Functions of the Liver
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
What are the three metabolic phases of the liver?
Absorptive phase, postabsorptive phase, and starvation phase.
What are the three specific functional categories of the liver?
Synthesis, biotransformation/detoxification, and storage.
What characterizes the absorptive phase?
It is an anabolic phase occurring 3-4 hours after a meal when nutrients are being digested and absorbed.
How do amino acids and monosaccharides reach the liver after digestion?
They are released by enterocytes into the enteric vasculature and reach the liver via the portal system.
What is the primary role of insulin during the absorptive phase?
It responds to high blood glucose and amino acid levels to stimulate nutrient uptake, storage, protein synthesis, and lipogenesis.
How does the Growth Hormone/IGF-1 axis function during the absorptive phase?
It responds to high amino acid levels; insulin upregulates hepatic GH receptors, leading to IGF-1 production which promotes protein anabolism.
What are the three stages of glucose oxidative ATP production?
Glycolysis, the Krebs cycle, and the electron transport chain.
What is the structural composition of glycogen?
A branched polysaccharide containing both 1-4 and 1-6 alpha glycosidic linkages.
What enzyme is required to create the 1-6 branching points in glycogen?
A branching enzyme.
What is the result of deaminating amino acids in the liver?
They are converted into keto acids, which can be used for non-essential amino acid synthesis, lipogenesis, or ATP production.
How does the liver handle excess ammonia (NH3/NH4) produced during amino acid metabolism?
It detoxifies it into urea for excretion via the kidneys or secretion into the forestomach in ruminants.
What are the primary transport forms of nitrogen from muscle to liver?
Alanine and glutamine.
What is the difference between free cholesterol and cholesterol esters?
Free cholesterol is amphipathic and used for membranes/precursors, while cholesterol esters are lipophilic and serve as a storage form.
Why do fats require transport mechanisms in the blood?
Because they are non-polar and would aggregate in the watery medium of the blood.
What is the role of albumin in lipid transport?
It acts as a water-soluble transport protein for free fatty acids, bilirubin, and certain drugs.
What is the structural composition of a lipoprotein shell?
Phospholipids, free cholesterol, and apoproteins.
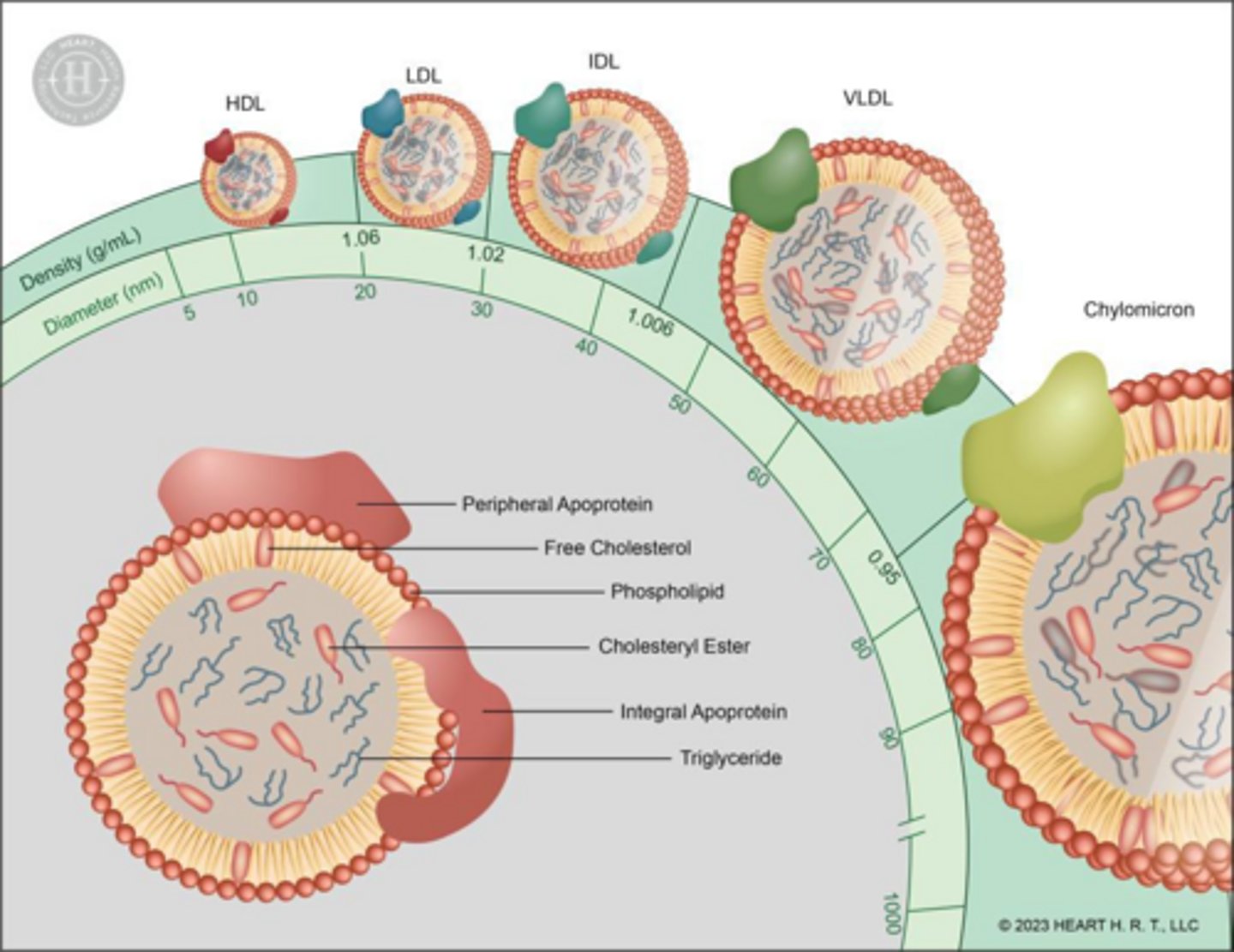
What is the structural composition of a lipoprotein core?
Triglycerides, cholesterol esters, and vitamins A, D, E, and K.
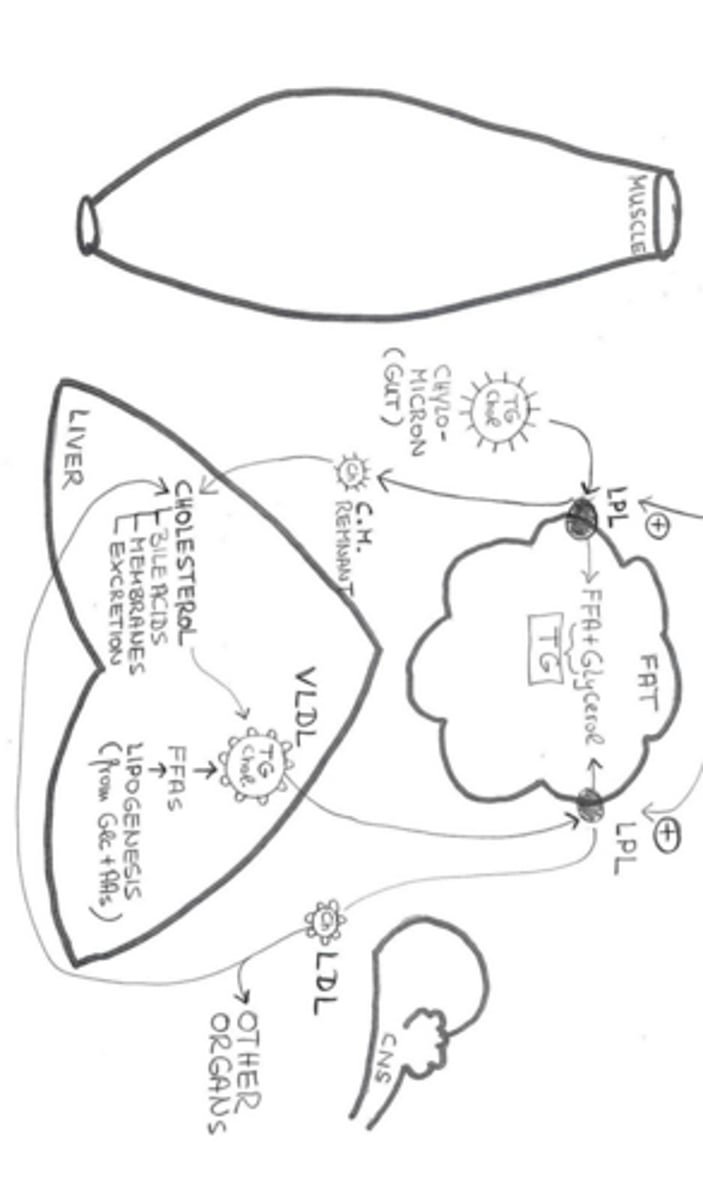
Where are chylomicrons produced and what do they contain?
