WJEC AS Biology Unit 1.4 - Enzymes and Biological Reactions
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
65 Terms
Metabolism
Series of enzyme controlled reactions
Combination of anabolic and catabolic reactions that are catalysed by enzymes
Anabolic reactions
Reactions which construct molecules from smaller units, eg protein synthesis
Catabolic reactions
Reactions when break down molecules into smaller units, eg digestion
Enzyme
Biological catalysts
Speed up reactions without being changed themselves
Found in living things
All are proteins
Often end in -are, e.g. amylase
Catalyst
An arm or molecule that alters the rate of a chemical reaction without taking part in the reaction or being changed by it
Substrate
The substance on which an enzyme acts
Enzyme-substrate complex
Intermediate structure formed during an enzyme-catalysed reaction in which the substrate and enzyme bind temporarily, such that the substrates are close enough to react
Forms a product
Product
The substances that are made, e.g. maltose etc.
Active site
The specific three-dimensional site on an enzyme molecule to which the substrate binds by weak chemical bonds. Formed by amino acids
Specific
Only one type of substrate can bind
Denaturation
The breakdown of the bonds that hold the protein together, including the active site, in shape
This changes the 3D structure of the enzyme incl the active site so the substrate can no longer bind
Activation energy
The minimum energy that must be put into a chemical system for a reaction to occur
Collision theory of enzyme reactions
For molecules to react, they have to collide with energy to break and form bonds
Lock and key model
The specific, unique shape of the active site means that an enzyme can only catalyse one type of reaction
Induced fit model
Where changed of shape of both active site and substrate being reactive groups of enzyme and substrate close to each other, weakening bonds in the substrate so the reaction takes places at a lower activation energy
Sites of enzyme action
Extracellular (inside cells); some enzymes are secreted from cells by exocytosis and catalyse extracellular reactions
Intracellular (outside cells);
In solution; intracellular enzymes may act in solution inside cells
Membrane-bound; intracellular enzymes may be attached to membranes such as on the crustal of mitochondria
Catalysis
The lowering of activation energy
Metabolic pathway
A sequence of enzyme-controlled reaction in which a product of one reaction is a reactant in the next
Many enzymes can be close together and can catalyse separate reactions in a series
Structure of enzymes
Globular proteins
Proteins with tertiary structure
Properties of enzymes
Speed up reactions
Not used up
Not changed
Catalyse many reactions per second
Only catalyse reactions that are energetically favourable and would happen anyway
Protein nature of enzymes
Globular proteins
Proteins with tertiary structure
The protein chain folds into a spherical or globular shape with hydrophilic R groups on the outside of the molecule, —> enzymes = soluble
