Nitrogenous compounds + polyesters module 6
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
amine structures ( Image)
N attached to 1 R group = primary
N attached to 2 R group = secondary
N attached to 3 R group = tertiary
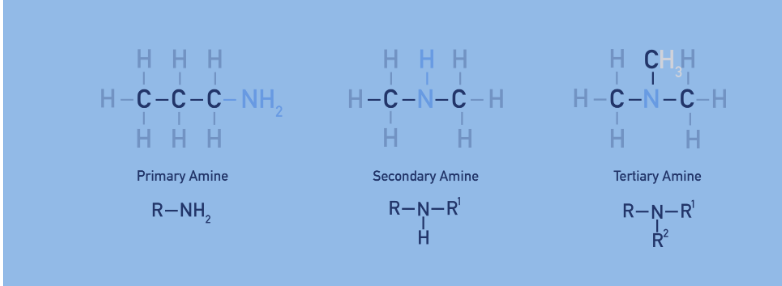
what are type of molecule are amines
Bases
lone pair on N accepts protons
How to name amines
Suffix - amine
prefix - Alkyl group (alphabetical order )
amine + acid →
Amine + Acid → Salt
(Amine + Acid ) explanation
NH₂ acts as base and accepts proton
Acid ion ionically bonds to N⁺H₃
Phenylamine + Nitric acid ( Image)
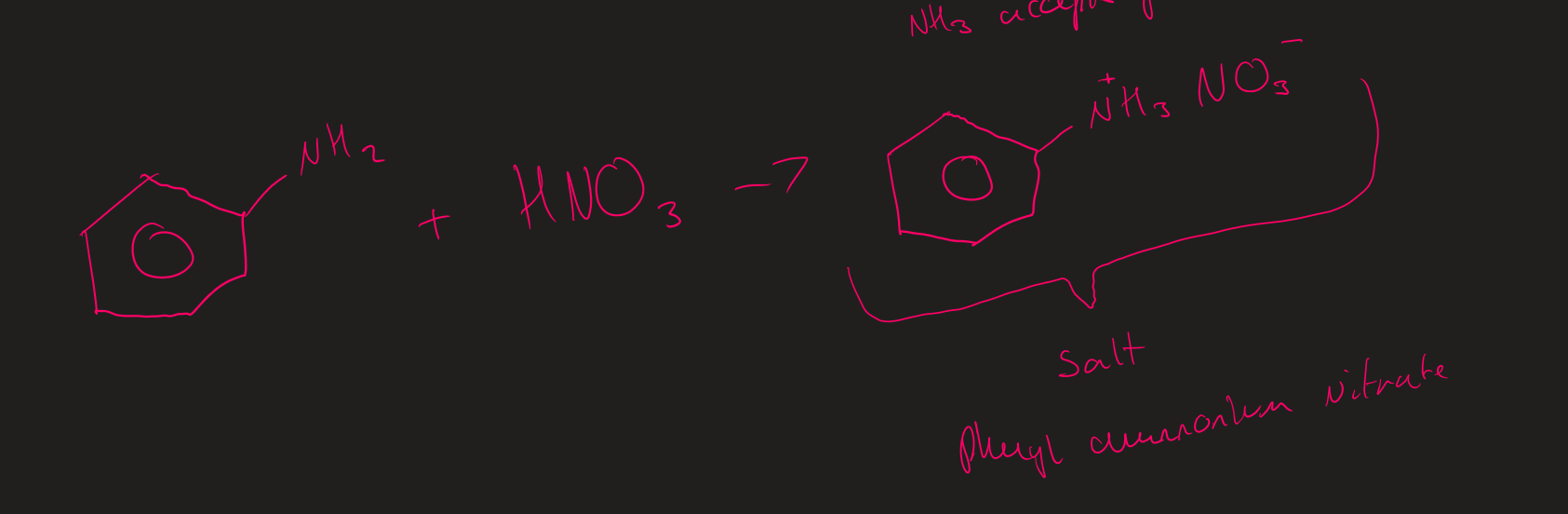
Amine + Acid how to name salt
Alkyl ammonium ion
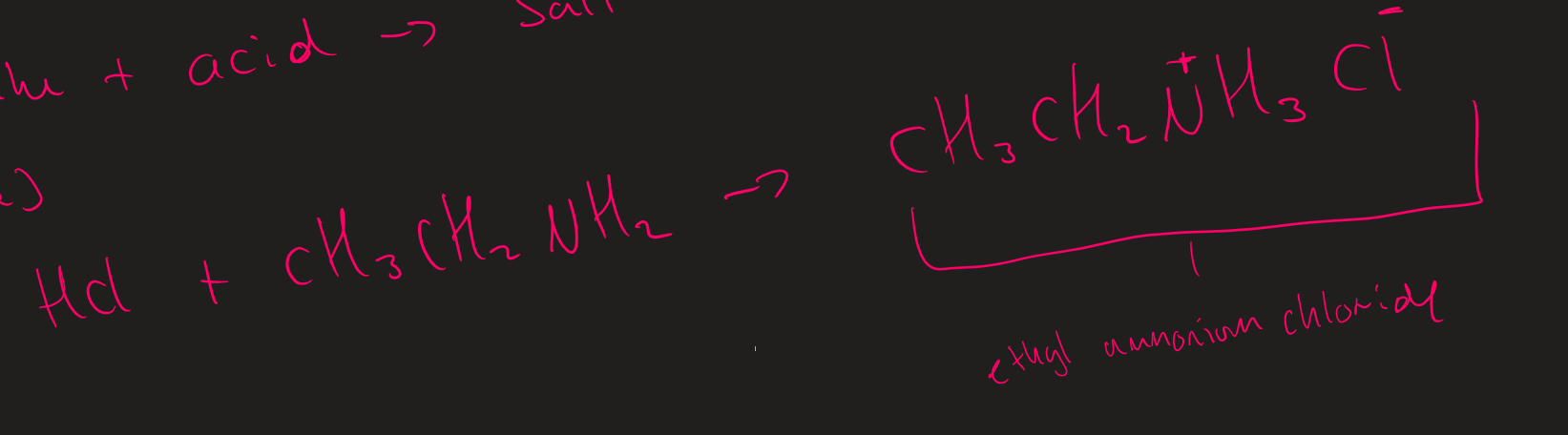
