Exam 3 - Biochemistry Honors
1/288
Earn XP
Description and Tags
No regrets
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
289 Terms
What is the name of the catabolic process that breaks down amino acids?
oxidative deamination (oxidative degradation of amino acids)
The three circumstances under which amino acids undergo oxidative degradation
During the normal synthesis and degradation of cellular proteins
When the diet is rich in protein and there is an excess of amino acids
During starvation or uncontrolled diabetes mellitus
Under the metabolic conditions that allow amino acids to undergo oxidative degradation, amino acids…
lose their amino groups to form α-keto acids
The two key features of amino acid degradation
Separation of α-amino group from carbon skeleton
Separate pathways for amino group and skeleton
Following the separation of the α-amino group from the carbon skeleton, the amino group ends up in the
urea cycle
Following the separation of the α-amino group from the carbon skeleton, the carbon group ends up in the
citric acid cycle
Dietary amines are always associated with carriers. What are the primary carriers to the liver?
glutamine and alanine
Dietary amines are always associated with carriers. What is the primary pathway in muscle?
Ammonia is coupled to pyruvate, forming alanine
Dietary amines are always associated with carriers. What is the primary carrier from the liver to the renal system?
urea
Most amino acids are metabolized in the
liver
The urea cycle primarily occurs in hepatocytes, beginning in their mitochondria. Excess ammonia generated in extrahepatic tissues travels to the _ for conversion to the excretory form.
liver
Draw the structure of ammonia.

Draw the structure of urea.
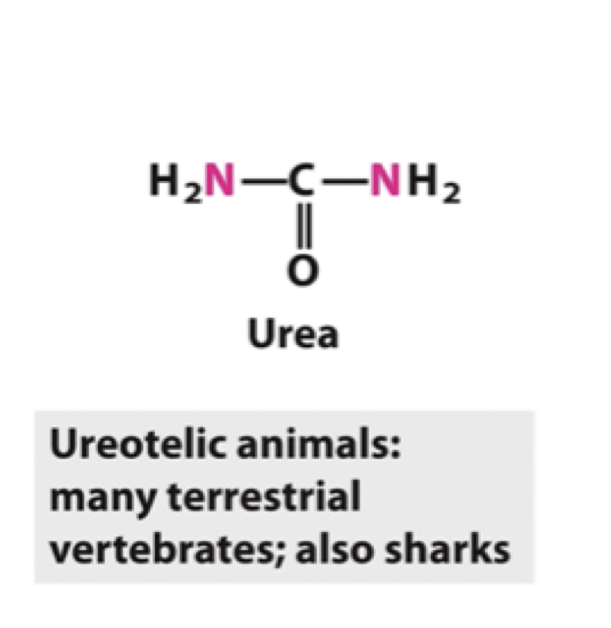

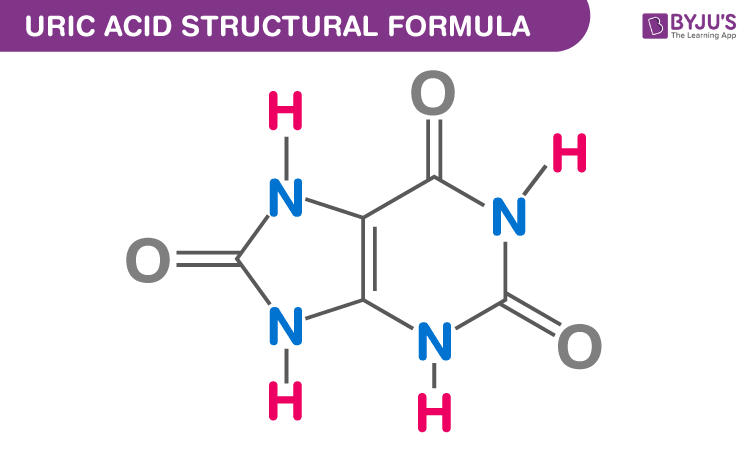
Draw the structure of uric acid.
Hormones control digestion. Dietary protein in the stomach stimulates the _ to secrete _
gastric mucosa, gastrin
Gastrin stimulares the secretion of (1) (2)
HCl by the parietal cells
Pepsinogen by the chief cells of the gastric glands
_ triggers secretion of the hormone secretin into the blood.