Each enzyme has a particular sequence of amino acids
The elements in the R groups determine the bonds the amino acids make with each other
These bonds (eg hydrogen bonds, ionic bonds, disulphide bridges) hold the enzyme molecules in its tertiary form
Small area with specific 3D shape = active site which gives enzyme many of its properties
Effect of temperature on enzyme controlled reactions
Increased temp —> increased KE of enzyme + substrate molecule; collide with enough energy more often, increasing rate of reaction
Above optimum, molecules have more KE but rate goes down as increasing vibration breaks H bonds, changing tertiary structure
Alters shape of active site and substrate will not fit. Enzyme = denatured, a permanent change in structure
Low temps, enzyme = inactivated as molecules have v low KE. Shape = unchanged and enzyme will work again if the temp is raised

Effect of pH on enzyme controlled reactions
At optimum, rate = highest
Small changes around the optimum cause small reversible changes in enzyme structure and reduce its activity, but extremes of pH denatured enzymes
Changes on amino acid side chains of active site age affected by hydrogen ions or hydroxide ions
Low pH, excess H ions = attracted to - charges + neutralise them
High pH, excess OH ions neutralise + charges
Disrupts ionic and H bonds maintaining shape of active site
Shale changes, denaturing the enzyme
No e-s complexes form + enzyme activity is lost

Effect of enzyme concentration when substrate is unlimited
product leaves active site, enzyme molecule can be reused, so only low enzyme conc is needed to catalyse a large number of reactions
Number of substrate molecules that one enzyme molecule can turn into products in a given time is the turn-over number
As the enzyme conc increases, there are more active site available + for increases
Effect of substrate concentration when enzyme concentration is limited
if enzyme conc is constant, rate increases as substrate conc increases
At low substrate conc the enzyme molecules have only a few substrate molecules to collide with. Active sites are not colliding to full capacity
With more substrate, more active sites filled
Conc of substrate is controlling the ror and so is a limiting factor
More substrate is added, at a critical conc, all the active sites become occupied and rate is at its max
Active sites all full, enzyme is saturated
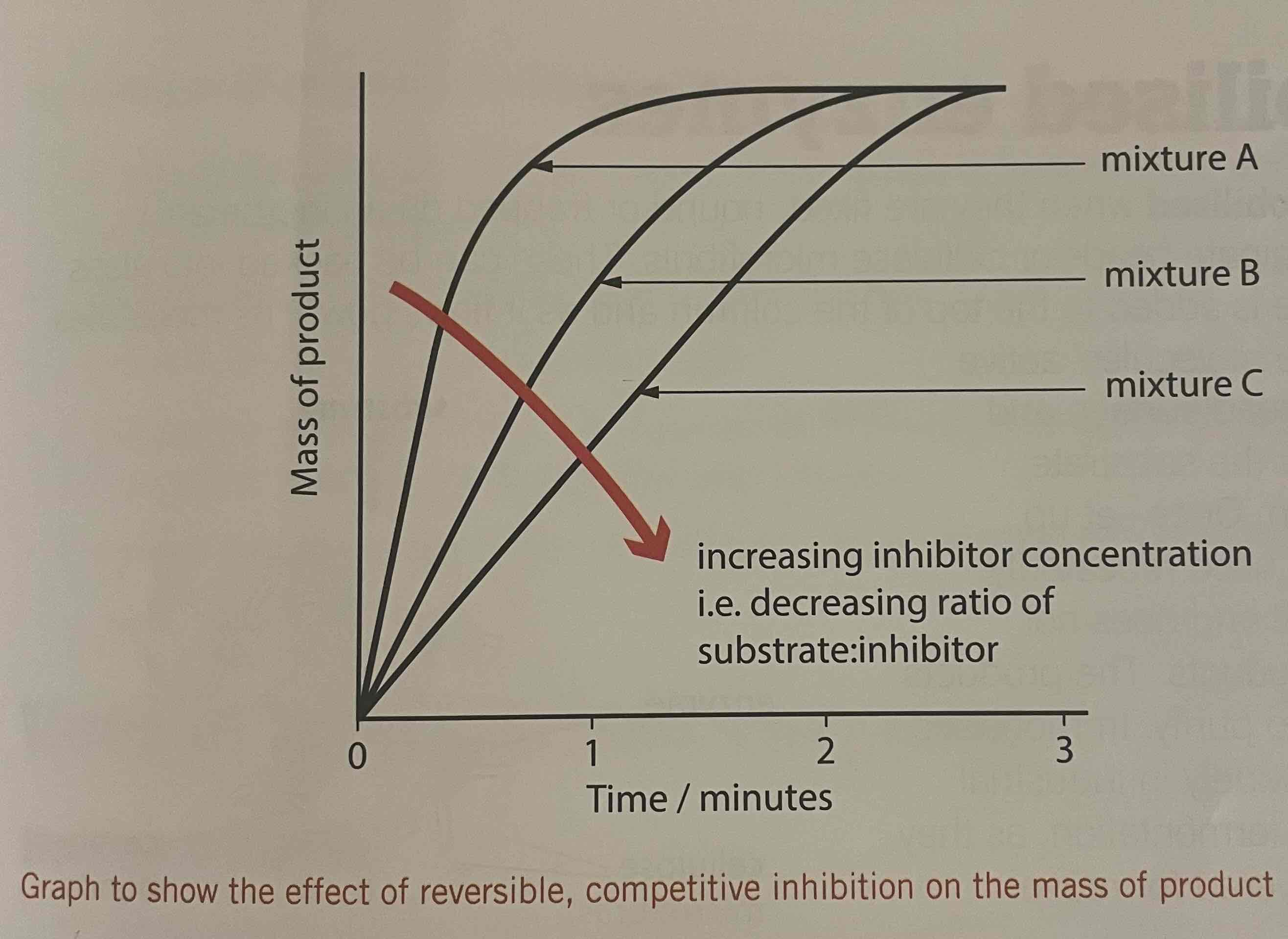
Effect of competitive inhibitors
the greater the substrate conc, in relation to inhibitor, the greater the chance that the substrate and enzyme will collide
No reaction takes place to form products
Effect is overcome when the substrate is in excess
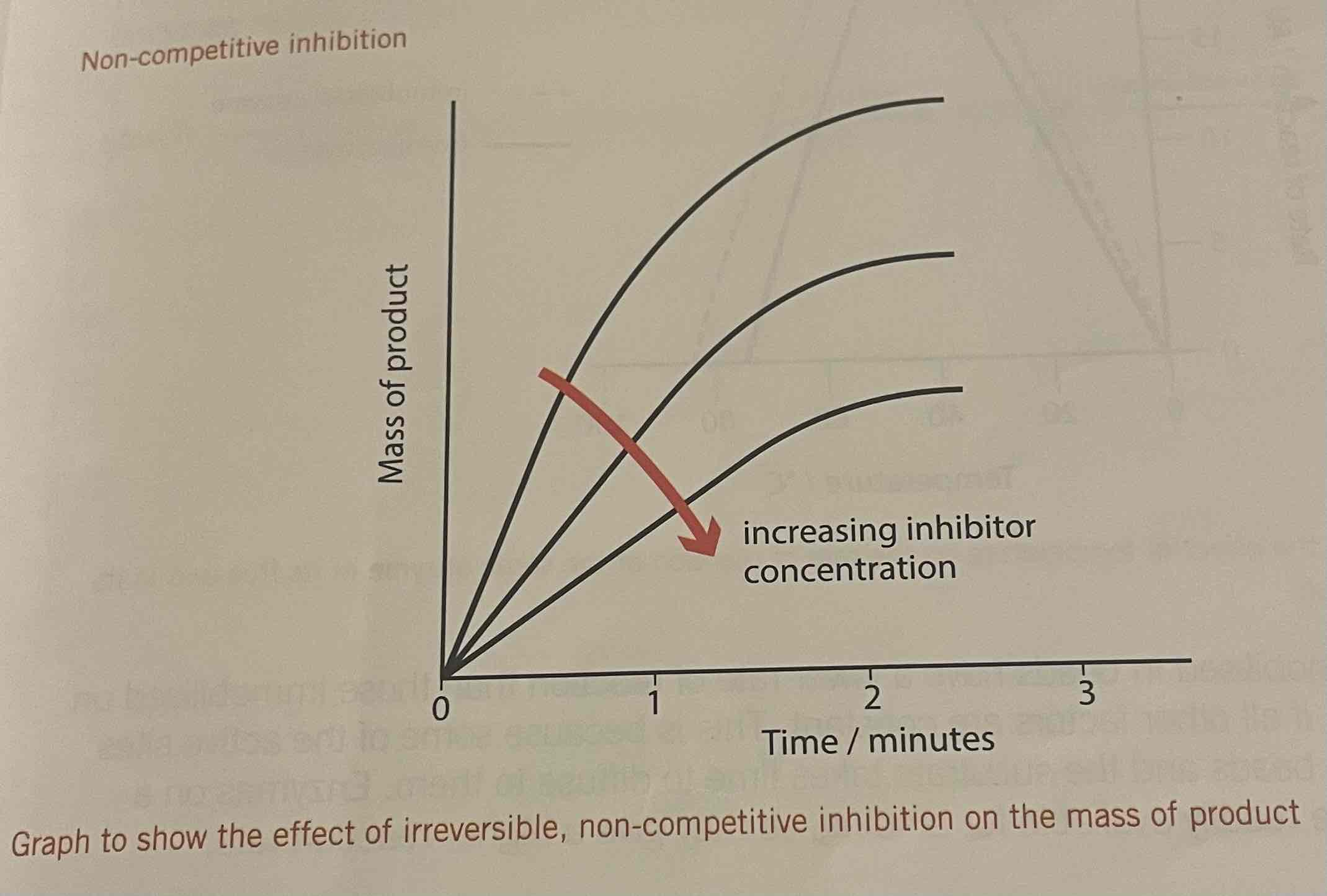
Effect of non-competitive inhibitors
as inhibitor conc increases, the rate of reaction and final mass of product decrease
Enzymes and activation energy
Modify the substrate so the reaction requires a lower activation energy
Inhibitor
A molecule or ion that binds to an enzyme and reduces the rate of reaction the enzyme catalyses. Can be reversible or irreversible