They are produced in enterocytes and contain dietary fats.
Where are VLDLs produced and what is their composition?
They are produced in the liver and contain endogenous fat with a high fat-to-protein ratio.
What are the two primary functions of apoproteins?
They act as ligands for tissue-specific receptors and as activators of lipid metabolism enzymes like lipoprotein lipases.
What hormone stimulates lipoprotein lipase (LPL) in fat cells?
Insulin.
What happens to chylomicrons after lipoprotein lipases hydrolyze their triglycerides?
They become chylomicron remnants, which are cleared by the liver.
What does the liver do with the cholesterol found in chylomicron remnants?
It uses it for bile acid production, membrane synthesis, or excretion if levels are excessive.
What is the postabsorptive phase of metabolism?
A phase characterized by low energy intake where the body mobilizes stored energy.
What is the primary hormone driving the absorptive phase?
Insulin
What is lipogenesis?
The production of free fatty acids and triglycerides from excess glucose and amino acids.
What transport vehicle does the liver use to export endogenous fats?
VLDLs (Very Low-Density Lipoproteins)
What enzyme facilitates the storage of fats from VLDLs and chylomicrons into fat tissue?
LPL (Lipoprotein Lipase)
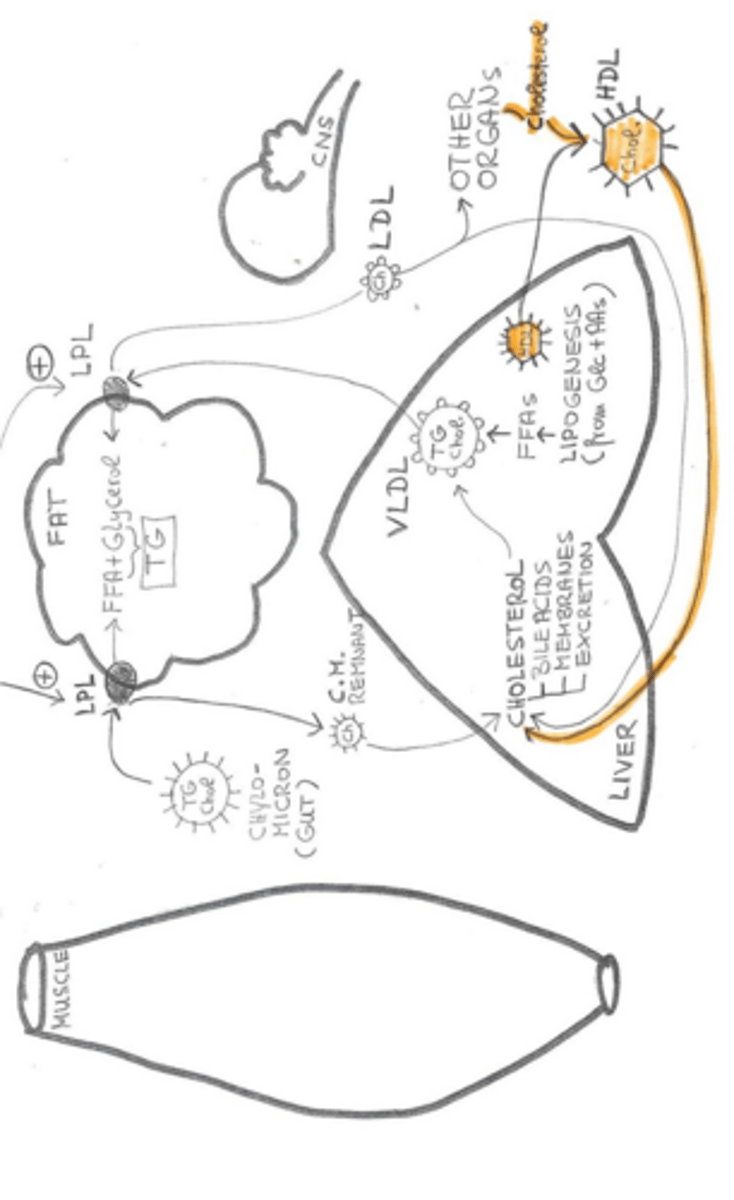
Why are LDLs often referred to as 'bad cholesterol'?
