CHEM 12!! - Kinetics
1/105
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
How is the speed of a chemical reaction defined?
As the rate of appearance of a product or disappearance of a reactant with respect to time.
What is the most common way to measure the rate of a reaction?
By the change in concentration of reactants or products over time.
What happens to reactant concentrations over time during a reaction?
Reactant concentrations decrease over time.
What happens to product concentrations over time during a reaction?
Product concentrations increase over time.
In the combustion of methane, how does the rate of oxygen disappearance compare to methane?
Oxygen will disappear at twice the rate that methane disappears.
What is the formula for calculating the rate of appearance of carbon dioxide?
rate = ∆[CO2]/∆t
What is the rate of appearance of water in the combustion of methane if it is 0.50 mol/L·s?
0.50 mol/L·s
How do we express the rate of disappearance of a reactant?
By putting a negative sign in front of the change in concentration.
What is the average rate of disappearance of methane if it is 0.25 mol/L·s?
−0.25 mol/L·s
What does the graph of concentration vs. time look like for a reaction?
It is typically curved, indicating a slowing reaction rate.
What is the difference between average rate and instantaneous rate?
Average rate is over a time period, while instantaneous rate is at a specific time.
What graphical methods can be used to measure average and instantaneous rates?
Secant lines for average rate and tangent lines for instantaneous rate.
What happens to the rate of a reaction as reactants are consumed?
The reaction rate typically slows down.
What is the general reaction formula for A → B?
A converts to B over time.
How can we monitor the amount of reactant or product over time?
By using various methods summarized in a table.
What is the balanced chemical equation for the reaction of magnesium with hydrochloric acid?
Mg(s) + 2 HCl(aq) → MgCl2(aq) + H2(g)
What is the significance of the coefficients in a balanced chemical equation?
They indicate the relative rates of disappearance and appearance of reactants and products.
What does a 1:1 ratio in a reaction graph indicate?
It indicates that reactants and products are changing at the same rate.
What is the impact of fewer collisions among reactants on the reaction rate?
It leads to a decrease in the reaction rate.
What is the role of time in calculating reaction rates?
Time is a critical factor in determining both average and instantaneous rates.
What is a common method for measuring the concentration of reactants or products?
Using spectrophotometry or titration.
What does a negative rate indicate in a chemical reaction?
It indicates the rate of disappearance of a reactant.
What is the importance of specifying the compound when stating the rate of a reaction?
Different compounds may have different rates of change.
What does a rate law express?
The relationship between the rate of a reaction and the concentrations of the reactants raised to an exponent, along with the rate constant.
What is the rate constant (k)?
A measure of the inherent speed of the reaction that changes with temperature.
Do products appear in the rate law?
No, only the reactants appear in the rate law.
What can the exponents in a rate law indicate?
They can indicate the order of the reaction with respect to each reactant and can be integers or fractions.
What does an exponent of zero mean in a rate law?
It means that the rate will not depend on the concentration of that reactant.
How are the exponents in a rate law determined?
They must be determined experimentally and cannot be predicted from the coefficients in the balanced chemical reaction.
What are elementary steps in a reaction mechanism?
They are individual steps that make up the overall chemical reaction, showing the sequence of bond-breaking that occurs.
What is a reaction intermediate?
A stable molecule produced in one step of a reaction and consumed in a subsequent step.
What is the rate-determining step?
The slowest step in a reaction mechanism that determines the overall reaction rate.
How can the rate-determining step be used?
It can be used to predict the rate law equation for the overall reaction.
What is the significance of the order of steps in a reaction mechanism?
It provides information about the sequence of bond-breaking that occurs during the reaction.
What happens in the reaction of nitrogen dioxide with carbon monoxide?
It occurs in two elementary steps, not by a direct collision of the two molecules.
What is the relationship between rate laws and reaction mechanisms?
The rate law for a reaction is related to the mechanism, particularly the rate-determining step.
What is the role of experimental data in determining rate laws?
Experimental data is used to find the concentrations of reactants and their corresponding exponents in the rate law.
What is the overall reaction derived from elementary steps?
It is obtained by adding the individual elementary steps together.
What is the importance of understanding reaction mechanisms?
It helps in interpreting how reactions occur and predicting their rates.
Can the rate law be derived from the balanced chemical equation?
No, the rate law must be determined experimentally and cannot be directly derived from the balanced equation.
What is the significance of the coefficients in a chemical reaction?
They are used in the balanced equation but do not directly determine the exponents in the rate law.
What is the relationship between temperature and the rate constant?
The rate constant (k) changes with temperature.
What does it mean if a reactant has a concentration raised to the power of zero in the rate law?
The reaction rate is independent of that reactant's concentration.
What is the purpose of analyzing reaction mechanisms?
To understand the steps involved in a reaction and how they contribute to the overall reaction rate.
What does a rate law express?
The relationship between the rate of a reaction and the concentrations of the reactants raised to an exponent, along with the rate constant.
What is the general form of a first-order rate law equation?
rate = k[Reactant]^1
What information do rate law equations lack?
They do not provide information about the specific concentration of reactants or products over time.
What is the purpose of an integrated rate law?
To determine the concentration of a reactant at any given time.