Competitive inhibition
Reduction of the rate of an enzyme-controlled reaction by a molecule or ion that has a complementary shape to the active site, similar to the substrate, and binds to the active site, preventing the substrate from binding
Have a molecular shape complementary to the active site and similar to that of the substrate
Fits in enzyme active site —> e-I complex, blocking active site
Prevents binding of substrate
Inhibitor and substrate compete for active site
Substrate molecules unable to occupy active site
No reaction takes place to form products
Effect of increasing substrate concentration on competitive inhibition
the greater the chance of a substrate binding to the active site
Leaves fewer available for the inhibitor
If the inhibitor conc increases, it binds to more active sites to reaction rate is slower
Non-competitive inhibition
An atom, molecule or ion that reduces the rate of an enzyme-controlled reaction by binding to the enzyme at a position other than the active site, altering the shape of the active site and preventing the substrate from successfully binding to it
Bind to enzyme somewhere other than active site - an allosteric site
Substrate and inhibitor do not compete with the substrate
Affect bonds within enzyme molecule and alter its overall shape, including that of the active site, distorts shape of active site
Substrate cannot bind with the active site, and no enzyme-substrate complexes form
If substrate conc is increased, the degree of inhibition is unaffected
Rate of reaction is always lower when nci is present
Effect of increasing substrate concentration on non-competitive inhibition
No effect
End-point inhibition
product of enzyme-controlled reaction
Acts as an inhibitor
Binds to enzyme, slowing down its own production using negative feedback
Immobilised enzymes
Enzyme molecules bound to an inert material, over which the substrate molecules move
Can be packed into glass columns
Substrate is added to the top of the column and as it flows down, its molecules bind to the enzyme molecules’ active sites, both on the bead surface and inside the beads as the substrate molecules diffuse in
Column can be used repeatedly
Enzyme is fixed and does not contaminate products, therefore easy to purify
Surface area of immobilised enzymes
Large beads —> smaller total surface area then if the same volume had been used to make small beads
Smaller beads; substrate molecules will have easier access to enzyme molecules and so they will produce a higher rate of reaction
Effect of immobilisation of enzymes on rate of reaction
Makes enzymes more stable as it creates a micro environment allowing reactions to occur at higher temperatures or more extreme pHs than normal
Prevents shape change that would denature its active side so the enzyme can be used in a wider range of physics conditions
Enzymes immobilised in beads = lower rate of reaction than those immobilised on a membrane, if all other factors are constant
Some of the active sites are inside the beads + the substrate takes time to diffuse them