Because excess cholesterol delivered by LDLs can be deposited on endothelial membranes, potentially causing plaques.
What is the primary function of HDLs?
To scavenge excess cholesterol from peripheral tissues and deliver it back to the liver.
What condition can occur in species with limited VLDL export mechanisms when fed high-caloric diets?
Hepatic fat accumulation (Fatty Liver).
What is the main role of the liver in glucose homeostasis during the postabsorptive phase?
To release glucose into circulation to maintain levels for glucose-dependent tissues.
Which tissues are considered glucose-dependent?
Neurons and red blood cells (RBCs).
What are the primary hormones that dominate the postabsorptive phase?
Glucagon and epinephrine.
What hormones become dominant as the postabsorptive phase progresses into fasting and starvation?
Cortisol and growth hormone (GH).
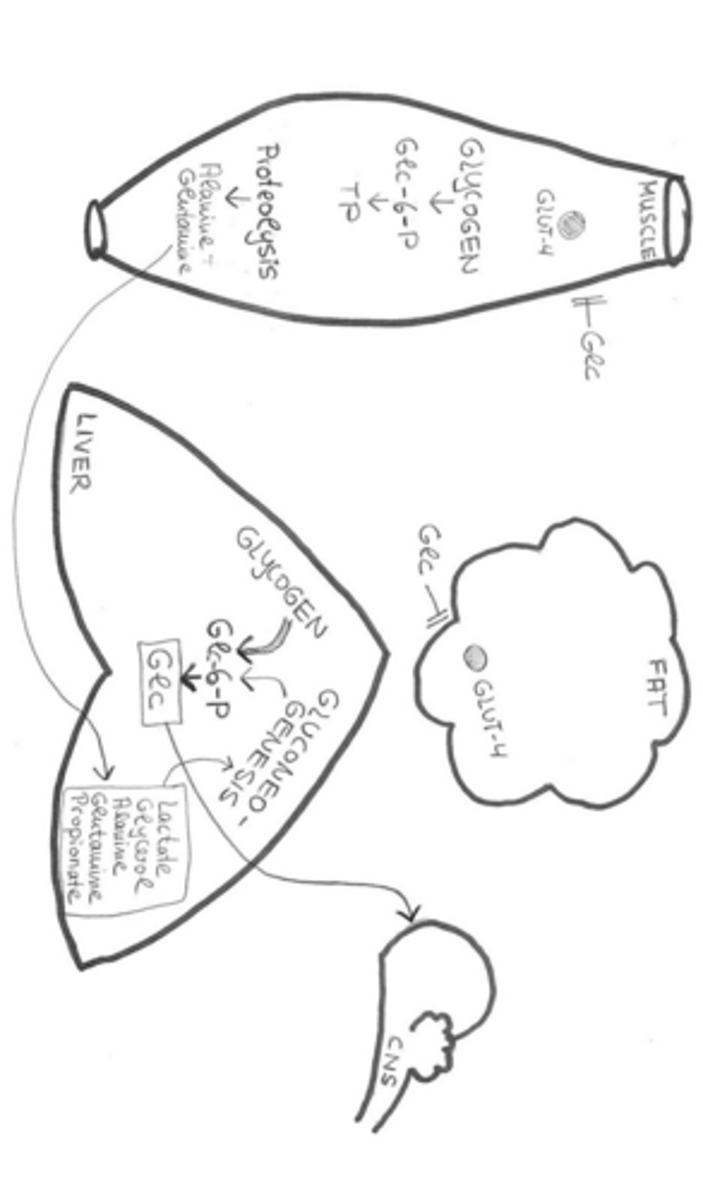
What is the difference between liver and muscle glycogenolysis regarding glucose release?
Only the liver contains phosphatases to release free glucose into circulation; muscles use glucose-6-phosphate for their own energy.
What are the primary precursors for gluconeogenesis?
Lactate, glycerol, amino acids (alanine and glutamine), and propionate.
Which organs are responsible for gluconeogenesis?