What mathematical operation is used to generate an integrated rate law?
Integration, which is the reverse of differentiation.
What is a common example of a zero-order reaction?
A catalyzed reaction where only a small fraction of reactant molecules are available to react.
How can the order of a reaction be determined?
By generating concentration-time data and plotting it to observe the shape of the curve.
What characterizes a first-order reaction graph?
The plot of concentration versus time will yield a straight line when the natural logarithm of concentration is plotted.
What characterizes a second-order reaction graph?
The plot of 1/concentration versus time will yield a straight line.
What characterizes a zero-order reaction graph?
The plot of concentration versus time will yield a straight line with a slope equal to -k.
What is the role of the rate constant (k) in a rate law?
It is a proportionality constant that relates the rate of the reaction to the concentrations of the reactants.
What happens to the rate of a zero-order reaction if the concentration of reactants increases?
The rate remains constant and does not depend on the concentration of reactants.
In a first-order reaction, how does the rate change with concentration?
The rate is directly proportional to the concentration of the reactant.
What is the significance of the exponent in a rate law?
It indicates the order of the reaction with respect to that reactant.
Why are zero-order rate equations less common?
Because most reactions depend on one or more reactant concentrations.
What is the integrated rate law for a first-order reaction?
ln([A]t/[A]0) = -kt
What is the integrated rate law for a second-order reaction?
1/[A]t - 1/[A]0 = kt
What is the integrated rate law for a zero-order reaction?
[A]t = [A]0 - kt
What does the slope of the concentration vs. time graph indicate for a zero-order reaction?
The slope is equal to -k, indicating the rate of reaction.
How does the concentration of a reactant change over time in a first-order reaction?
It decreases exponentially over time.
What is the relationship between reactant concentration and reaction rate in a second-order reaction?
The rate is proportional to the square of the concentration of the reactant.
What effect does temperature have on reaction rates?
Reactions proceed faster at higher temperatures due to increased molecular energy.
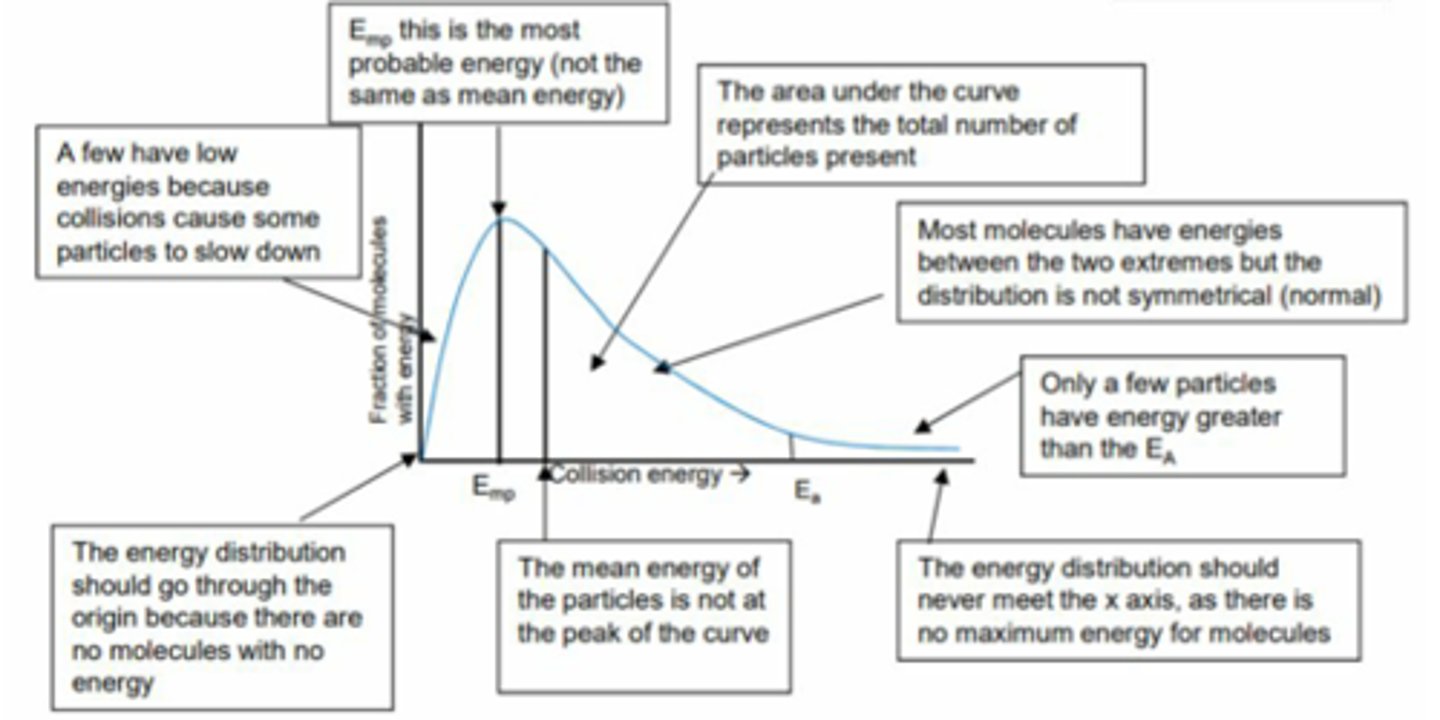
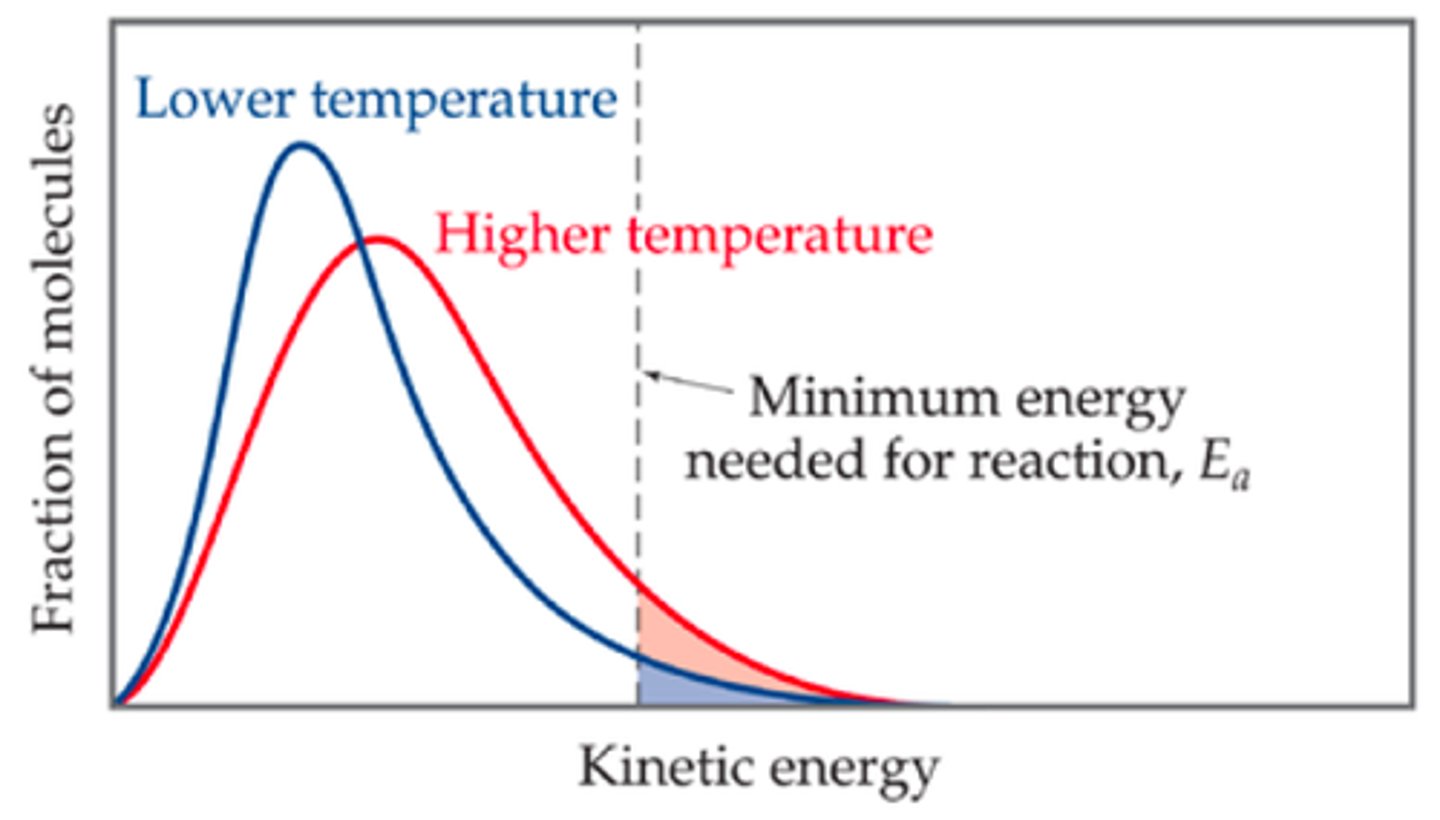
What is the Maxwell-Boltzmann distribution?
It shows the distribution of kinetic energies for all molecules in a sample.