Acidic stomach contents lower the pH which
Secretin stimulates the _ to secrete _
pancreas, bicarbonate
The pancreas secretes bicarbonate to
neutralize gastric HCl
How does the secretion of bicarbonate by the pancreas affect the pH?
it increases it to about 7
The presence of protein in the small intestines signal the release of
cholecystokinin
Cholecystokinin stimulates secretion of
several pancreatic digestive enzymes
At a very low pH (~1), pepsinogen is converted to _ by an _
active pepsin, autocatalytic cleavage
Pepsin is active at a very low pH (~1), and its function is
to cleave denatured proteins into peptides
What are the zymogens that are inactive precursors to proteases, secreted by the pancreas?
Trypsinogen, chymotrypsinogen, procarboxypeptidases A and B
Which enzyme is secreted by the small intestines to cleave trypsinogen into trypsin?
Enteropeptidase
Following the cleavage of trypsinogen,
the other two proteases are activated
What tissue absorbs amino acids?
intestinal mucosa
Free amino acids (generated by digestion) are transported into the epithelial cells lining the (organ), enter the blood capillaries in the (?), and travel to the (organ)
small intestine, villi, liver
The suffix “-ogen” indicates that the enzyme is
a precursor
The suffix “-ase” indicates that the enzyme is
active
Pepsin cleaves proteins
N-terminal to L, F, Y, and W
Trypsin cleaves peptides
C-terminal to K and R
Chymotrypsin cleaves peptides
C-terminal to F, Y, and W
Carboxypeptidase cleave
C-terminal residues
Aminopeptidases cleave
N-terminal residues
In the liver, aminotransferases (transaminases) complete a _ reaction
transamination reaction
Transamination Reaction
The α-amino group is transferred from the L-amino acid to the α-carbon of α-ketoglutarate.
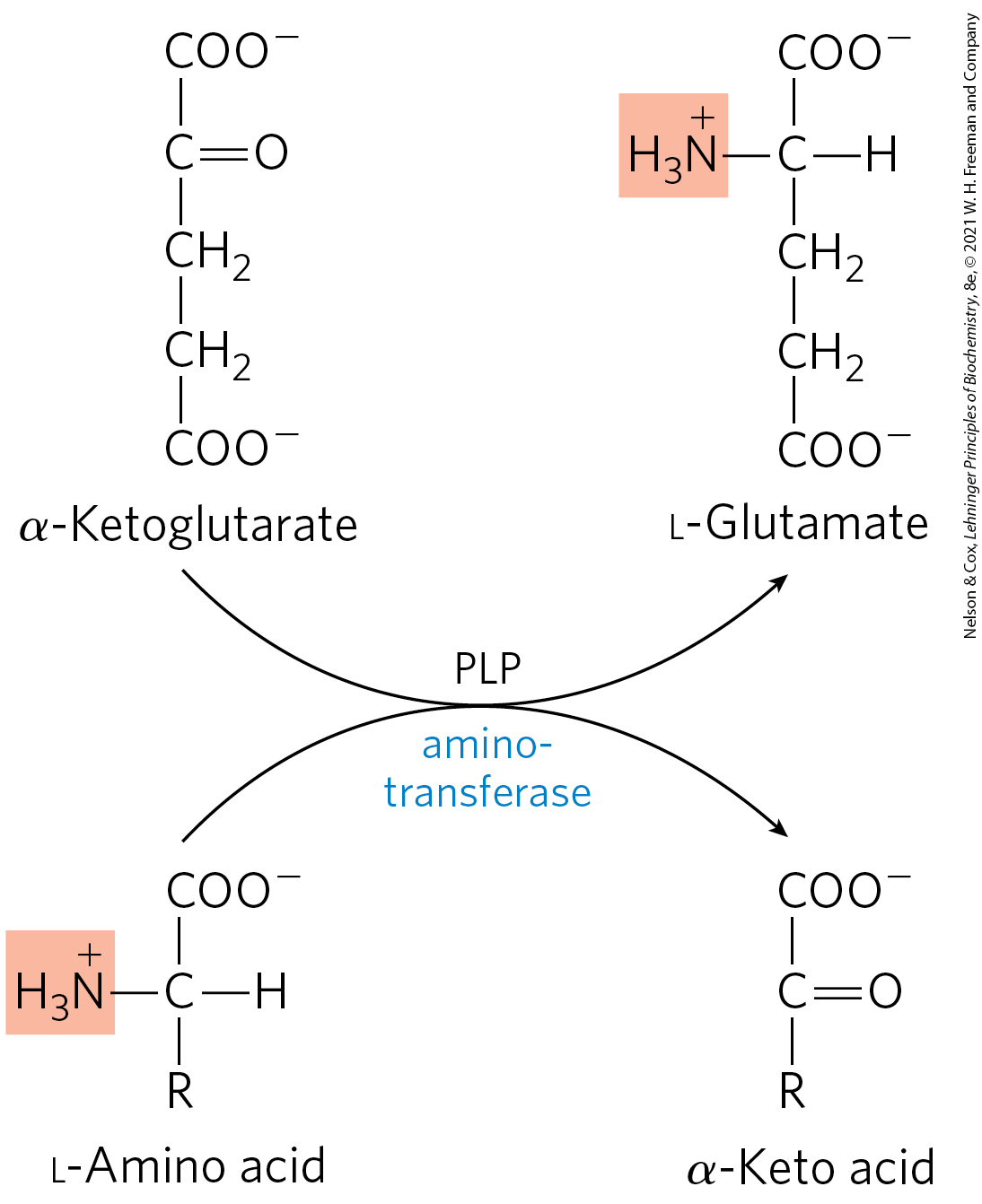
The effect of transamination reactions is
to collect the amino groups from many different amino acids in the form of L-glutamate
This product of transamination reactions functions as an amino group donor for biosynthetic pathways or for excretion pathways that lead to the elimination of nitrogenous waste products
L-glutamate
The prosthetic group of all aminotransferases
Pyridoxal phosphate (PLP)
Coenzyme form of Vitamin B6
Pyridoxal phosphate (PLP)
PLP is covalently bound to an enzyme’s active site by
an aldimine linkage to the α-amino group of a Lys residue
The coupled form (PLP–Lys) is called (?) and helps to (?)
internal aldimine, resist oxidation of the aldehyde in the inactive form
Intermediate of transaminase reactions
external aldimine, formed when an amino substrate replaces the Lys’ bond to PLP
