Organic Chem: Amines and Amides
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
What is an Amine
Ammonia molecule that has 1 or more hydrogens replaced by alkyl group (hydrocarbons)
General formula: CnH2n+3N
Types of amines
Primary: 1 alkyl group attached to the N
Secondary: 2 alkyl groups attached to N
Tertiary: all hydrogens replaced by alkyl groups
What are amides
Carbonyl group bonded with an AMINE
General formula: CnH2n+1NO
How are amines made?
Made from alkyl halide and ammonia:
CH3CH2I + NH3 —→ CH3CH2NH3 +HI
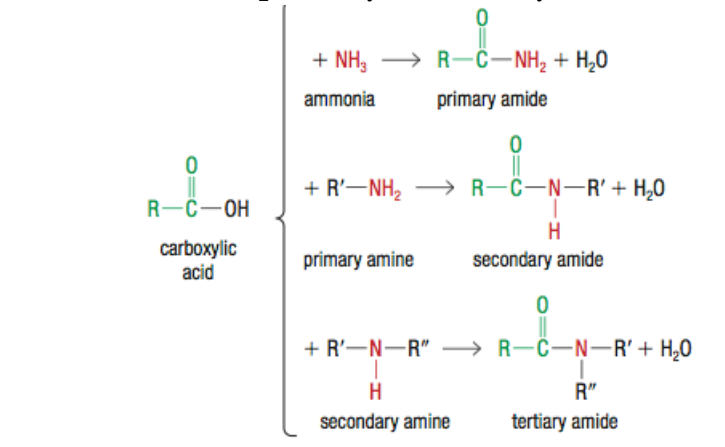
How are Amides made?
Through a condensation rxn between a carboxylic acid and ammonia or primary and secondary AMINES ONLY
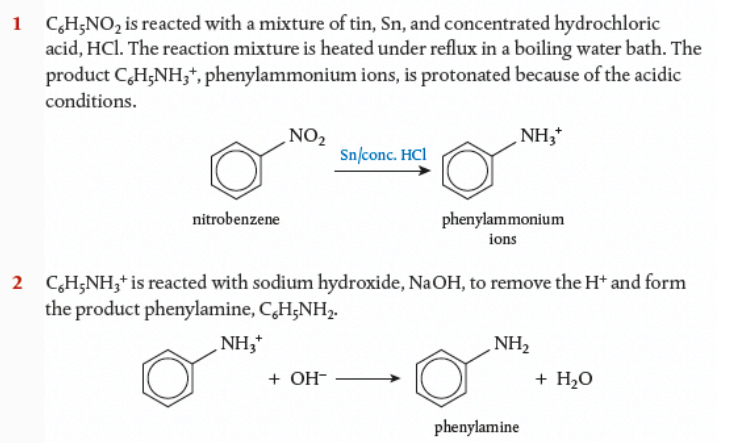
Reduction of nitrobenzene
Nitrobenzene, C6H5NO2, can be converted into phenylamine, C6H5NH2 in a 2-stage reduction process:
Primary Amide?
Carboxylic acid + NH3 (ammonia)
Secondary Amide
Carboxylic acid + primary amine
Tertiary amide
Carboxylic acid+ secondary amine