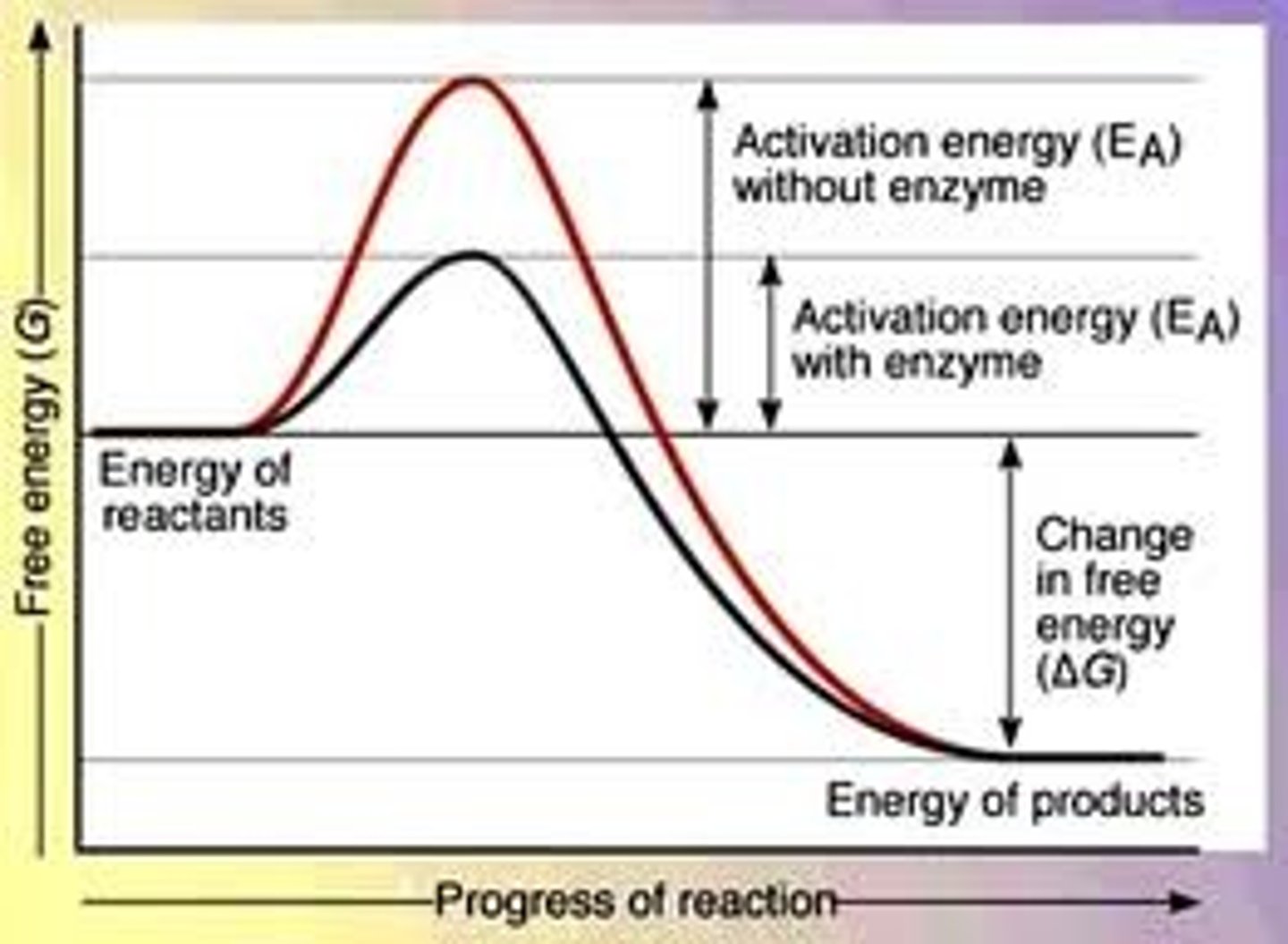
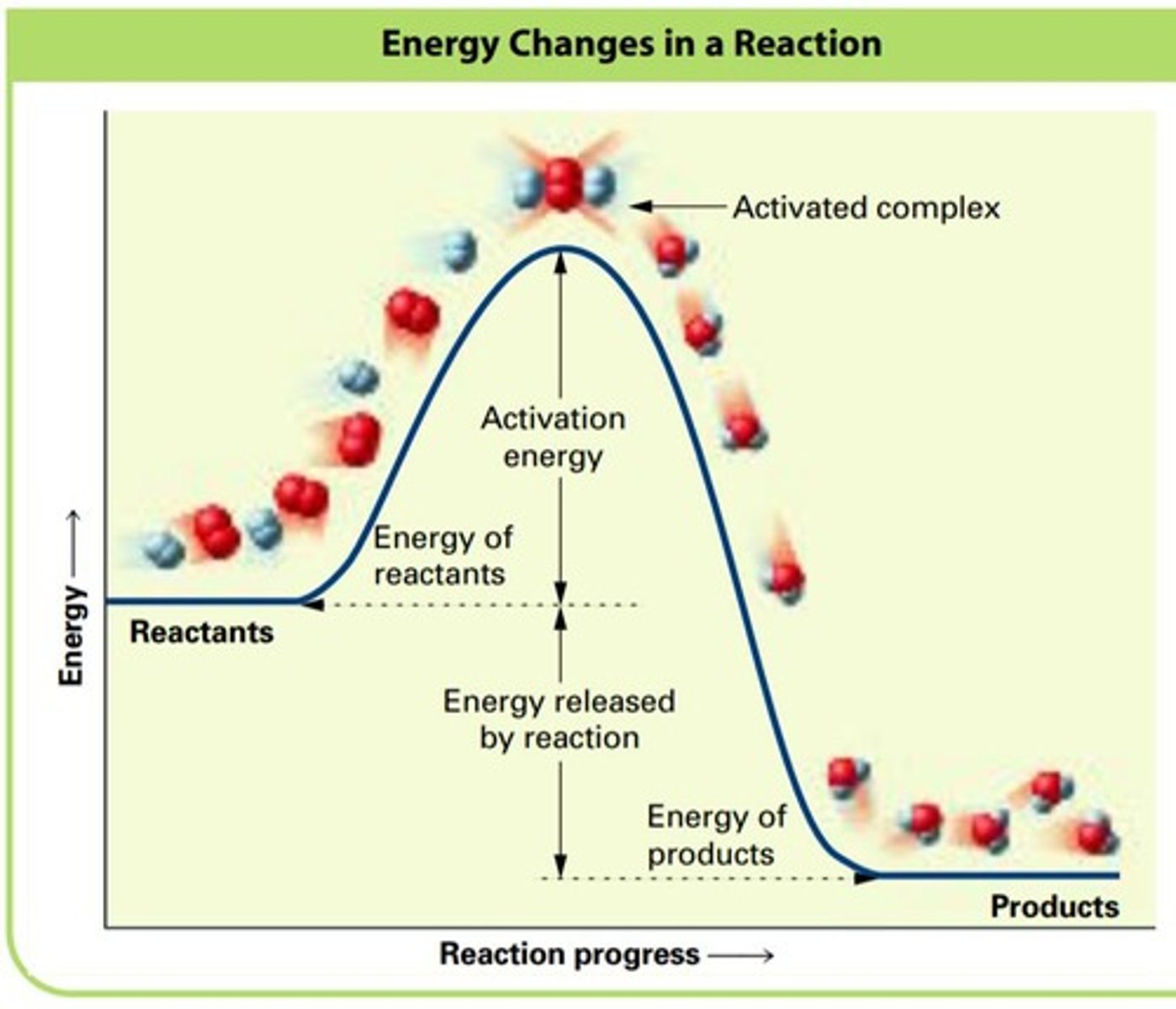
What is activation energy (Ea)?
The energy required to undergo a reaction.
How does increasing temperature affect the Maxwell-Boltzmann distribution?
It shifts the distribution curve to the right, increasing the percentage of molecules with energy greater than Ea.
What happens to the rate of reaction when the concentration of a reactant increases?
The number of collisions increases, making successful collisions more likely.
What are the two features required for a successful collision?
The collision must occur with enough force and the appropriate geometry.
How does increasing the surface area of a solid reactant affect reaction rates?