How to make an aliphatic amine reaction
Haloalkane + Excess NH₃ dissolved in ethanol → Primary amine + ammonium halide
What do amines act as in nucleophilic substitution reactions
Nucleophile → electron pair donor
Why do we use excess NH₃
Prevents further substitutions
no undesired secondary / tertiary amines
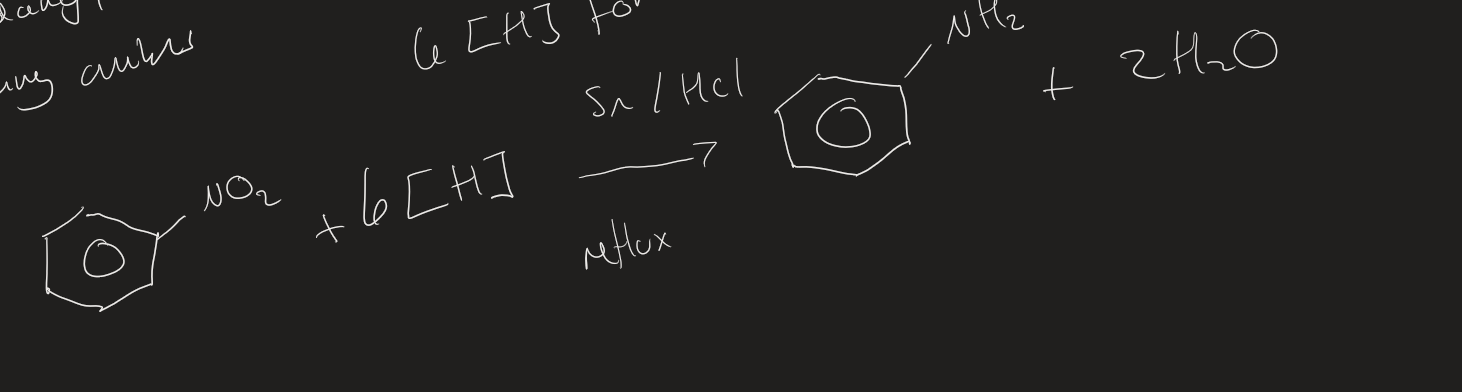
How to make a aromatic amine equation
Reduce nitrobenzene → Phenylamine
Reactants to make primary amine
Haloalkane
excess NH₃ dissolved in ethanol
By product of making a primary amine
Ammonium halide
Why is NH₃ dissolved in ethanol
Prevents H₂O from reacting with haloalkane → produces alcohol
Conditions to form a phenylamine
Sn/ HCl
reflux
How many [ H ] to reduce 1 nitro group on nitrobenzene to form phenylamine
