Edexcel IGCSE Biology - Biological Molecules
1/26
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
are carbohydrates monomers or polymers?
polymers
what types of carbohydrate are there?
glycogen, starch, sucrose
what are carbohydrate monomers?
simple sugars
what types of carbohydrate monomers are there?
glucose, fructose, ribose
what elements are carbohydrates made up of?
carbon, hydrogen, oxygen
are proteins polymers or monomers?
polymers
what monomers make up proteins?
amino acids
what elements make up proteins?
carbon, hydrogen, oxygen, nitrogen
are lipids polymers?
no
what elements make up lipids?
carbon, hydrogen, oxygen
what sub-units make up lipids?
fatty acids and glycerol
what is an enzyme?
a biological catalyst, present in metabolic reactions
what kind of biological molecules are enzymes?
proteins
what is the molecule that is changed in a chemical reaction called?
substrate
what is the part of the enzyme where the substrate joins to it called?
active site
what feature of the active site is very important?
its unique shape - each enzyme can normally only catalyse one type of reaction
what model is used to describe enzyme-substrate reactions?
lock and key model
what is the lock and key model composed of?
lock - enzyme
key - substrate
combine to form enzyme-substrate complex
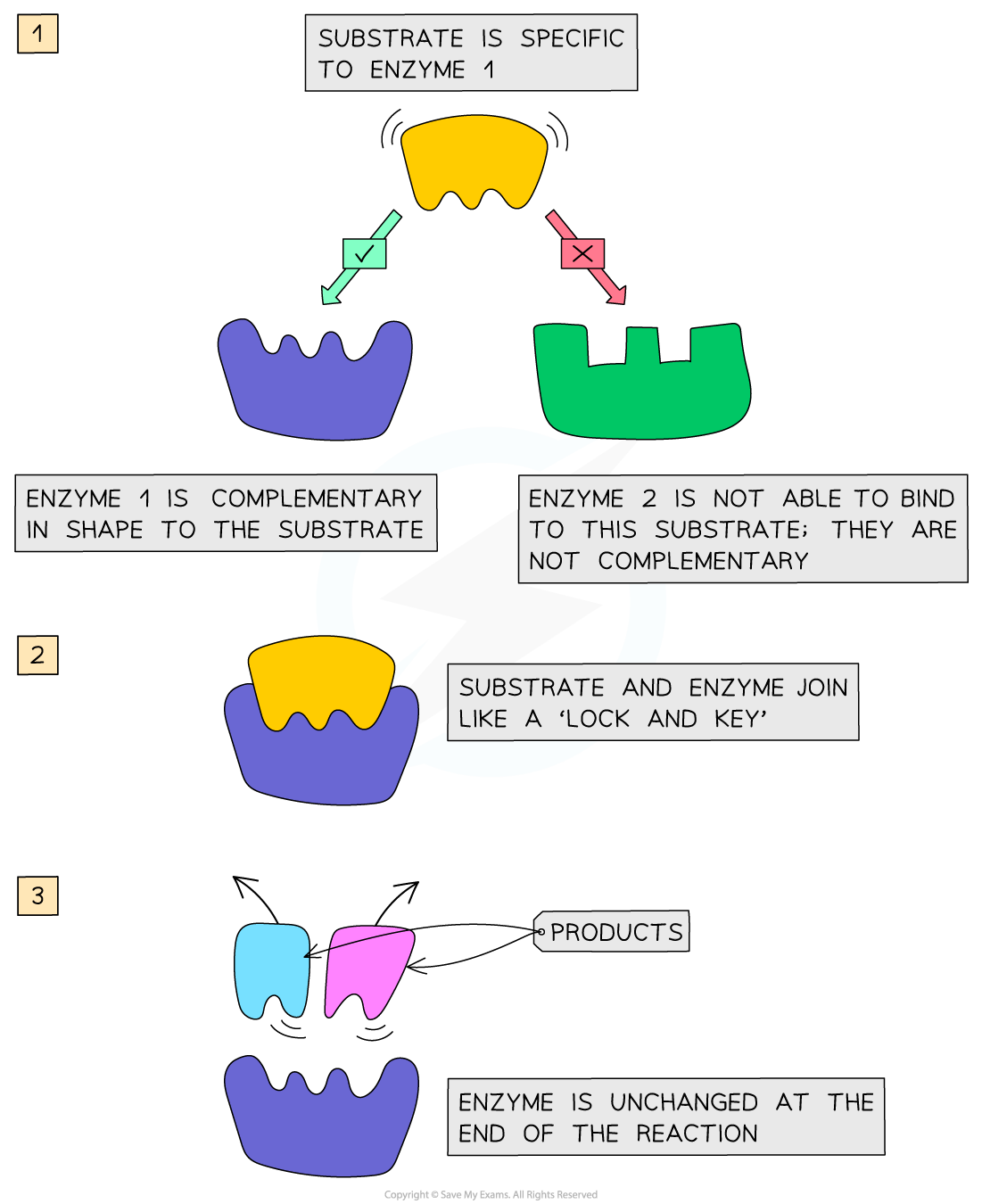
what aspect of enzymes does the lock-and-key model stress?
the fact that the active site of the enzyme must be complementary to the substrate for the reaction to take place
which factors affect enzymes + rate of reaction?
temperature
pH
substrate concentration
enzyme concentration
how does enzyme concentration affect rate of reaction?
directly proportional relationship
as long as the substrate concentration is constant, the ROR will increase proportionally as more active sites become available, as long as sufficient substrates are available - stops when something else is limiting factor
how does substrate concentration affect rate of reaction?
directly proportional relationship
increasing concentration increases rate of reaction because there are more particles available to collide with active sites so more enzyme-substrate complexes are formed
at some point, it will stop increasing ROR as something else becomes the limiting factor like enzyme concentration
how does temperature affect enzyme rate of reaction?