It exposes more of the reactant, increasing the likelihood of successful collisions.
Give an example of increasing the surface area of a solid reactant.
Cutting a piece of metal into small fragments or crushing a solid into powder.
Why do reactions with reactants in the same state proceed faster?
Due to better mixing and more collisions.
What is the difference between exothermic and endothermic reactions?
Exothermic reactions have reactants with higher energy than products, while endothermic reactions have reactants with lower energy than products.
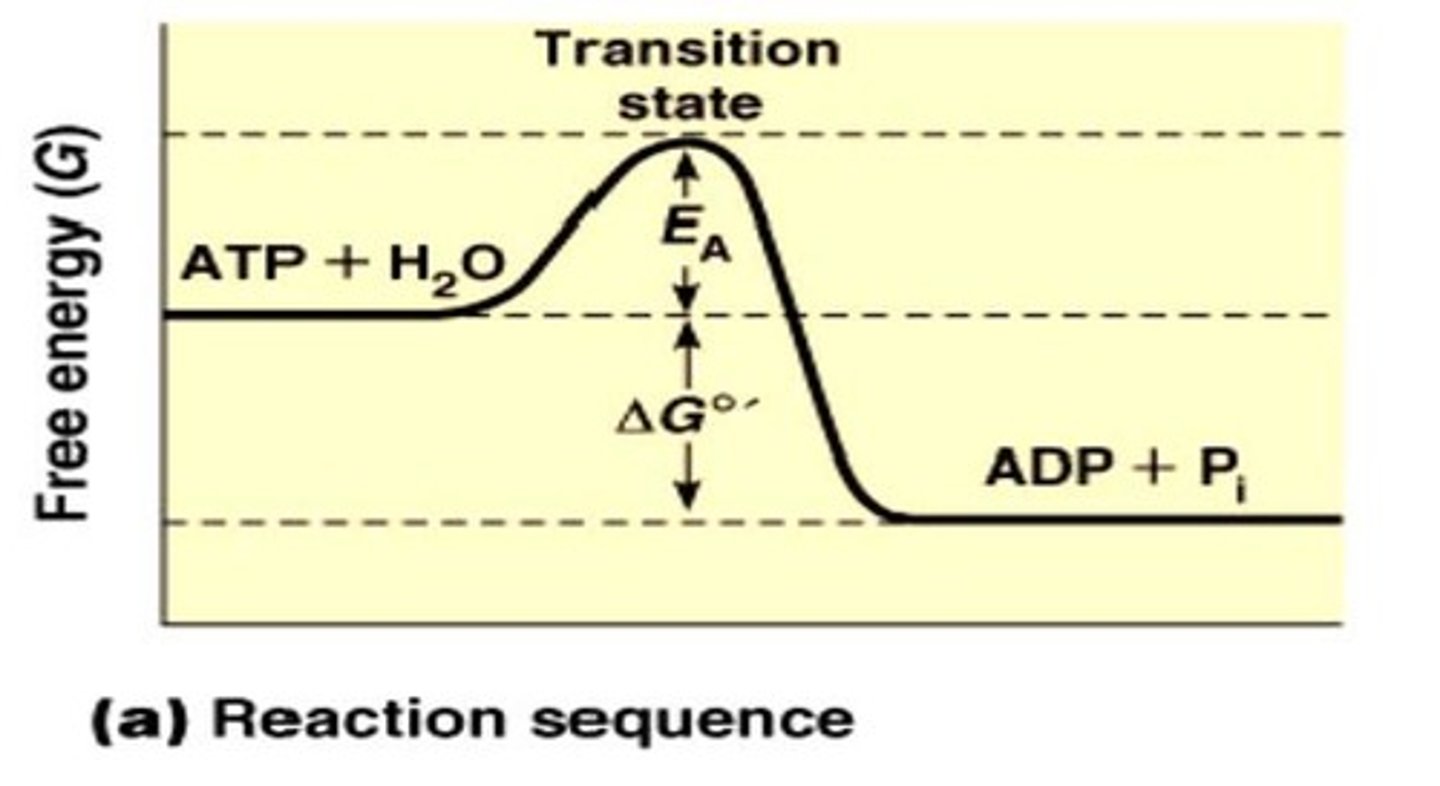
What is the transition state in a chemical reaction?