6 [ H ]
forming a aromatic amine word equation
C₆H₅NO₂ + 6 [ H ] → C₆H₅NH₂ + 2H₂O
forming aromatic amine by product
2 H₂O
Forming aromatic amine ( Image)
Amide structure ( Image)
CON bond
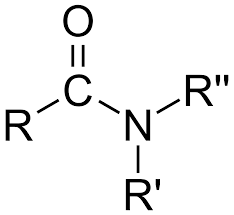
Amide structures ( Image)
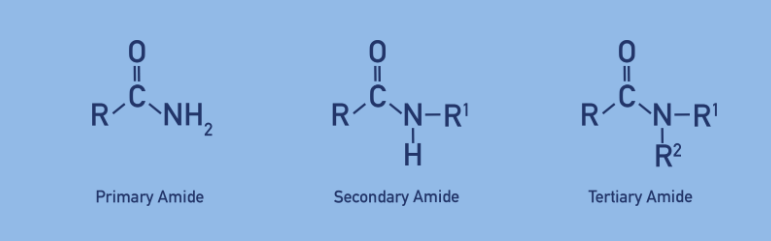
How to name primary amides
look at alkyl group then add amide
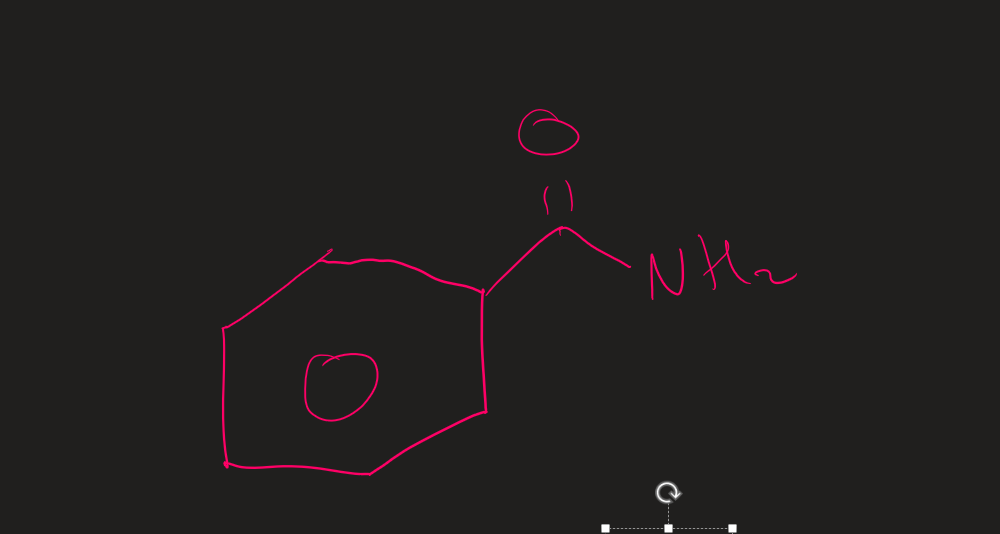
Name this structure
Benzamide
How to name secondary amines
Add N then Group attached to carbonyl = Stem + amide
R Group directly attached to N → Prefix
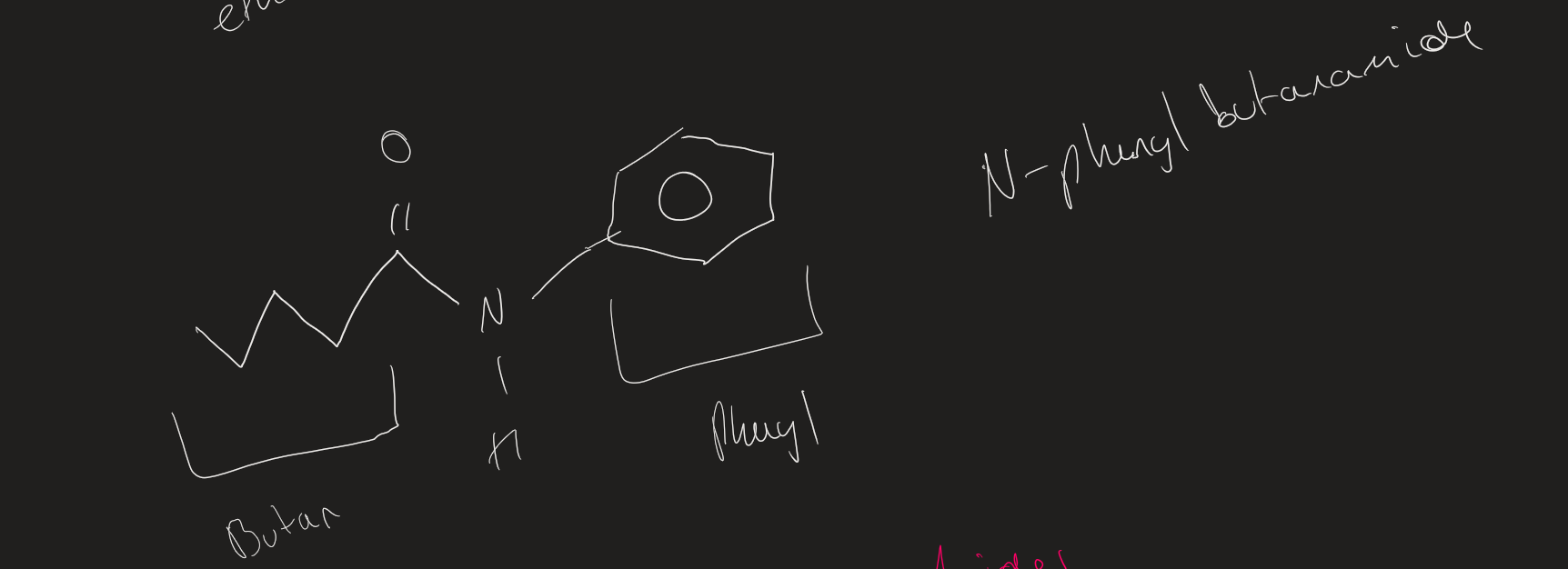
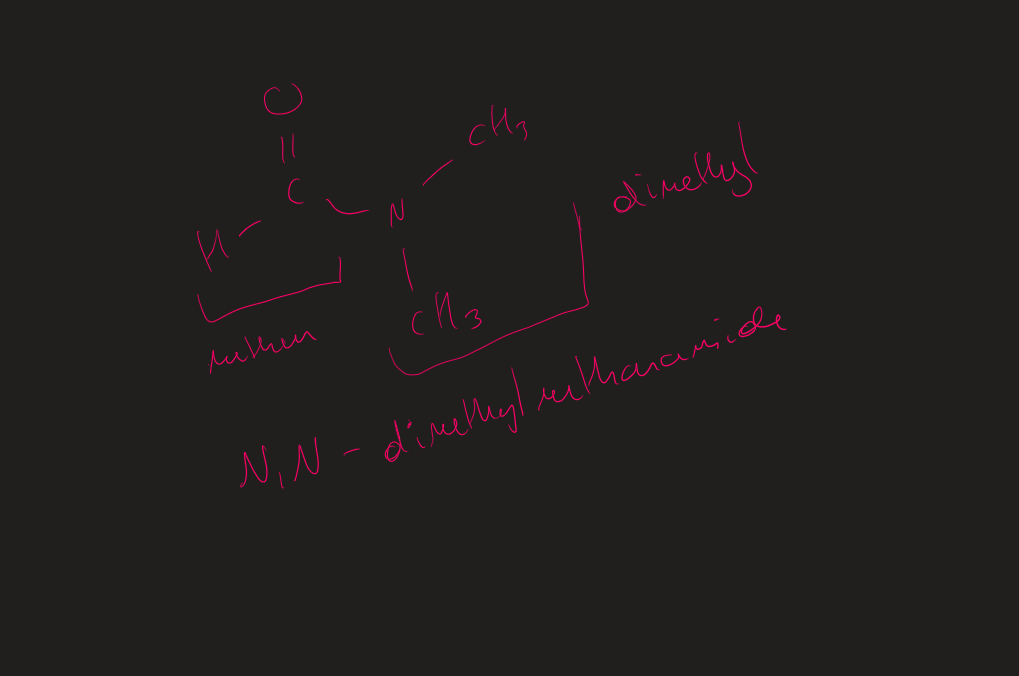
How to name tertiary amides
N,N then R group attached to N → prefix ( alphabetical order)