The liver (80%) and the kidney.
What happens to protein metabolism during the postabsorptive phase?
Protein synthesis ceases and proteolysis (net breakdown of proteins) begins, particularly in muscle tissue.
In what form are amino acids released during muscle proteolysis?
Mostly as alanine and glutamate.
What is the role of Hormone-sensitive Lipase (HSL) in fat tissue during the postabsorptive phase?
It hydrolyzes stored triglycerides into free fatty acids and glycerol for circulation.
How are free fatty acids transported in the blood?
They bind to albumin and are referred to as non-esterified fatty acids (NEFAs).
What process do glucose-independent tissues use to generate ATP from NEFAs?
Beta-oxidation, which yields acetyl groups for the Krebs cycle.
What is the fate of IDLs/LDLs after triglycerides are removed from VLDLs?
They carry mostly cholesterol to tissues for membrane synthesis, steroid hormone production, and bile acid production.
What is the effect of insulin on muscle tissue regarding fat uptake?
Fat tissue LPL is dominant under insulin stimulation, but without insulin, chylomicrons deliver fats to muscle tissue.
What is the purpose of cortisol and GH inducing mild insulin resistance during starvation?
To reserve 'metabolically expensive' glucose for glucose-dependent tissues.
What is the metabolic result of excess acetyl groups during lipogenesis?
The production of cholesterol.
When does the postabsorptive phase typically begin?
3 to 4 hours after a meal, once nutrient levels in the blood start to decline.
What are the three primary pathways the liver uses to process FFAs during the postabsorptive phase?
Beta-oxidation, ketogenesis, and VLDL production.
What are the three main ketone bodies produced by the liver?
Acetone, acetoacetate, and beta-hydroxybutyrate.
Why does muscle LPL become dominant over fat tissue LPL during the postabsorptive phase?
Because the lack of insulin inhibits fat tissue LPL activity.
Which hormones are dominant during the initial postabsorptive phase?
Glucagon and epinephrine.
Which hormones are dominant during prolonged starvation?
Cortisol and growth hormone (GH).
What are the three metabolic dysfunctions associated with excessive lipolysis during starvation?
Ketoacidosis, hepatic lipidosis (fatty liver), and hyperlipemia.
What is the primary cause of ketoacidosis in ruminants during starvation?
Excessive beta-oxidation of FFAs producing more ketone bodies than the body can utilize.
Why does hepatic lipidosis occur in cats and ponies during catabolic states?
Reduced lipoprotein synthesis prevents the export of TGs, leading to their accumulation in hepatocytes.
What characterizes hyperlipemia in ponies?
VLDL export into the blood exceeds the clearance capacity of tissues, resulting in milky plasma.
What is the primary function of albumin synthesized by the liver?
Maintaining oncotic pressure and transporting FFAs, calcium, hormones, and drugs.
Which liver-synthesized hormone is responsible for platelet production?
Thrombopoietin.
What is the role of angiotensinogen in the body?
It is part of the RAAS system, regulating blood pressure and blood volume.
Which liver-produced hormone mediates protein anabolism?
IGF-1 (stimulated by GH).
What is the function of hepcidin?
Regulation of iron metabolism.
What is the difference between Phase I and Phase II detoxification reactions in the liver?
Phase I prepares substrates (often using Cytochrome P450), while Phase II uses conjugation to make metabolites water-soluble for excretion.
How does the liver detoxify ammonia (NH3/NH4)?
By converting it into urea.
What is the purpose of bilirubin processing in the liver?
The degradation and excretion of heme groups.
Which fat-soluble vitamins are stored in the liver?
Vitamins A, D, E, and K.
Which water-soluble vitamins are stored in the liver?
Vitamin B12 and folate (B9).
In what form is iron stored in the liver?
As ferritin and hemosiderin.
What is the primary function of HDL formed by the liver?
Acting as a cholesterol scavenger.
What is the role of the liver in glucose homeostasis?
Glycogen storage and gluconeogenesis.
What are the main immune functions of the liver?
Production of complement factors (C1-C9) and acute phase proteins like C-reactive protein and serum amyloid A.
Why is the liver considered a 'fat sponge' during the postabsorptive phase?
Because hepatocytes readily absorb FFAs from non-esterified fatty acids (NEFAs) to process them for energy or transport.