Wk 6 pt1 - gas partial pressures and delivery
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Requirements for when exercise intensity increases
An increase in need for: ATP, O2, production of lactate and CO2, heat production, and fuel requirements.
Why do cardiopulmonary responses occur?
The occur to meet the requirements that happen due to an increase in exercise intensity. However, it focuses on sending more O2 to the muscles for aerobic generation of ATP, and to remove any additional CO2.
Oxygen delivery
O2 is carried in the blood through 2 ways: dissolved in solution, and in combination with Haemoglobin (Hb) in RBS’s.
O2 being dissolved in a solution
O2 is not very soluble in blood plasma. Only a small amount can be dissolved directly into plasma, which is around 0.3 ml of oxygen per 100 ml of blood. When it is dissolved into plasma, it is described in terms of partial pressure (PO2). PO2 is around 100mmHg.
Blood transportation through being bound to haemoglobin within RBC’s.
Each Hb molecule can bind up to 4 O2 molecules. Hb serves as a carrier for oxygen, allowing for efficient O2 transport from the lungs to tissues throughout the body and facilitating the removal of CO2 from tissues to the lungs for exhalation.
What is Hb?
An iron-containing globular protein pigment.
How do you describe the amount of oxygen carried with Hb?
As a % of saturation from 0-100.
How much O2 is combined with Hb?
It depends on how much was dissolved in the plasma. So although the amount dissolved is low, it’s important in determining how much is carried with Hb, and movement of O2 between lungs, blood, and muscle.
Kinetics of dissolved gases - movement across membranes
This depends on partial pressure of each gas in each area.
At sea level what is the barometric pressure (air pressure)?
760 mmHg
What is the composition of inspired air?
21% O2 or 159mmHg
79% N2 or 600 mmHg
0.04% CO2 or < 1 mmHg
Diffusion and gas exchange
Exchange of gases between lungs and blood due to process of passive diffusion. Gas moves down a partial pressure gradient.
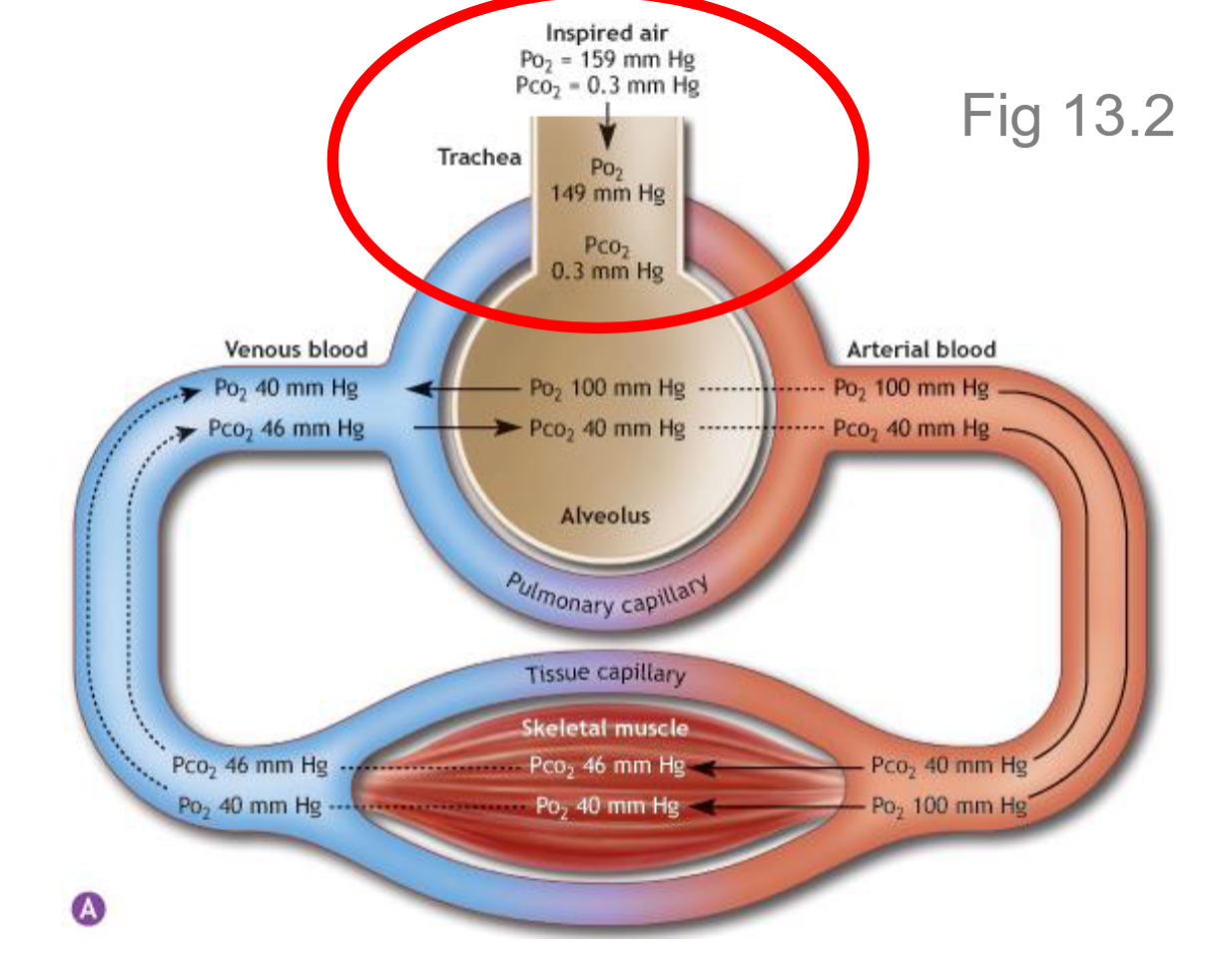
What is alveolar gas partial pressure?
It is the pressure of gases within the alveoli, which are small air sacs in the lungs where gas exchange occurs. It is primarily composed of O2 and CO2, along with small amounts of other gases.
Alveolar gas partial pressure during inspiration
Inspired air mixes with residual air in the lungs. Residual alveolar air is higher in CO2 but lower in O2. With mixing you get PO2 = 100-105 mmHg, and PCO2 = 40 mmHg.