The peak of the activation energy hill, where reactants may become products or revert back.
What is an activated complex?
An unstable short-lived species that is a hybrid of reactants and products at the transition state.
How does increasing temperature affect the activation energy barrier?
It does not change the size of the barrier; it supplies more reactants with enough energy to overcome it.
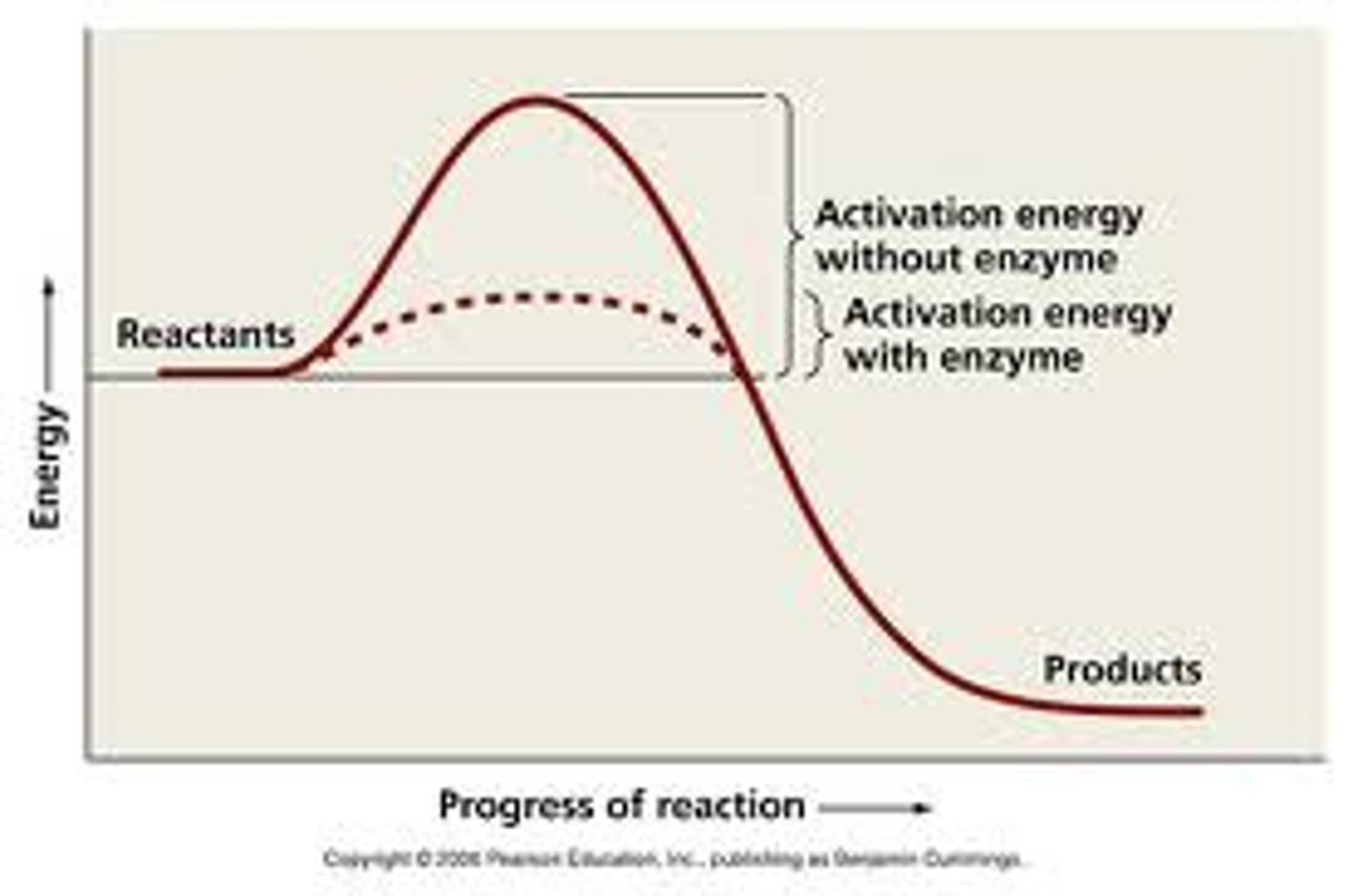
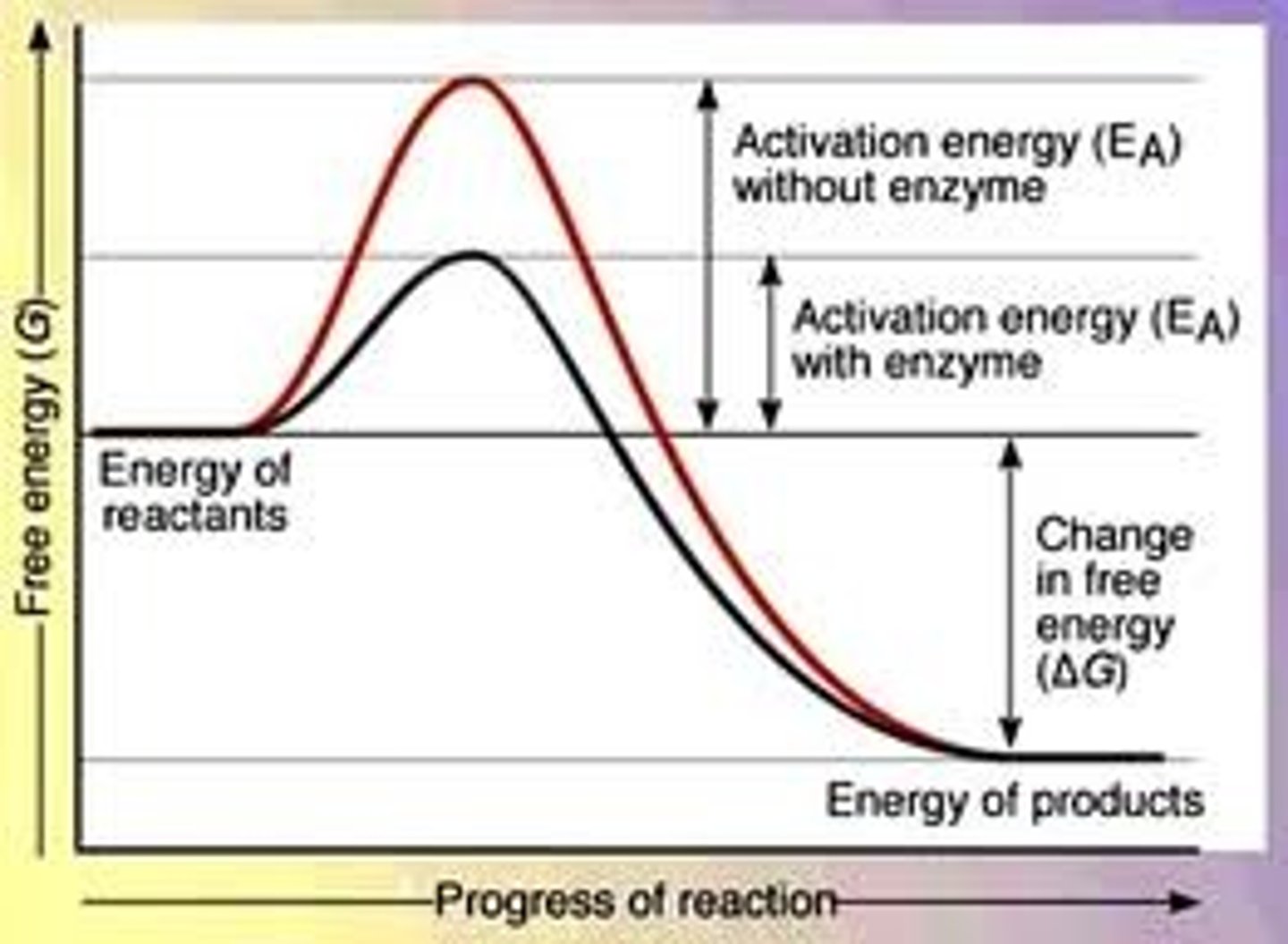
What is a catalyst?
A molecule that provides an alternate pathway for a reaction, lowering the activation energy.
What is the effect of lowering activation energy on enthalpy change?
Lowering activation energy has no effect on the enthalpy change.