as temperature increases to the optimum, rate of reaction increases as particles have more kinetic energy and so collide more frequently forming more complexes
as temperature increases past the optimum, rate of reaction increases as enzymes begin to denature, meaning their active sites disfigure and so a complex cannot form - this is irreversible
at low temperatures, enzymes do not have much kinetic energy so rate also decreases
how does pH affect the rate of reaction?
as pH increases or decreases to the optimum, rate of reaction increases as the active site becomes more complementary to the substrate
as pH increases or decreases (too high/low) past the optimum, rate of reaction increases as enzymes begin to denature, meaning their active sites disfigure and so a complex cannot form - this is irreversible
describe how to perform an experiment to investigate how enzyme activity is altered by temperature by measuring how fast a product appears
the enzyme catalase catalyses the breakdown of hydrogen peroxide into water and oxygen.
You can collect the oxygen and measure how much is produced in a set time
use a pipette to add a set amount of hydrogen peroxide to a boiling tube
put it in a water bath at 10C
set up the apparatus with an upturned measuring cylinder with a delivery tube going into it attached to the test tube, in a trough of water - record starting water volume
add a source of catalase - potato - to the hydrogen peroxide and attach the bung quickly
record how much oxygen is produced in the first minute, repeat 3x and calculate mean
repeat at 20,30,40,50 degrees
control variables
describe how to perform an experiment to investigate how enzyme activity is altered by temperature by measuring how fast a substrate disappears
amylase catalyses the breakdown of starch to maltose
starch can be detected using iodine solution: orange → blue-black
set up a spotting tile with a drop of iodine solution in each dimple
add some amylase to some starch solution and place in a water bath at 10C
start a timer and place a drop of solution in the spotting tile every 10 secs until the solution stops changing colour - remains orange - stop the timer
repeat with different temperatures
how would you alter the temperature-enzyme activity experiments to measure the effect of pH?
instead of using water baths at different temperatures, add different pH buffer solutions to the test tubes