Ch. 14 - Infrared Spectroscopy and Mass Spectroscopy
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
80 Terms
Spectroscopy
involves an interaction between matter and light
Light can take the form of…
waves of energy or packets of energy (photons).
Wavelength (λ)
distance between peaks of an oscillating field; inverse to energy
Frequency (v)
number of wavelengths that pass a point in space per time; directly related to energy.
Equation for Frequency (v)
v = c/λ
c = speed of light constant
Equation for Energy of Photon
E = hv
h = Planck’s constant (6.626 × 10-34 J x s)
Electromagnetic Spectrum
range of frequencies of light
IR (infrared) Spectroscopy
identifies functional groups
Nuclear Magnetic Resonance (NMR) Spectroscopy
radio waves; identifies the arrangement of all carbon and hydrogen in a compound.
UV-VIS Spectroscopy
visible light and UV; identifies any conjugated pi systems in a compound.
To jump orbitals…
the energy of light must match the energy between orbitals (energy gap - frequency).
Vibrational Excitement
Energy of a photon is absorbed and temporarily stored as vibrational energy.
Vibrational Excitement occurs…
If a photon possess exact energy as the energy gap, the bond can absorb the photon and promote vibrational excitement.
Molecular bonds can vibrate by…3 examples being….
bending or stretching in multiple ways
scissoring bend, symmetrical stretch, and asymmetrical stretch.
Energy gaps between vibrational states is dependent on…
the nature of the bond
An IR spectrophotometer measures…
the percent transmittance (as frequency).
Absorption Spectrum
a plot that measures the percent transmittance or absorption as a function of frequency; used in IR and UV-VIS spectroscopy.
Location of each signal is more often reported in terms of…
wavenumber (ṽ)
Wavenumber Equation
ṽ = v/c
IR Absoprtion Spectrum ranges from…
400 to 4000 cm-1
Higher wavenumber (ṽ) =
higher frequency and higher energy
A signal/peak on the spectrum has 3 important characteristics:
wavenumber, intensity, and shape
Wavenumber for stretching vibration depends on:
bond strength and mass difference of bonded atoms (shown by Hooke’s Law).
Hooke’s Law
allows us to approximate the frequency of vibration for a bond between two atoms of mass m1 and m2.
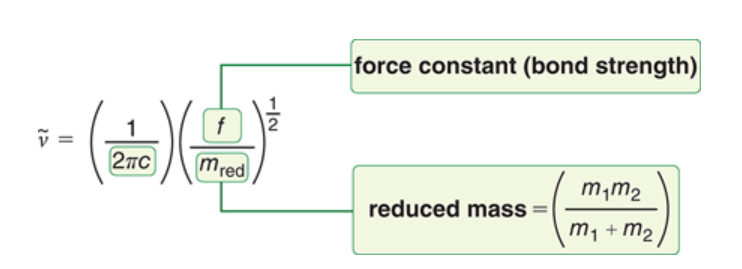
Smaller atom bonds give bonds that…
vibrate at higher frequencies, corresponding to a higher wave number
Stronger bonds will vibrate at…
higher frequencies, corresponding to a higher wavenumber.
Higher mass atoms in bond =
lower stretching frequency and lower wavenumber
Wavenumber Range
4000 - 2700 cm-1 = bonds to H
2300 - 2100 cm-1 = triple bonds
1850 - 1600 cm-1 = double bonds
1600 - 400 cm-1 = single bond
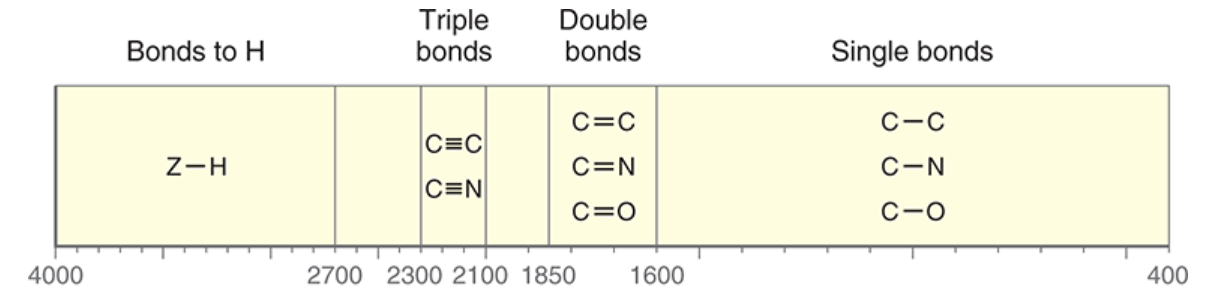
Diagnostic Region of IR Spectra
greater than 1500 cm-1 (includes bond to H, triple bonds, and double bonds); provides clear information.
Fingerprint Region of IR Spectra
below 1500 cm-1 (includes single bonds); many signals, difficult to analyze.