In the transaminase reaction, an active site (?) deprotonates the the external aldimine to form (?)
nucleophile, quinonoid intermediate
The acceptor that can reverse the transamination reaction
α-ketoglutarate, an acceptor keto acid
The keto acid formed during transamination reactions is formed by the reaction type
hydrolysis
Three main reactions facilitated by PLP
Transamination
Racemization (loss of stereochemistry)
Decarboxylation
In hepatocytes, glutamate is transported from
the cytosol into mitochondria
In the mitochondria, glutamate undergoes (reaction?) catalyzed by (enzyme?)
oxidative deamination, L-glutamate dehydrogenase
Where can you find L-glutamate dehydrogenase?
mitochondrial matrix
The only enzyme that can use either NAD or NADP as the acceptor of reducing equivalents
L-glutamate dehydrogenase
aminotransferase + glutamate dehydrogenase =
trans-deamination
Free ammonia produced in extrahepatic tissues is… + enzyme?
combined with glutamate to yield glutamine, catalyzed by glutamine synthetase
synthetases (do/do not) use ATP
DO USE ATP
Conversion of ammonia into a nontoxic compound.
Glutamate + ATP → (?) → (?)
ADP + γ-glutamyl phosphate + NH4+ (glutamine synthetase) → glutamine + Pi
Together, (AA?) + (AA?) contribute to the regulation of blood pH
Hint: conjugate acid and base
glutamine + glutamate
What amino acid transports ammonia to the heptocytes from most extrahepatic tissues
glutamine
What enzyme converts glutamine to glutamate and NH4+
glutaminase
During exercise, skeletal mmuslce tissue accumulates
pyruvate
In parallel to exporting lactic acid, ammonia from (AA?) is transferred to (?) to create (?)
glutamate, pyruvate, alanine
The aminotransferase that transfers ammonia from glutamate to pyruvate
alanine aminotransferase
The two precursors of pyruvate,
lactic acid
alanine
Lactic acid and pyruvate can be used by hepatocytes for
gluconeogenesis
Glutamine from extrahepatic tissue is the product of which enzyme
Glutamine synthetase
The three major processes of nitrogen excretion
Ammonotelic
Ureotelic
Uricotelic
Ammonotelic organisms excrete amino nitrogen as
ammonia
Ureotelic organisms extrete amino nitrogen in the form of
urea
Uricotelic organisms excrete amino nitrogen as
uric acid
Urea production (the fate of most ammonia) occurs mostly in the
mitochondria of liver cells
One amino group enters the urea cycle as (?), which is formed in the mitochondrial matrix
carbamoyl phosphate
One amino group enters the urea cycle as (?), formed in the matrix by transamination of oxaloacetate and glutamate, then is moved to the cytosol
Bonus: what enzyme catalyzes the transamination?