On a membrane = readily available for binding, so give a higher rate of reaction
Benefits of immobilised enzymes overall
Increased stability and function over a wider range of environments that enzymes free in solution
Products are not contaminated with the enzyme
Enzymes are easily recovered for reuse
A sequence of columns can be used so several enzymes with differing pH or temp optima can be used in one process
Enzymes can be easily added or removed, giving greater control over the reaction
Benefits of immobilised enzymes in industry
Separated easily from products and reused
More thermo- and pH-stable
Several enzymes can be used in the same enzyme continuous fermenters (reactors) despite having different optimum condition
Products can be removed continuously
Products are pure and free of enzyme
Enzymes in biosensors
Turn a chemical signal into an electrical signal
Rapidly + accurately detect, identify and measure even very low concentrations of important molecules - high sensitivity
Enzymes = specific and are able to select one type of molecule from a mixture, even at very low concentrations
Example; detection of blood glucose
Enzymes can be immobilised onto test strips, where different strips may detect a variety of molecules
Uses of immobilised enzymes
Lactose-free milk; milk is passed down a column containing immobilised lactase. The lactose binds to the active sites on the lactase and is hydrolysed to its components
Biosensors
High-fructose corn syrup (HFCS) manufacture
What is a catabolic reaction?
What is an anabolic reaction?
What is a catalyst?
What biological polymer are enzymes made out of?
What is the difference between an intracellular and extracellular enzyme?
Describe the lock and key theory of enzyme action
Describe what is meant by induced fit
Give an example of an enzyme that works by induced fit
Explain how the shape of the active site make enzymes specific to their substrate
What is the name of the energy lowered by enzymes that increases the rate of reaction?
Explain how increasing temperature increases the rate of reaction up to the optimum.
What is meant by optimum?
Explain how increasing temperature above the optimum decreases the rate of reaction.
What is meant by denaturation?
Describe the effect of small changes in pH away from the optimum.
Describe the effect of large changes in pH away from the optimum.
Describe the difference between competitive and non-competitive inhibition.
Sketch a graph to show the effects of increasing substrate concentration on the rate of an enzyme controlled reaction
Sketch a graph to show the effects of increasing substrate concentration on the rate of reaction affected by a competitive inhibitor
Sketch a graph to show the effects of increasing substrate concentration on the rate of reaction affected by a non-competitive inhibitor.
Explain the difference between the graphs for non-competitive and competitive inhibitors.
List two advantages of using immobilised enzymes in industry.
What are the advantages of biosensors?
Give an example of a biosensor.