R attached to carbonyl = stem + amide
Primary to secondary amine mechanism
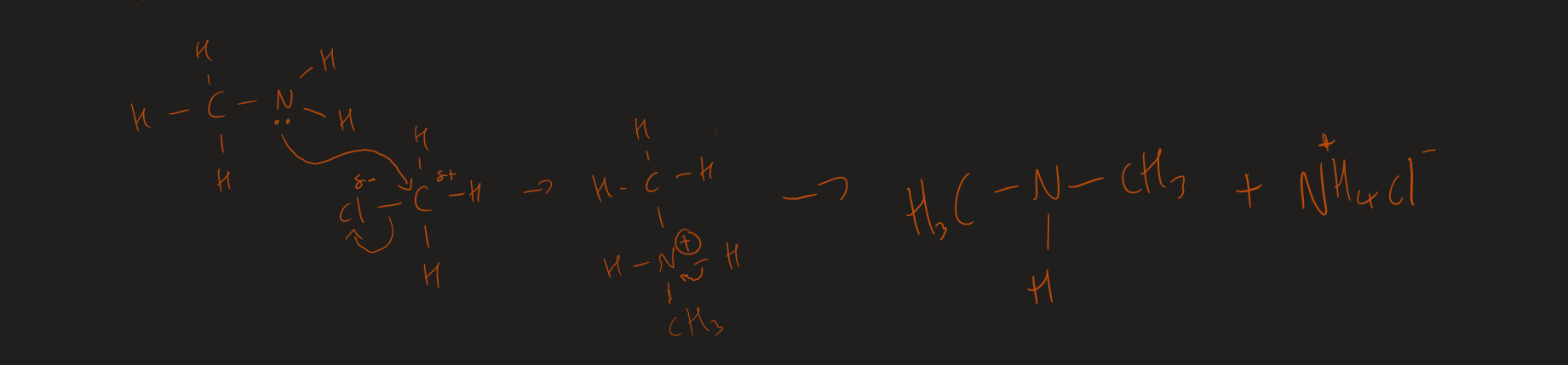
what are the ways to make a Amine
Haloalkane → Nitrile → Amine
Haloalkane + Excess NH₃ dissolved in ethanol
Acyl chloride + NH₃
Advantages of Nitrile to amine
makes only 1 product ( amine)
don’t have to purify mixture of products
How to form amine from Haloalkane ( Nitrile method)
React with KCN ( dissolved in ethanol) to form a nitrile ( 1 more carbon)
react nitrile with 2H₂ → Amine
Conditions for nitrile to amine
React with 2H₂
Nickel catalyst
ways to make a amide
Acyl chloride / acid anhydride + amine → secondary amide
what are optical isomers
Form non- imposable mirror images
Reagants and conditions for Nitrile to carboxylic acid
