Introduction to Organic Chemistry
1/57
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
Macromolecules
These are large molecules that are necessary for all life processes
Carbohydrate
These macromolecule is made up of carbon, hydrogen and oxygen
Saccharide
The unit structure of carbohydrates.
Monosaccharide
They are simple sugars such as glucose
Disaccharide
They are chains of two simple sugars such as lactose
Polysaccharide
they are chains of more than two simple sugars such as cellulose
Nucleic Acids
They are polymers made from the monomers called nucleotides. Examples are DNA and RNA
Effects of Impurities
They increase the boiling point and reduce the melting point
Melting points of impure compounds
They have a wider melting point range that is relatively lower
Melting points of pure compounds
they have a smaller melting point range that is relatively higher
The higher the molecular weight...
...the higher the boiling point.
What is Boiling point
It is the temperature at which vapor pressure of a liquid equals the atmospheric pressure.
Boiling sticks/Stones/Chips
Used to prevent super heating.
For an impure substance...
...The temperature during boiling/melting does not remain constant.
Refractive index "n"
The measure of how much a ray of light bends as it travels from one medium to another.
Formula for refractve index
Speed of light in vacuum/speed of light in given sample
Abbe-3L refractometer
this is used for measuring the refractive index of a sample.
Factors that influence the choice of a purification method
Stability of the compound. Amount of impurities present. Physical state of the compound. Nature of the compound.
Purification of compounds
The separation of the components of a compound and the removal of impurities.
Purification Method
Distillation. Re crystallization. Sublimation. Chromatography. Extraction.
Organic Chemistry
The study of all carbon compounds and the reaction it undergoes in exception of carbonates (CO32-), cyanide (CN-) and oxides
Catenation
The property of certain elements, such as, carbon to form rings, chains or branched structures with other atoms of the same element
The ability catenate....
...decreases down the group
Elements with the Catenation property
Carbon, Silicon, Gallium, Tin, Lead (all group 4 elements)
If there are four atoms bonded to a carbon atom e.g CH4 (Methane)
The shape is tetrahedral. Bond Angle is 109.5 degrees
If there are three atoms bonded to the central atom e.g C2H2 (Ethene)
The shape is trigonal Planar. The bond angle is 120 degrees
If there are two atoms bonded to the central atom e.g C2H2 (Ethylene)
The shape is linear. Bond angle is 180 degrees
Source of Formic Acid CH2O2
Ants, Sting of bees
Source of Ethylene C2H4
Plants
Use of Ethylene C2H4
Promotes ripening of fruits
Examples of Macromolecules
Protein, Carbohydrate, Nucleic Acids.
Metabolism
this refers to chemical reactions in cells that change food into energy
Proteins
Macromolecules that are polymers of amino acids.
Chromatography
a technique for the separation of a mixture by passing it through a stationary phase in which the components move at different rates while being carried by the mobile phase
In Chromatography, the component that travels the least distance…
…is the least soluble/polar to the solvent
In Chromatography, the component that travels the most distance…
…is the most soluble/polar to the solvent
Theory of Chromatography
Solubility of the compounds in the mixture
It uses the different polarities of the stationary phase and mobile phase
Compounds attracted to the stationary phase will move slower than compounds attracted to the mobile phase.
common substances used in the stationary phase of chromatography
Silica gel, and alumina
Substance used in the mobile phase
any solvent that can move through the stationary phase and carry components of the compound.
Retardation Factor
(distance traveled by solute) / (distance traveled by solvent)
Types of chromatography
Gas chromatography
Thin-layer chromatography
Column chromatography
Column chromatography
The chromatography method used to separate a single compound from a mixture.
Gas Chromatography
The chromatography method used to separate small quantities of volatile mixtures
Some carrier gas used in Gas Chromatography
Helium and Nitrogen
High Pressure Liquid Chromatography (HPLC)
A chromatographic technique that uses high pressure to separate and analyze compounds in a liquid sample based on their interaction with a stationary phase.
Thin Layer Chromatography (TLC)
an affinity-based chromatography method used to separate compounds in a mixture.
Uses of Chromatography
For Identifying poisons and drugs
To determine the purity of a given substance
To detect traces of unlawful dyes and other additives in foodstuffs.
Sublimation
is the process where a substance transitions directly from a solid to a gas without passing through the liquid phase.
Distillation
This is the process used to separate components in a liquid mixture through a selective evaporation and condensation method.
