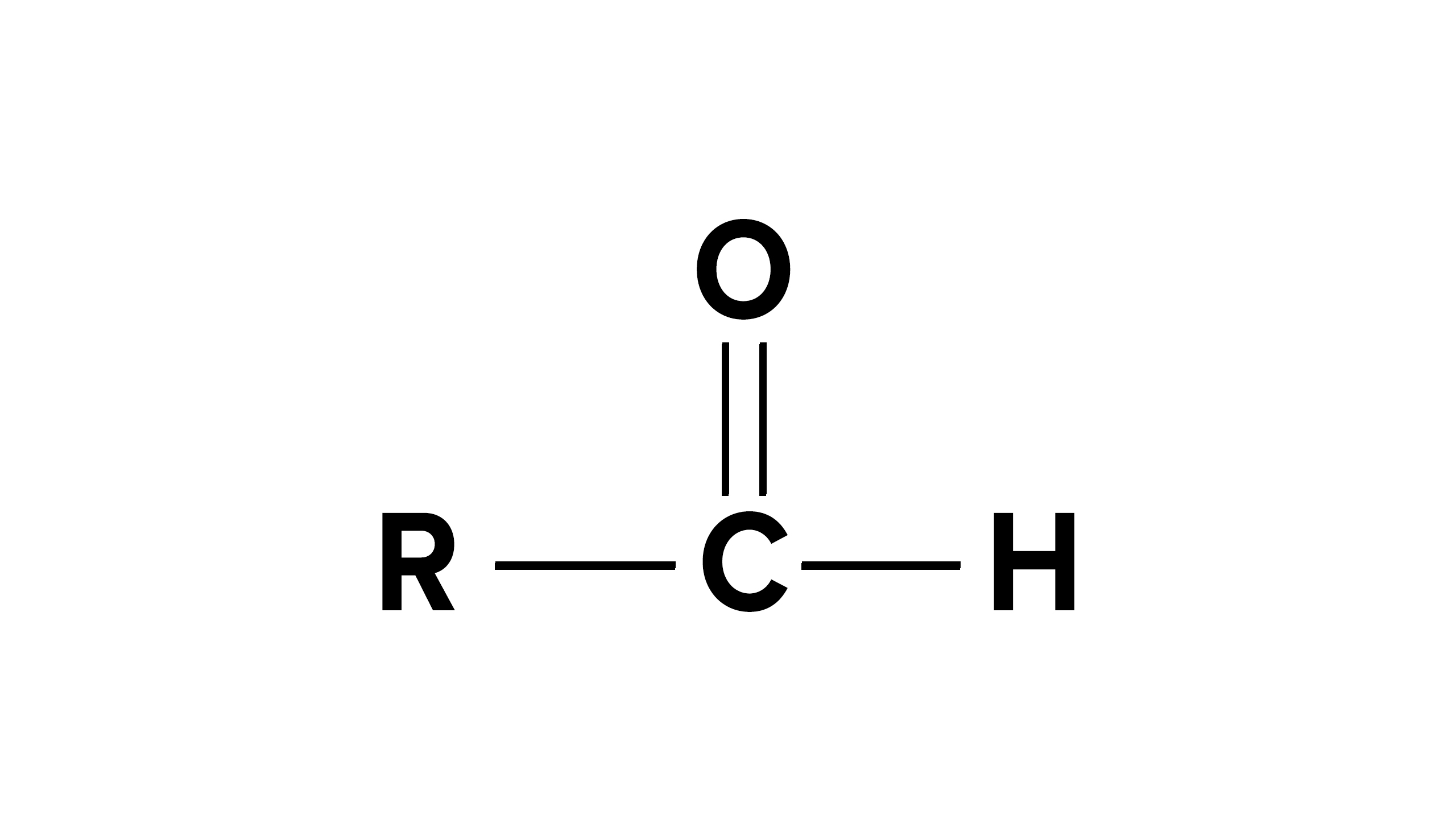
Carbonyl Group
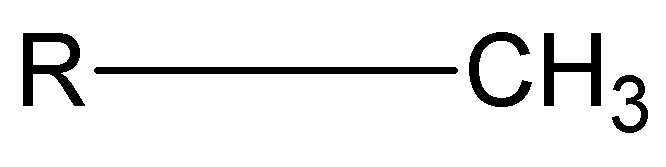
Methyl Group
Why is carbon such a versatile element?
It can form long chains and carbon skeletons to support organic molecules.
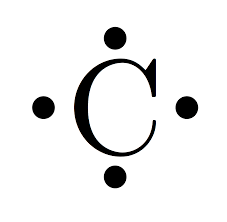
Lewis dot carbon molecule
How many valence electrons does carbon have?
4
How do functional groups affect the structure and behavior of organic molecules?
