Module 3: Ceramics
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
97 Terms
Atomic bonding in ceramics:
What 4 factors influence atomic bonding in ceramics?
Electronegativity
Ionic/covalent character
Ionic radius and relative sizes of cations and anions
Coordination numbers
Factor 1: Electronegativity (1 of the factors which influences atomic bonding in ceramics)
What is electronegativity? Do metals have a high or low electronegativity?
Electronegativity is a measure of an atom's ability to attract shared electrons to itself.
Metals have a low electronegativity.
True or false: ceramics must have an overall positive charge
False, ceramics must have an overall neutral charge
Factor 2: Ionic/Covalent character (1 of the factors which influences atomic bonding in ceramics)
What does atomic bonding in ceramics look like?
Ranges from purely ionic
To totally covalent
Combination of the two
What does an ionic crystal structure look like?
Cations (+ve) give valence electrons to anions (-ve)
Factor 3: Ionic Radii and Cation/Anion ratio (1 of the factors which influences atomic bonding in ceramics)
What influences an ionic crystal structure?
crystal must be electrically neutral
ratio of cation/anion must be less than 1
Factor 3: Ionic Radii and Cation/Anion ratio (1 of the factors which influences atomic bonding in ceramics)
Why must rcation/ranion be less than 1?
ratio maximizes the number of anion neighbors that a cation has → stable structure
Factor 4: Coordination numbers (1 of the factors which influences atomic bonding in ceramics)
What does a higher coordination number mean in ceramics and why do you want a higher coordination number?
Higher coordination number → larger rc:ra ratio
larger rc:ra ratio → greater stability within ceramics
What are the three common ceramic crystal structures?
AX type
AmXp type
AmBnXp type
Tell me more about an AX type crystal
Cations and anions?
Example
Type of atomic bonding?
equal numbers of cations and anions
One type of cation and one type of anion
Example: NaCl (Na is the cation, Cl is the anion)
Highly covalent atomic bonding
Tell me more about an AmXp type crystal
Cations and anions?
Example
Type of atomic bonding?
charges on the cations and anions are not
the same, not an equal number of cations and anionsOne type of cation and two types of anions
Example: Fluorite (CaF2), Calcium is the cation and the two fluoride ions are the anions
Tell me more about an AmBnXp type crystal
Cations and anions?
Example
Type of atomic bonding?
Two types of cations (A and B)
Example: Barium titanate (BaTiO3), Ba2+ and Ti4+ are the cations, O3(2-) is the anion
Barium titanate→perovskite crystal structure
What are silicate ceramics?
SiO4 tetrahedron
Covalent because of Si-O bond (strong)
True or false: Silicate ceramics are ionic
False, silicate ceramics are covalent
What is the significance of SiO4-?
SiO4- is the basic unit of silicates
SiO4- has different 1D, 2D, and 3D arrangements
Tell me more about the SiO2 arrangement?
Polymorphs if crystalline?
Densities?
3 polymorphs:
Silica
Quartz
Cristobalite
Polymorphs→relatively low densities but high melting points
Why do polymorphs have relatively low densities but high melting points?
The Si-O interatomic bonds are very strong so they require a lot of energy to break
Silica glasses…tell me more about them
Atomic randomness
Basic unit?
Network formers, modifiers, intermediates
great disorder
SiO4- is the basic unit
Types include network formers, modifiers, and intermediates
What are network formers?
Backbone
Building blocks of glass (“backbone”)
Have a direct influence on the structure of glass
Examples: Silicon, Boron, Phosphorus
What are network modifiers?
modify, not form
Don’t form the backbone of glass like network formers;
Network modifiers sit in the network structure and change it to stabilize the network
Examples: CaO and Na2O
What are intermediates?
substitute teachers
intermediates substitute for silicon to stabilize the network
What does the addition of network modifiers or intermediates do?
Lower the melting point and viscosity of glass
Make glass easier to form at lower temperatures
What is the difference between simple and layered silicates?
Layered:
Net charge?
Electroneutrality?
Simple: involve isolated tetrahedra
Layered: 2D sheet
Net negative charge
Electroneutrality: second layered sheet with an excess of cations cancels out the first layered sheet with the net negative charge
True or false: Layered silicates form a 3D structure with the tetrahedra
False: they form a 2D layered structure by sharing 3 O’s in the tetrahedra
True or false: Layered silicates have neutral base units
False- (Si2O5)2- has an unshared oxygen atom that is balanced by a cation-rich planar sheet structure
Are clays examples of simple or layered silicates?