Higher the s character of a carbon in a C-H bond…
the stronger the C-H bond and the higher stretching frequency of the bond.
sp3 → ~2900 cm-1
sp2 → ~3100 cm-1
sp → ~3300 cm-1
Alkyl C-H bonds…
just under 3000cm-1
Alkenyl and Alkynyl bonds…
are over 3000cm-1; the absence of a signal above 3000cm-1 does not always indicate the absence of a alkene or alkyne.
The more delocalized the p electrons (resonance)…
the weaker the p bond and lower the stretching frequency
additional resonance structure → more single bond character → lower wavenumber → lower stretching frequency
Conjugated carbonyls (double bonds between carbon and oxygen atom) have…
lower stretching frequency and lower wavenumber
Intensity (signal characteristic)
the strength of IR signals; strong signal = deeper peak, more light absorbed by sample
More polar a bond…
the greater the opportunity for interaction between the waves of the electrical field and IR radiation
greater bond polarity = stronger IR signal
If more light is absorbed…
percent transmittance is lower
If a bond is symmetrical…
no dipole moments; stretching frequency is not observed in IR spectrum.
When there are multiple bonds of the same type vibrating…
stronger signals are observed; often observed of C-H bonds (many in an organic compound).
Shape (signal characteristics)
broad and narrow IR signals
Affects of H-bonding on bond strength?
weakens existing bonds; lowers wavenumber
O-H (alcohol) stretching signals are often…
broad
H-bonds are often transient, meaning?
will have molecules with varying O-H bond strengths.
When an alcohol is diluted with a solvent that cannot form hydrogen bonds, what shape of signal forms?
narrow
Carboxylic acids often more pronounced because…
they can form 2 H-bonds (H-bonding dimers).
Amines IR spectra
~3000 cm-1
Primary and secondary amines exhibit different N-H stretching signals…
Secondary: exhibit one signal for N-H bonds
Primary: exhibits two signals for N-H bonds
Mechanics of Primary Amines
The two N-H bonds stretch in phase with each other; one symmetrically and the other asymmetrically).
Half symmetrically and the other half asymmetrically; gives rise to two signals.
Mass Spectroscopy
primarily used to determine molar mass and formula of a compound.
In a mass spectrometer:
compound is vaporized, ionized, then undergoes fragmentation.
masses of the ions are detected and graphed
Most common method of ionizing molecules is:
Electron Impact (EI)
sample is bombarded with a beam of high energy electrons.
usually causes an electron to be ejected from the molecule.
Mass of a radical cation is equal to…
the mass of the parent compound
If the radical cation remains intact it is known as the…
molecular ion or parent ion
Most radical cations fragment into…
a radical and a cation.
Mass-to-Charge Ratio
(m/z); detected when ions are deflected by magnetic field
In mass spectroscopy, neutrally charged fragments (radical)…
are not detected.
Mass Spectrum
shows relative abundance of each cation detected.
Base Peak
tallest peak; most abundant fragment in mass spectrum.
Peaks with a m/z less than the molecular ion represents…
fragments
M+⋅ Peak
located where m/z of the parent ion = molar mass of the compound; can be base peak.
Nitrogen Rule: Odd massed M+⋅ peak generally means…
there’s an odd number of N atoms.
Nitrogen Rule: Even massed M+⋅ peak generally means…
absence or even number of N atoms
(M+1)+⋅ Peak
more abundant the peak, the more carbon atoms in a compound.
comparing heights of M+⋅ and (M+1)+⋅ allows us to estimate number of carbons in a molecule.
(M+2)+⋅ Peak
affected by Cl and Br
compounds containing a Cl atom will have a 3:1 ratio of M+⋅ and (M+2)+⋅ peaks
compounds containing a Br atom have (M+2)+⋅ peaks equally strong as M+⋅.
Clusters on Mass Spectrum represent…
further fragmentation; loss of H atoms
To form fragments, alcohols often undergo…
alpha cleavage and dehydration
To form fragments, amines undergo…
alpha cleavage
To form fragments, carbonyls undergo…
McLafferty rearrangement
M - 15
loss of methyl group
M - 29
loss of ethyl group
M - 43
loss of propyl group
M - 57
loss of butyl group
M - 18
loss of water
M - X (where X is an even number)
McLafferty rearrangement (ketone or aldehyde)
High-Resolution Mass Spectroscopy allows…
m/z to be measured with up to 4 decimal places; used to distinguish between compounds that have the same molecular weight when rounded to nearest amu.
Saturated hydrocarbons and alkanes will always follow…
CnH2n+2
For every pi bond or ring (degree of unsaturation), the number of hydrogens…
are reduced by 2
1 degree of unsaturation =
1 unit of Hydrogen Deficiency Index (HDI)
HDI Formula
½ (2C + 2 + N - H -X), where X is a halogen