Reactivity 3.14 The pH scale
0.0(0)
0.0(0)
Card Sorting
1/6
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
7 Terms
1
New cards
why the pH scale should be logarithmic
because majority of acids are weak and therefore produce H+ concentrations with large negative exponents
2
New cards
inventor of pH scale
Soren Sorensen, 1909
3
New cards
definition of pH scale
pH= -log10[H+]
4
New cards
features of pH scale
usually positive
has no units
change of one pH=10 fold change in H+ concentration
5
New cards
relationship pH and H+ ion concentration
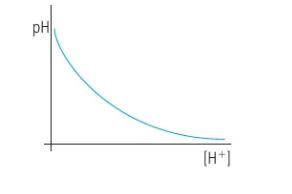
6
New cards
measuring pH
universal indicator paper/ solution
colour estimation shows pH
more exact:
pH meter
has to be calibrated prior to each measurement
temperature must be standardised
7
New cards
pH calculations
relationship between H+ and OH- is inverse in aqueous solutions, so low H+ means high OH-