C5: energy changes
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
What is an exothermic reaction? Give examples.
They release energy to the surroundings, so the temperature of the surroundings increases.
EXAMPLES: combustion, many oxidation reactions, neutralisation, self-heating cans, hand warmers
What is an endothermic reaction? Give examples.
They take in energy from the surroundings, so the temperature of the surroundings decreases.
EXAMPLES: thermal decompositions, citric acid + sodium hydrogencarbonate reaction, some sport injury packs
When it comes to endo and exothermic reactions, what does the thermometer measure?
Temperature of the surroundings
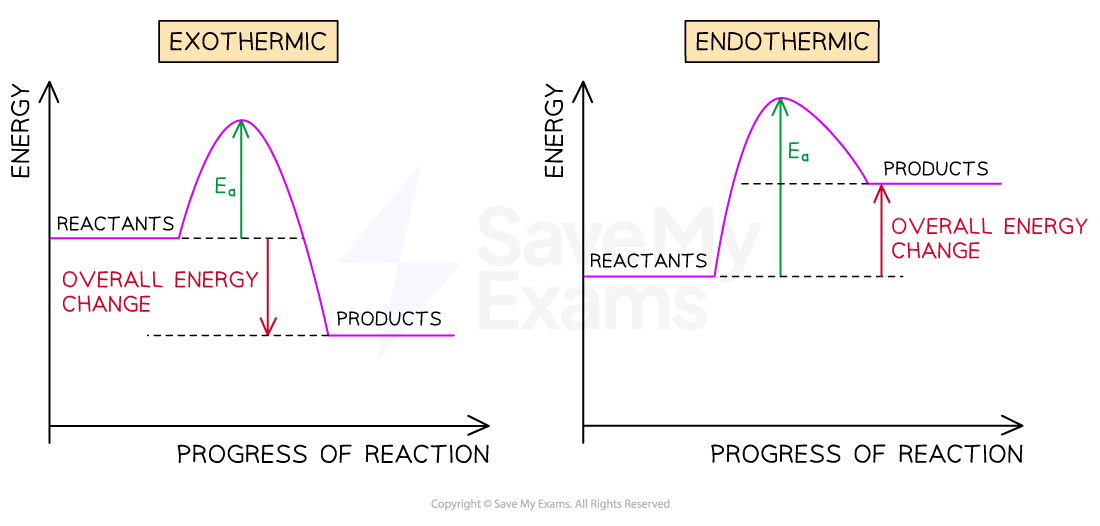
What do reaction profiles for endo and exothermic reactions look like?
Ea meaning activation energy.
Be able to do bond energy calculations.