aspartate
aspartate aminotransferase
The four main steps of the urea cycle
First amino group: Ornithine + carbamoyl phosphate → citrulline → cytosol
Second amino group: Argininosuccinate formed via citrullyl-AMP intermediate
Argininosuccinate → arginine (+ fumarate → TCA)
Urea + ornithine
(#?) amines are required for the synthesis of urea by hepatocyte, which come from (AA?)
2, glutamine or glutamate
Urea Cycle: Activation Step
Enzyme:
Product:
Starting Material:
Cofactors:
Matrix or Cytosol?
carbamoyl phosphate synthetase I
carbamoyl phosphate (an amide)
NH4+ (ammonia) + HCO3- (bicarbonate)
2 ATP
Matrix
The Carbamoyl Phosphate Synthetase I Reaction (3 steps)
ATP + Bicarbonate → carbonic-phosphoric acid anhydride + NH3 → carbamate + ATP → carbamoyl phosphate
Phosphorylation of bicarbonate
Ammonia substitutes Pi
Phosphorylation of carbamate
What is the first committed step of the urea cycle?
Mitochondrial synthesis of carbamoyl phosphate, catalyzed by carbamoyl phosphate synthetase I
Urea Cycle: Step 1
Enzyme:
Product:
Starting Material:
Cofactors:
Matrix or Cytosol?
ornithine transcarbamoylase
citrulline + Pi
ornithine + carbamoyl phosphate
X
Matrix → Cytosol (citrulline is transported OUT)
mitochondrial antiport
citrulline/ornithine
Urea Cycle: Step 2
Enzyme:
Product:
Intermediate?
Starting Material:
Cofactors:
Matrix or Cytosol?
arginosuccinate synthetase
arginosuccinate
citrullyl-AMP intermediate
citrulline + aspartate
ATP
cytosol
One molecule of urea requires hydrolysis of (?) ATP molecules
3 ATP
The Argininosuccinate Synthetase Reaction (Two Activation Steps)
ATP + citrulline → citrullyl-AMP + aspartate → argininosuccinate + AMP
ATP + citrulline → Citruyll-AMP intermediate
Aspartate substitutes AMP
Urea Cycle: Step 3 - Connected to TCA
Enzyme:
Product:
Starting Material:
Cofactors:
Matrix or Cytosol?
argininosuccinase
arginine + (fumarate → malate → TCA)
argininosuccinate
X
cytosol
Urea Cycle: Step 4 - The Big One!
Enzyme:
Product:
Starting Material:
Cofactors:
Matrix or Cytosol?
arginase
urea + (ornithine → matrix for urea cycle)
arginine
X
cytosol → matrix
Where is carbamoyl phosphate synthetase I located?
hepatocyte mitochondria
4 mitochondrial enzymes of the urea cycle
glutamate dehydrogenase
aspartate aminotransferase
carbamoyl phosphate synthetase I
ornithine transcarbamoylase
3 cytosolic enzymes of the urea cycle
argininosuccinate synthetase
argininosuccinase
arginase
glutaminase releases
NH4+ from Q (glutamine)
aspartate aminotransferase transfers the amine group from
glutamine to oxaloacetate to create aspartate
carbamoyl phosphate synthetase creates
carbamoyl phosphate from bicarbonate, ammonia, and ATP
ornithine transcarbamoylase couples
carbamoyl phosphate and ornithin to make citrilline
argininosuccinate synthetase couples
citrilline and aspartate to form argininosuccinatea
rgininosuccinase cleaves
arginionsuccinate into arginine and fumaratear
arginase cleaves
arginine into ornithine and urea
(?) concentration needs to remain low to drive the TCA forward
oxaloacetate
aspartate-argininosuccinate shunt
links TCA and urea cycle
the three main one-carbon transfer cofactors
biotin
tetrahydrofolate
S-adnosylmethionine
biotin carries carbon as
CO2 or carbonic acid
tetrahydrofolate (THF or H4 folate) carries carbon in
intermediate oxidation states