Answer the question
Example of clay
How many layers (if any)
Charge
Bonding within the sheet and between the two sheets?
Layered
Kaolinite
2 layer sheet structure
Neutral
Bonding within the two-layered sheet: strong (ionic/covalent)
Bonding between the two sheets: weak Van Der Waals
Which of the following are examples of network formers in silica glasses?
Silicon, boron, and phosphorus
Sodium and potassium
Aluminum and zinc
Calcium and magnesium
(Pick one)
Silicon, boron, and phosphorus
True or false: Network formers form glassy structures and polyhedral oxide structures
True
True or false: Network modifiers form glassy structures and polyhedral oxide structures
False: modifiers sit in the network and modify it
True or false: Intermediates form glassy structures and polyhedral oxide structures
False: intermediates like TiO2 and Al2O3 substitute for silicon to stabilize the network
True or false: Silica glasses are crystalline solids
False, silica glasses are noncrystalline solids with high atomic randomness
Carbon
2 famous allotropes?
Metal, ceramic or polymer?
Other allotropes?
Diamond and graphite
None of the above, graphite is sometimes classified as a ceramic
Nanocarbons
What is the primary structural difference between diamond and graphite?
Diamond has a hexagonal layer structure, while graphite has a 3D tetrahedral network
Diamond is held by weak Van der Waals forces, while graphite is bonded covalently
Diamond and graphite have identical structures but differ in density
Diamond has a tetrahedral network, while graphite has hexagonal layers with weak interlayer forces
(Pick one)
Diamond has a tetrahedral network, while graphite has hexagonal layers with weak interlayer forces
True or false: Graphite has basal plates connected to each other with covalent bonds
False- the basal plates are covalently bonded, but they are connected to each other by Van der Waals forces
Defects in ceramics?
Frenkel and Schottky defects
Defects
What is the difference between a Frenkel and a Schottky defect?
Frenkel: cation leaves its place in the crystal structure and moves to a nearby interstitial site, 1 vacancy
Schottky: cation-anion pair leave their place in the crystal structure, 2 vacancies
Does rcation/ranion change with the presence of Schottky or Frenkel defects?
No
Stoichiometry vs Nonstoichiometry?
Examples for each
What does nonstoichiometry induce in a crystal structure?
Stoichiometry: exact rcation/ranion ratio based on the chemical formula
Example: NaCl (ratio is 1:1)
Nonstoichiometry: deviation from rcation/ranion ratio predicted by the chemical formula
Example: FeO
Induces a defect to achieve electroneutrality
Impurities
2 types of impurities?
Interstitial and substitutional
Interstitial: impurity atoms occupy interstitial spaces of lattice structure
ionic radius of impurity must be smaller than the interstitial space!
Substitutional: foreign atom replaces host atom in crystal lattice without changing the overall structure of the material
True or false: Interstitial impurities in ceramics should be the same size as the anion
False, they should be relatively small compared to the anion
True or false: Substitutional impurities in ceramics always substitute for cations
False: they substitute for whichever host ion is most similar (electrically)
Solid solubility
What must the ionic radius and charge be compared to the host atom for a substitutional impurity to achieve solid solubility?
What if the ionic radius or charge is different?
What does the crystal want to achieve?
To achieve solid solubility, the charge and ionic size of the impurities must be nearly the same as the host ion
If the charge or ionic radius of the foreign atom is different from the host, a defect is induced to achieve electroneutrality
YOU ALWAYS WANT TO TRY TO DO WHATEVER POSSIBLE TO ACHIEVE ELECTRONEUTRALITY!!
Binary phase diagrams
two components are compounds that
share ___
Oxygen
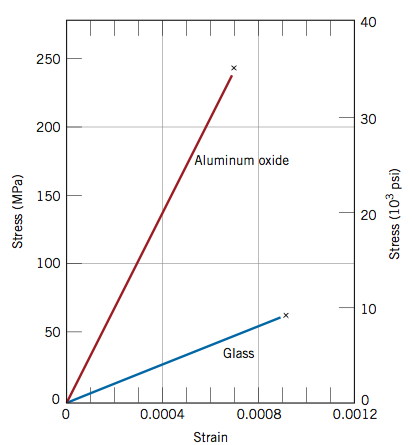
Stress-strain curve
What conclusions can you draw from this stress-strain curve of aluminum oxide and glass?