H₂O / HCl
Number of structural isomers ( Image)
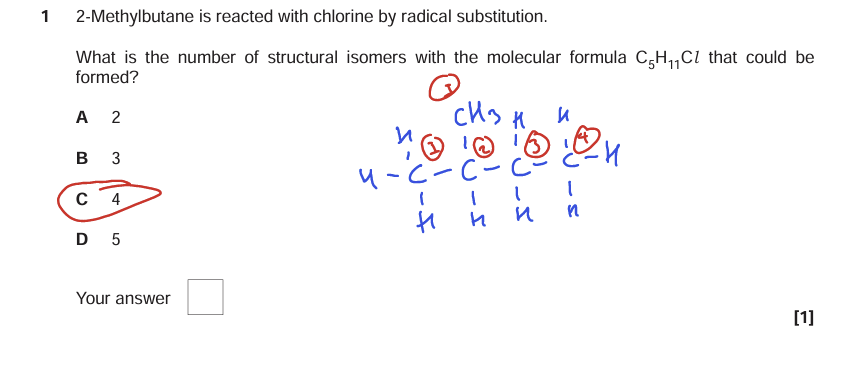
what are the types of polymers
Addition
Condensation
Difference between addition and condensation polymers
Condensation → Biodegradable
Addition → Non -biodegradable
Carboxylic acid + Amine →
Carboxylic acid + Amine → Amide + H₂O
what are the 2 monomers of polyester
Dicarboxylic acid + Diol → polyester + H₂O
hydroxycarboxylic acid + hydroxycarboxylic acid + H₂O
Product of all acyl chloride reactions
HCl
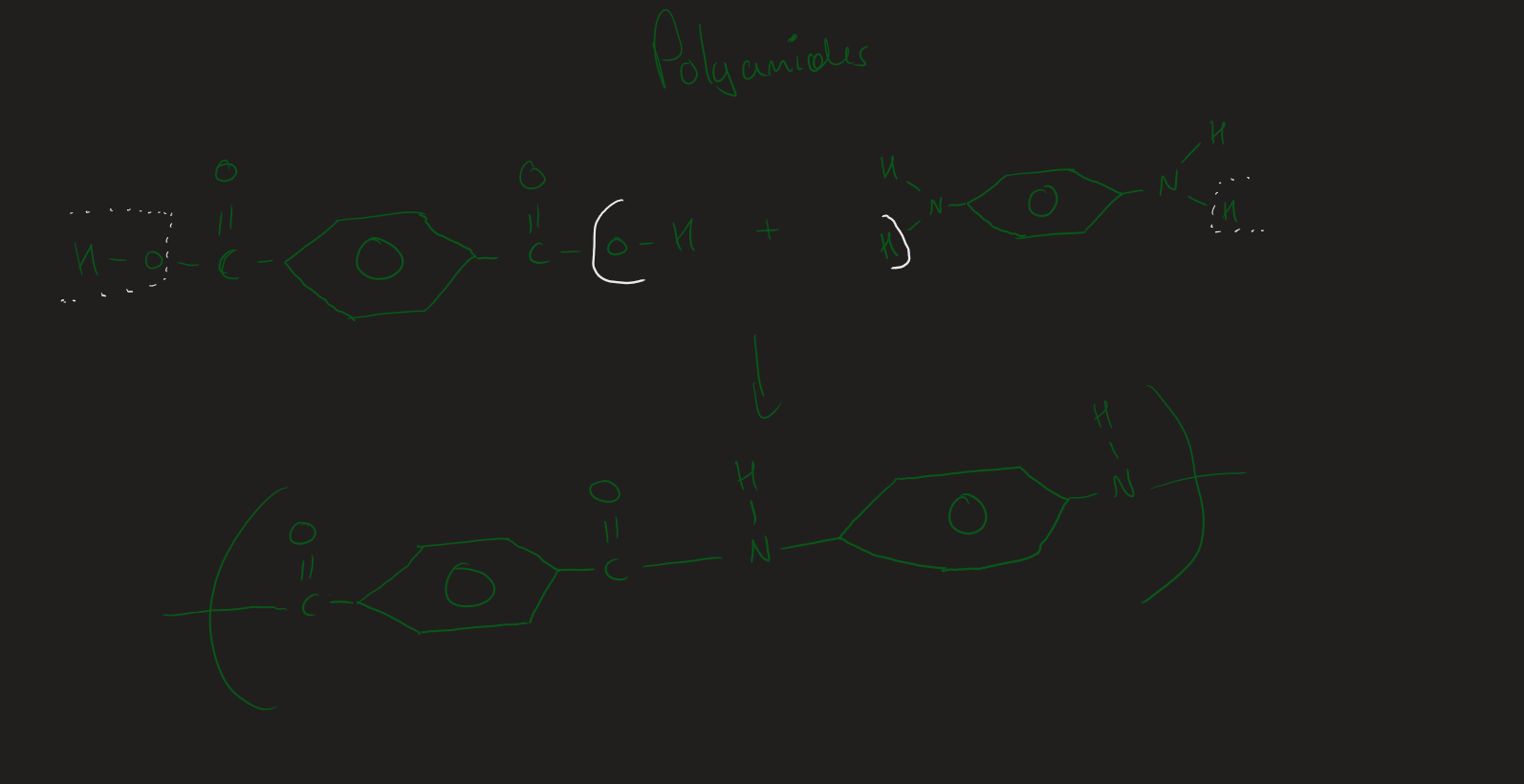
what are the 2 monomers of polyamide
Amino acid + Amino acid
Dicarboxylic acid + Diamine
what are the polymers of amino acids
Polypeptide
Dicarboxylic acid + Diol reaction explained