It affects the reactivity and polarity of an organic molecule
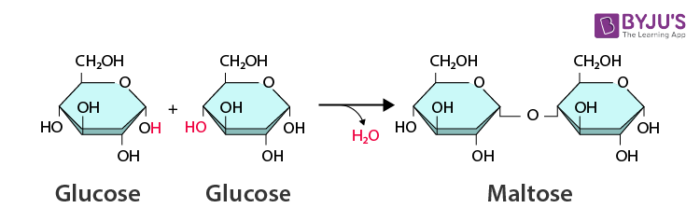
Dehydration Reaction
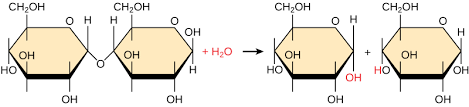
Hydrolysis
How many monomers of proteins are there?
20
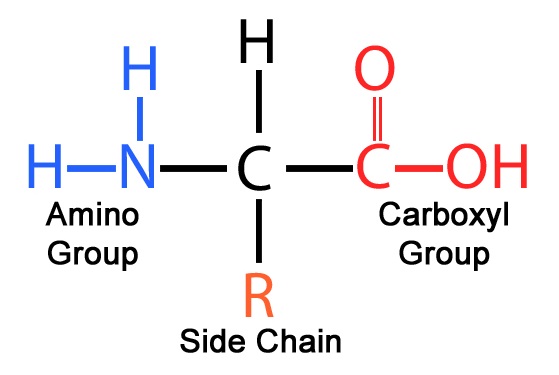
Amino Acid
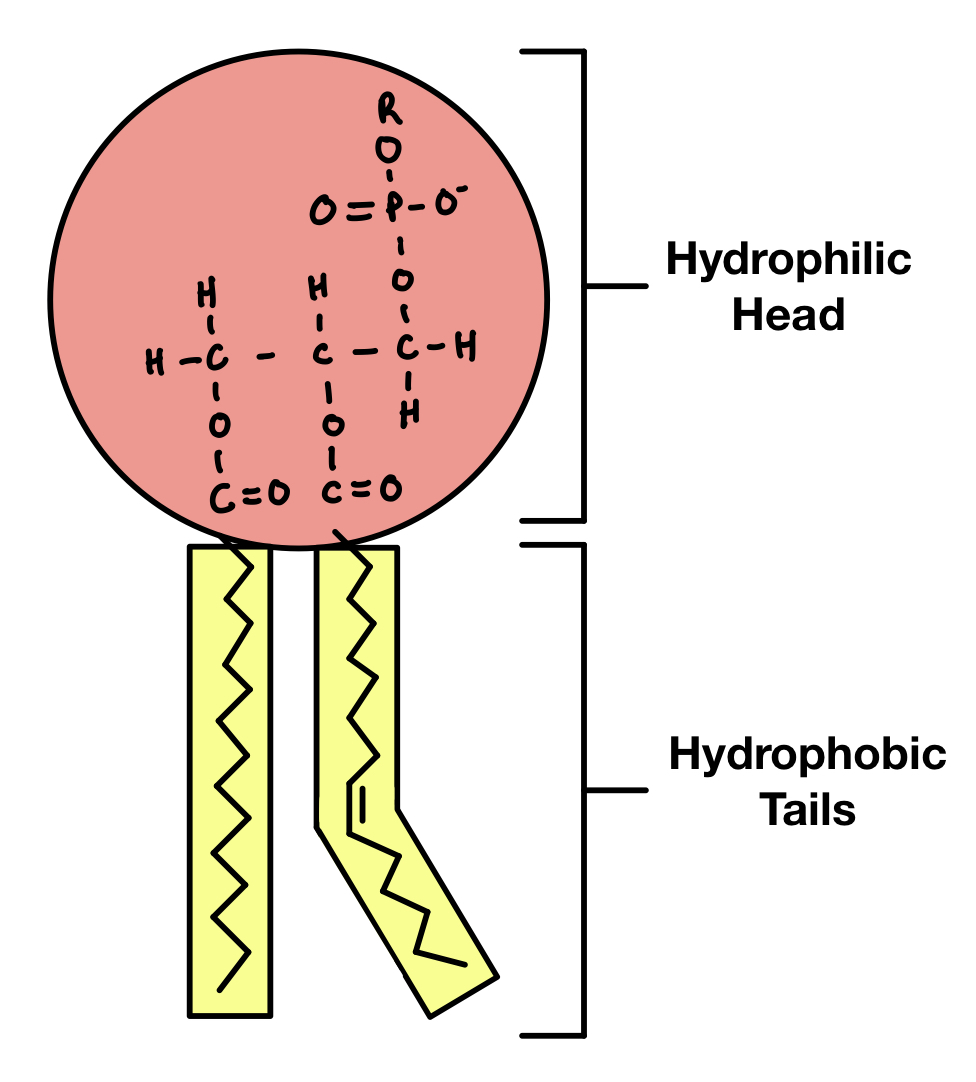
Phospholipid
Where can phospholipids be found in a cell?
Cell membrane