The curve for aluminum oxide is steeper than the curve for glass. This means that aluminum oxide has a higher Young’s Elastic Modulus (stress/strain) than glass which corresponds with higher stiffness.
Fracture toughness
What is fracture toughness?
measure of a ceramic
material’s ability to resist
fracture when a crack is
present
Porosity
Increased porosity increases E and reduces strength
False: it reduces both E and strength
How does porosity affect the properties of ceramics?
Makes ceramics more flexible
Improves electrical conductivity
Increases strength and toughness
Reduces strength but enhances applicability in filtration
(Pick one)
Reduces strength but enhances applicability in filtration
Properties and applications of glass-ceramics, clay products, refractory ceramics, and abrasives
What is a glass-ceramic?
Glass processed by heat treatment to produce a fine-grained polycrystalline structure
Are all glass-ceramics transparent?
Only some glass-ceramics may be made optically transparent
only very fine uniform grains are transparent
What are the most attractive attributes of glass-ceramics?
Ease of fabrication
Strength and excellent resistance to thermal shock
True or false: Glasses only crystallize above the critical cooling rate
False: they only crystallize below the critical cooling rate
True or false: Shifting transformation curves in glass crystallization affects how glasses crystallize with time
True
True or false: Transformation curves in glass crystallization are shifted by changes in viscosity
False: they are shifted by the use of nucleating agents
True or false: Glass-ceramics have high coefficients of thermal expansion
False: they have low coefficients to reduce cracks from cycling stress (and therefore avoid thermal shock)
True or false: Larger grains increase transparency
False: this will only work if it is a single crystal
True or false: Glass-ceramics are electrical and thermal insulators
True
What are the steps in manufacturing clay products?
Clay and water are mixed for shaping
Shaped piece is dried
Fired at a high temperature to improve mechanical strength
2 classifications of clay products?
Structural clay products
Whitewares
True or false: Structural clay products have very high structural integrity
True
True or false: Refractory ceramics are more reactive than other ceramics
False: they don’t degrade (even in high temps) and they are inert in extreme environments
Refractory ceramics provide thermal insulation and support mechanical loads
True
True or false: Refractory ceramics are used for products that need to withstand high temperatures
True
True or false: Abrasives are used to cut harder materials
False: they can only grind/cut softer materials
True or false: Abrasives need to be hard, wear resistant, and tough
True
What is the Mohs hardness of most abrasive materials?
7 or greater
True or false: Abrasive particles remove large chunks of material
False: they remove very small amounts of material
Phase transformation of glass ceramics
Most inorganic glasses transform from _____ to ______ at high-temperatures
through ____
noncrystalline to crystalline, nucleation
What is the critical cooling rate?
“just misses the nose”
What happens above the critical cooling rate?
does recrystallization/nucleation happen?
no, nucleation is prevented
What happens below the critical cooling rate?
nucleation takes place, some glass-ceramic material will form
Glass forming
What are some glass-forming examples?
Pressing, blowing, drawing, fiber forming
Particulate-forming examples
casting (slip/tape), powder pressing
Ceramics are produced by these 3 methods:
glass-forming (bottles, jars, sheet glass)
particulate-forming (electronic and magnetic ceramics)
cementation (dental veneers)
What is the glass transition temperature (Tg)?
temperature @ which amorphous polymer transitions from a hard, glassy state→softer, viscous state
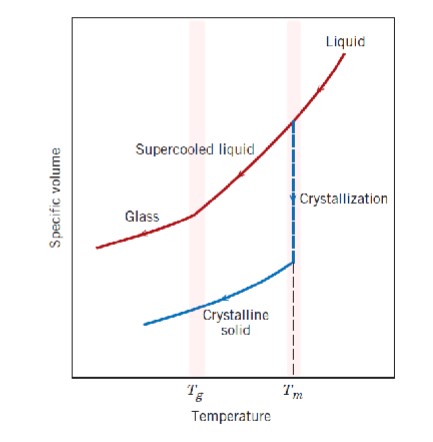
What is the difference between
glass and crystalline solids?
At the melting temperature (Tm), crystalline materials experience a sharp drop in specific volume (crystallization) before they become a crystalline solid.