Diol → loses H
Dicarboxylic acid → Loses OH
Dicarboxylic acid + Diamine reaction explained
Diamine → loses a H
Dicarboxylic acid → loses OH
Hydroxycarboxylic acid (image)
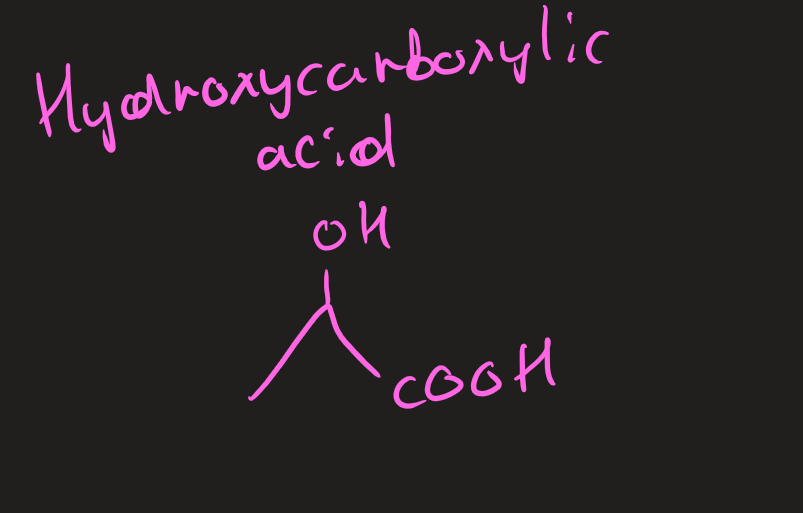
General formula for making polymer
n Reactant + n Reactant → polymer n + 2n-1 H₂O
Making polyester ( Image)
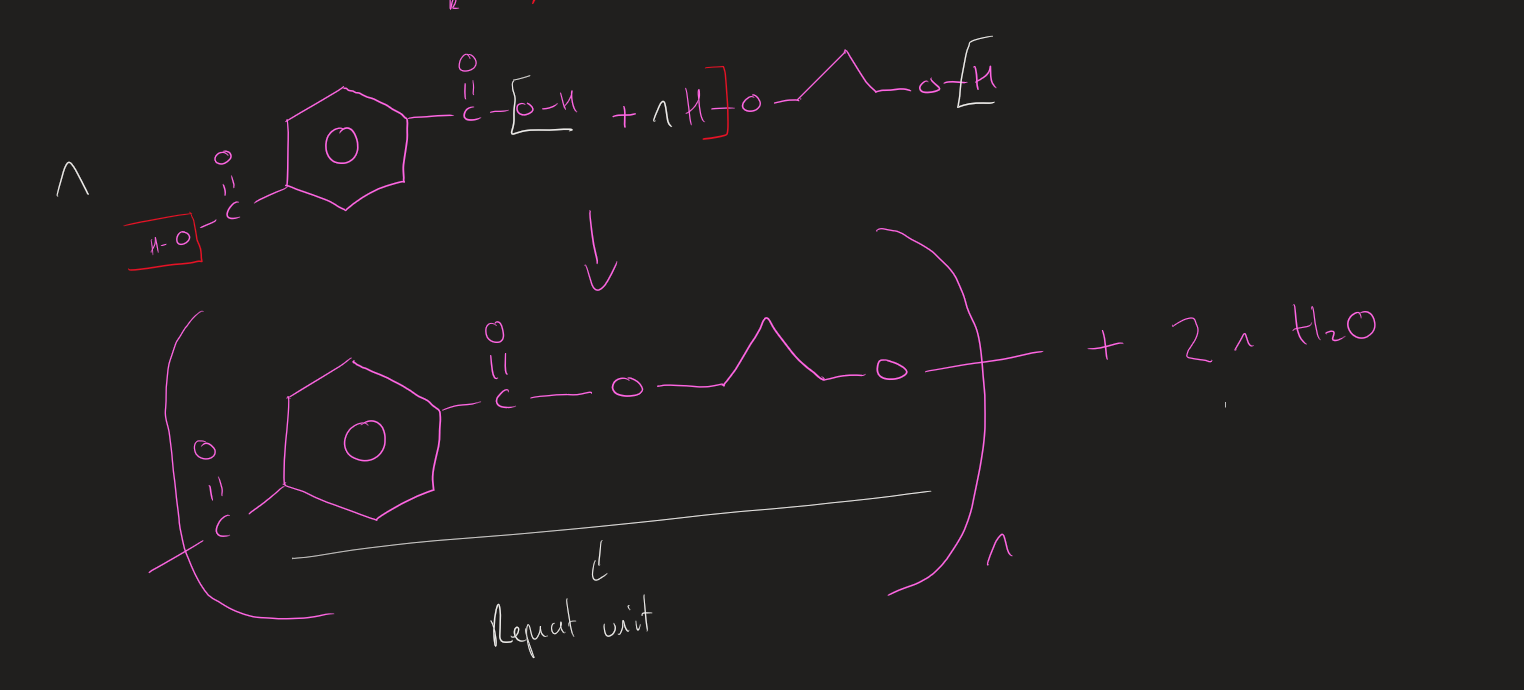
How to work out monomers of Polyester
Break through ester bond
monomers → Diol + Dicarboxylic acid
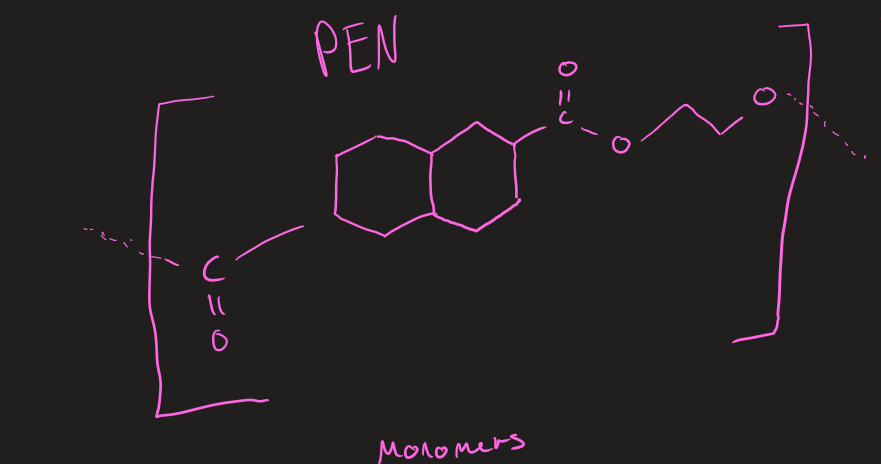
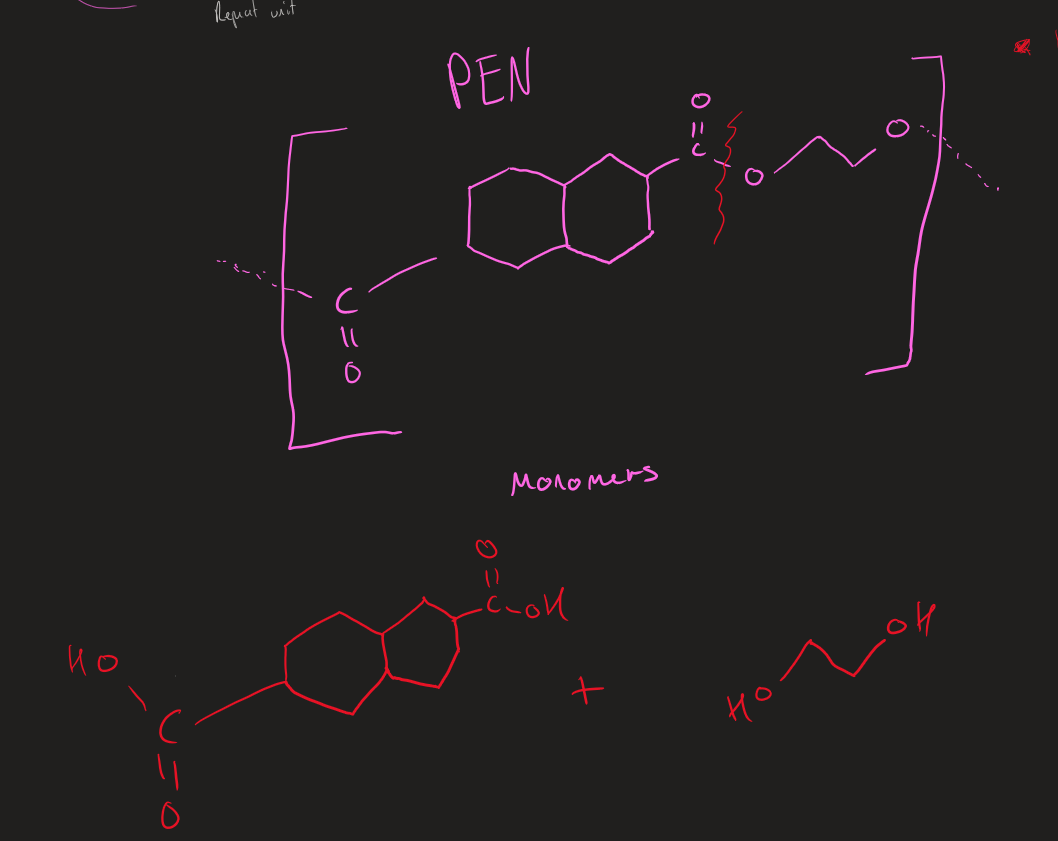
Monomers of PEN
Dicarboxylic acid + Diamine reaction explained
Diamine → Loses H
Dicarboxylic acid → loses OH
Forming polyamide ( Image)
Hydrolysis of polymers general equation ( Image)