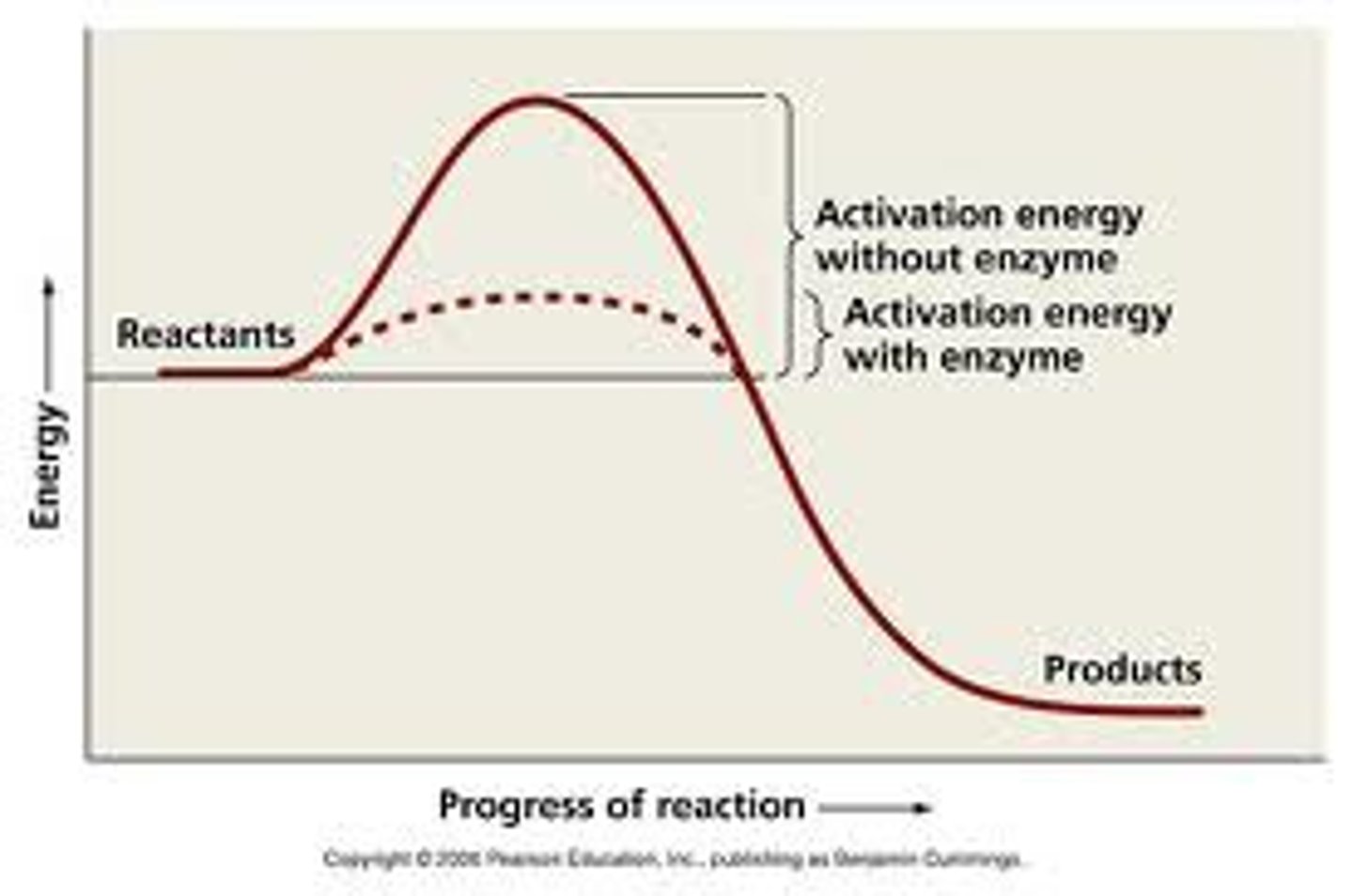
What is the relationship between activation energy and reaction speed?
The higher the activation energy barrier, the slower the reaction will be.
What can happen if too much heat is applied to a reaction?
It may decompose the reactants before they can react.
What should you do to safely speed up a reaction?
Use a catalyst.
What is the significance of the area under the curve in a Maxwell-Boltzmann distribution?
It represents the percentage of molecules with energy greater than the activation energy.
What is the effect of the nature of reactants on reaction rates?
Reactions with reactants in the same state are faster due to better mixing.
What are the two types of energy changes in chemical reactions?
Exothermic and endothermic.
What does the rate constant (k) incorporate?
The effects of temperature, nature of reactants, and surface area of solid reactants.
What is catalysis?
Catalysis is the process of increasing the rate of a chemical reaction by the addition of a substance known as a catalyst.
Why is catalysis important in industrial processes?
It can provide significant cost savings and enable reactions that would not occur without a catalyst.
What happens to the rate of a reaction in the absence of a catalyst?
The rate may be too slow, rendering the reaction impractical.
Give an example of a reaction that requires a catalyst.
The addition of hydrogen to a double bond in organic chemistry.
What role does palladium play in a car's catalytic converter?
Palladium acts as a catalyst to convert toxic combustion products into harmless substances quickly.
What are multi-step energy diagrams used for?
They illustrate reactions that occur in multiple elementary steps, each with different activation energies.
What is an activation energy?
The minimum energy required for a reaction to occur.
What is a rate-determining step?
The slowest step in a multi-step reaction that determines the overall reaction rate.
What is an intermediate in a chemical reaction?
A species that is produced in one step of a reaction and consumed in a subsequent step.
What is the steady-state approximation?
A method used to derive a rate law when rate information is unavailable, assuming that the concentration of intermediates remains constant.
How can the steady-state approximation help in deriving rate laws?
It allows for the estimation of reaction rates without knowing which step is the slow step.
How many elementary steps are involved in a multi-step reaction?
The number of steps can vary; it is determined by the specific reaction mechanism.
What is the relationship between catalysts and activation energy?
Catalysts lower the activation energy required for a reaction to occur.