At the melting temperature (Tm), liquid glass becomes a supercooled liquid and at the glass transition temperature, it becomes hardened glass.
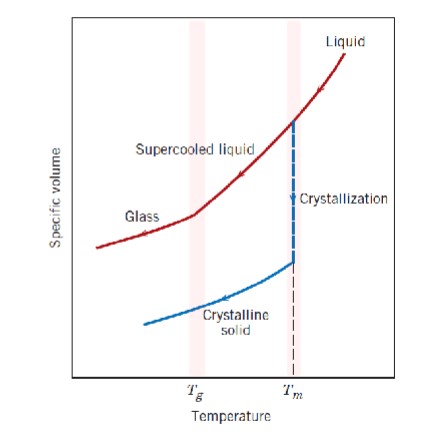
Interpret this graph
Both glass and crystalline solids start out as liquids. At the melting temperature (Tm), liquid glass becomes a supercooled liquid while crystalline solids experience a sharp drop in specific volume (crystallization) and become crystalline solids. The glass transition temperature (Tg) is significant for glass as that is when glass transitions from a supercooled rubbery state to a hard, brittle glass.
True or false: Glass is always amorphous
True
What is the difference between amorphous and crystalline materials?
Crystalline materials: repeating atomic structure
Amorphous materials: random arrangement of atoms in structure
If there is no crystallization, how do
you define Tm?
Viscosity
Is glass crystalline or amorphous?
Glass is amorphous. However, glass can be heat-treated to become crystalline (glass-ceramics)
What are the different points in glass forming and describe each?
Melting point: glass is fluid enough to be a liquid
Working point: glass is easily deformed
Softening point: glass can be handled without causing significant dimensional alternations
Annealing point: glass is heated to relieve any internal stresses that arise because of the glass working process
Strain point: Glass solidifies completely and will fracture if any extra loads are added
True or false: The melting point of glasses is determined by when they become liquids
False, the melting point of glasses is determined by viscosity
True or false: The glass transition temperature is below the strain point
False, the glass transition temperature is above the strain point
What are the points in glass forming from low to high viscosity and high to low temperatures?
Melting→working→softening→
annealing→strain
True or false: Glass-forming should be done between the working and softening points
True, this is called the working point of glass
True or false: At the strain point, glass will plastically deform and then fracture
False, at the strain point, glass will fracture and then plastically deform
Does glass have residual stresses when heated?
No, glass has residual stresses when cooled
True or false: Stresses from cooling can be fixed just above the annealing point
True
How does viscosity influence the temperature-dependent behavior of glass, and where does the glass transition temperature (Tg) fall in relation to viscosity levels?
Viscosity as glass is heated?
Viscosity as glass is cooled?
Viscosity before Tg?
Viscosity above Tg?
As glass is heated, viscosity decreases (resistance to flow decreases).
As glass is cooled, viscosity increases (resistance to flow increases).
Before Tg (material is glassy), viscosity is higher than above Tg.
Above Tg (material is rubbery), viscosity is lower than below Tg.
Which of the following statements is true about the viscosity that glass reaches at different points?
All statements are true
Viscosity at the melting point is very high, i.e. glass is fluid enough to behave as a liquid.
Viscosity is very high at the strain point, at which the material becomes so rigid that it will fracture before undergoing plastic deformation.
Viscosity at the annealing point is lower than at the softening point, such that atomic diffusion occurs rapidly enough to relieve residual stresses within about 15 minutes at the annealing point.
Viscosity at the working point is lower than at the melting point, such that the glass can be easily shaped and deformed at the working point.
(Pick one)
Viscosity is very high at the strain point, at which the material becomes so rigid that it will fracture before undergoing plastic deformation.
What are the steps in the press and blow technique?
Put gob into mold
Press it to make a concave shape
Blow in compressed air to fit the mold
True or false: The float process makes hollow containers
False: the float process makes sheets
True or false: Glass tempering decreases strength
False, it increases strength by inducing compressive surface stresses
True or false: Glass tempering occurs above the softening point
False, it occurs below the softening point
How is glass cooled in glass tempering?
With air or oil
True or false: Powder processing compacts powder into a solid
False: it compacts powder, but it needs to be sintered to be a bonded solid
What is the difference between ceramic jet 3D printing and the stereolithographic technique for 3D printing?
Difference in curing:
SLA uses a laser to cure
Ceramic jet 3D printing uses ceramic particles to cure