disadvantages of water in hydrolysis
Too slow
use Hot aqueous acid / alkali
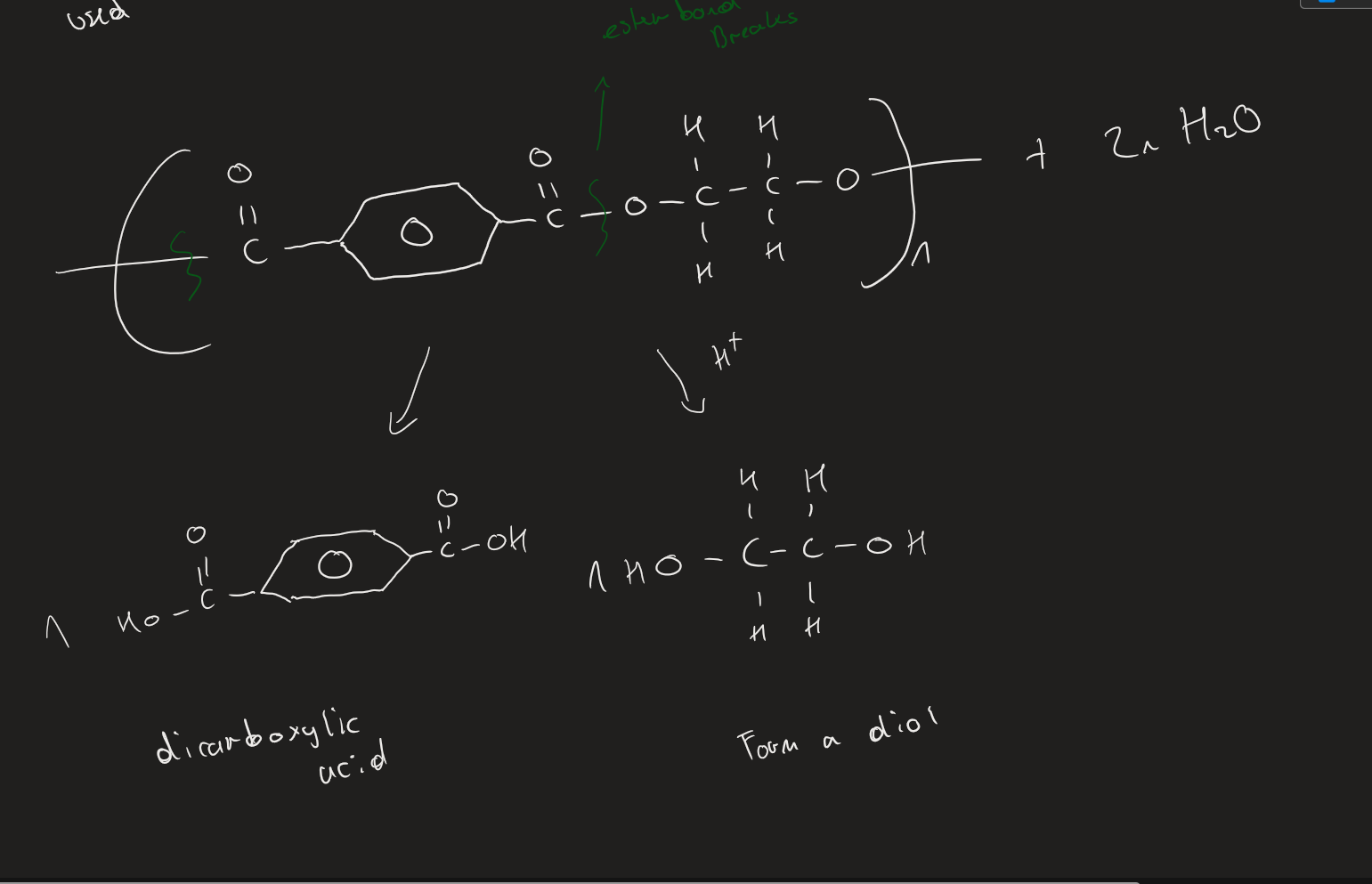
Products of acid hydrolysis on polyester
Dicarboxylic acid + Diol
Polyester acid hydrolysis ( Image)
Reactants in hydrolysis reaction
(Polymer) n + n H₂O
Monomer of acid hydrolysis of polymer with one ester bond
amino acid
hydroxycarboxylic acid
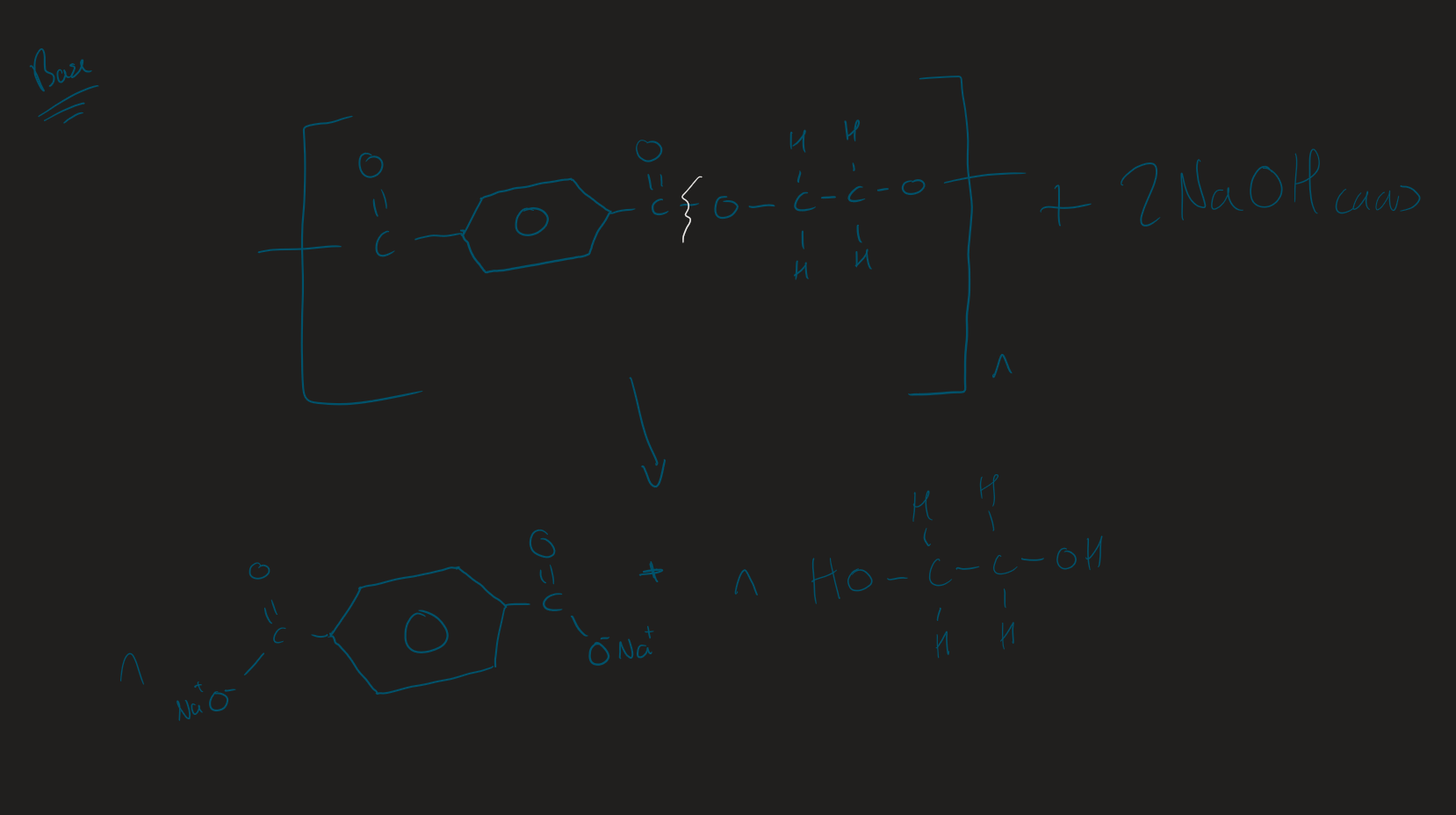
Products of alkali hydrolysis on polyester
Dicarboxylate salt + Diol
Base hydrolysis polyester ( Image)
what are the types of Hydrolysis
Acid
Base
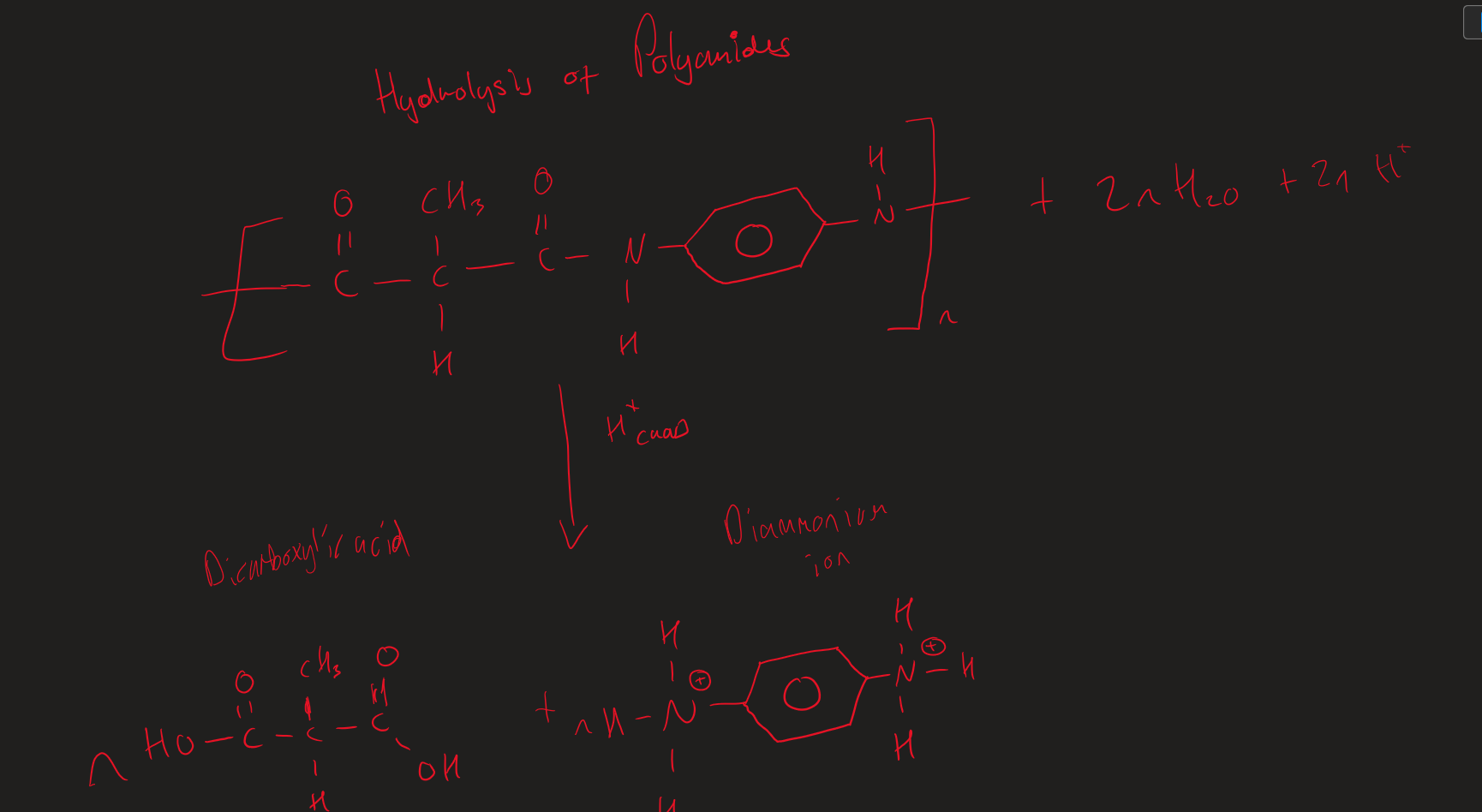
products of the acid hydrolysis of Polyamides
Dicarboxylic acid
Diammonium ion
Acid hydrolysis of polyamides ( Image)
what is meant by a chiral centre