Types of Distillation
Simple Distillation
Vacuum Distillation
Fractional Distillation
Steam Distillation
Simple Distillation
The distillation method used when the boiling points of the samples vary widely, usually greater 25 degree Celsius
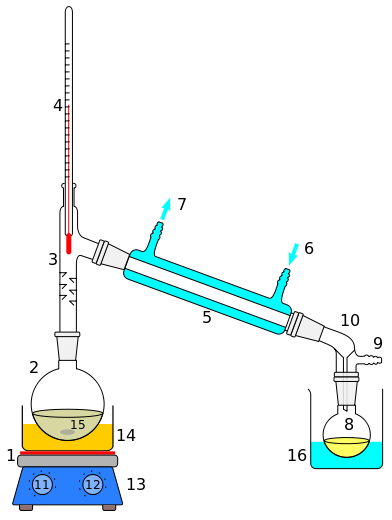
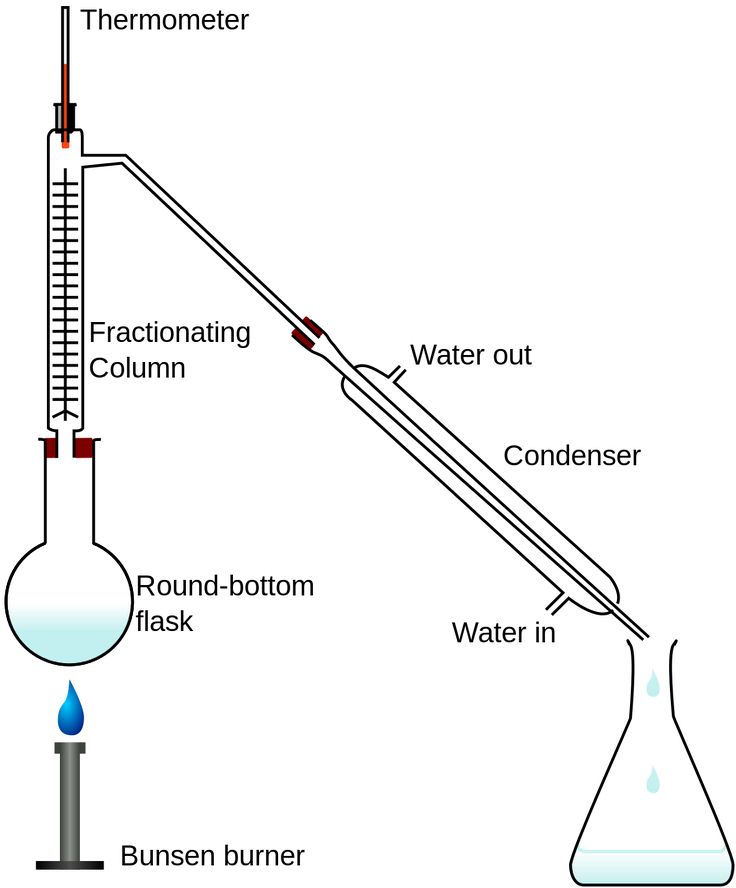
Fractional Distillation
This distillation method is used when the difference in the boiling points of the samples is very small.
Ways to increase the efficiency of Fractional Distillation
Increasing the length of the fractionating column
Increasing the surface area of the beads in contact.
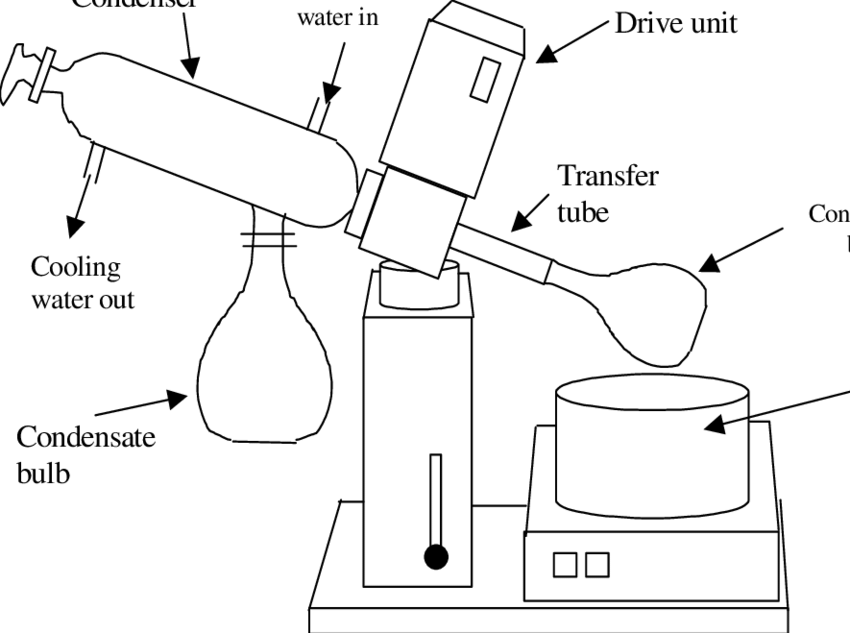
Vacuum Distillation
The distillation method used to separate samples that decompose before reaching their boiling points by decreasing their boiling points.
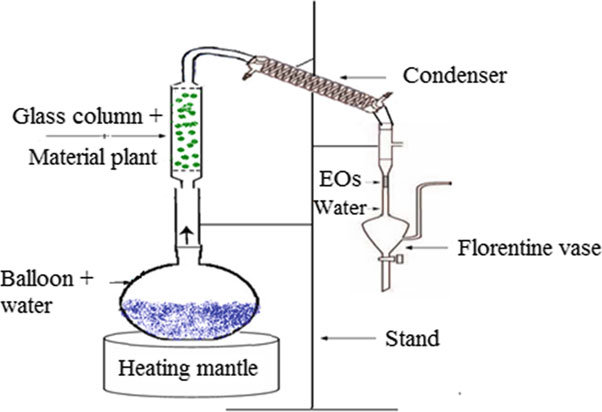
Steam Distillation
This distillation is used for separating samples that are heat sensitive and are nearly insoluble in water
Heat sensitive substances…
…decompose before reaching their boiling points.
The method used to extract fragrant compounds for perfumes, essential oils, and those used in food processing and skincare products is…..
…Steam Distillation.
Recrystallization
Purification method to get super clean crystals!
Dissolves impure solid in hot solvent, then cools it down.
The pure stuff forms crystals, leaving gunk behind in the solution.