Carbon has 4 different groups attached
when are carbons never chiral
Two or more hydrogens attached
C=C
How many optical isomers do molecules with 1 chiral centre have
2 optical isomers
How many optical isomers do molecules with 2 chiral centres have
4 optical isomers
Which molecules are always chiral
Amino acids
What are Enantiomers
Optical isomers
How to work out number of optical isomers a chiral centres form

Is benzene chiral?
No
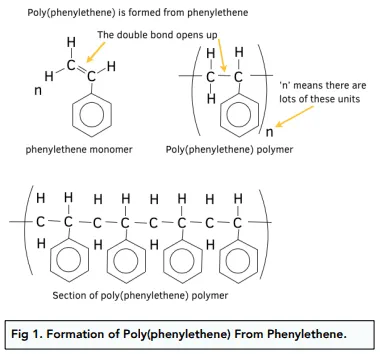
addition polymers (image)
everything revolves around broken C=C bond
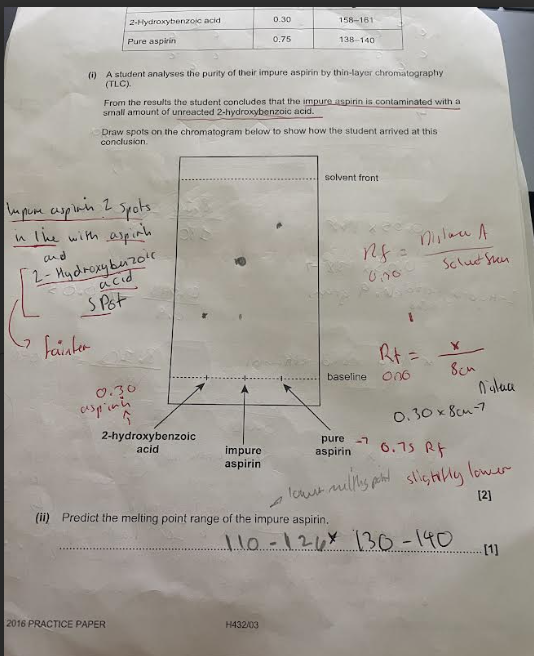
TLC question on Aspirin image
melting of pure aspirin was 138 - 140
impure 130 - 140
What does the Amine group ( NH₂) contain
Hydrogen bonding