Spectroscopy Exam 336
1/77
Earn XP
Description and Tags
OCHEM 336 spectroscopy exam (Peak is one like spike in a signal, signal is a cluster (i.e doublet, singlet), full signal is like all of the signals for one H)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
Degrees of Unsaturation Equation
DU = nc - (nH+X/2) + (nN/2) + 1
OR DU = 1/2(2C + 2 + N - H - X)
What does proximity to an electronegative atom or a pi bond do to a full signal?
It causes it to be shifted downfield
Integration numbers are _____?
relative
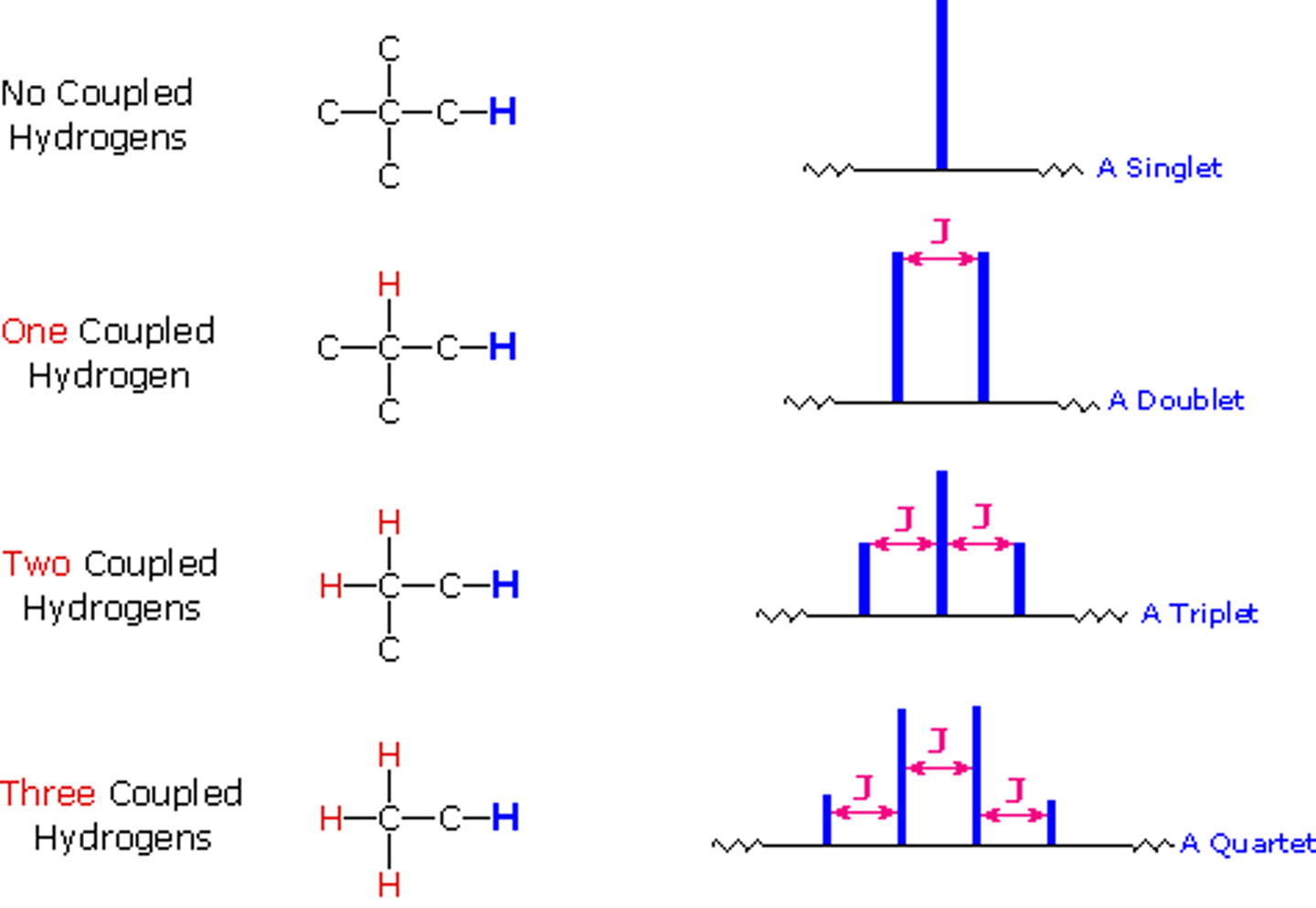
n + 1 rule (H NMR)
Peaks in a signal = individual (not type of) neighbors + 1
n + 1 rule (C NMR)
N = number of H on the carbon + 1
Coupling constants (J values) measure
the distance between peaks in Hz
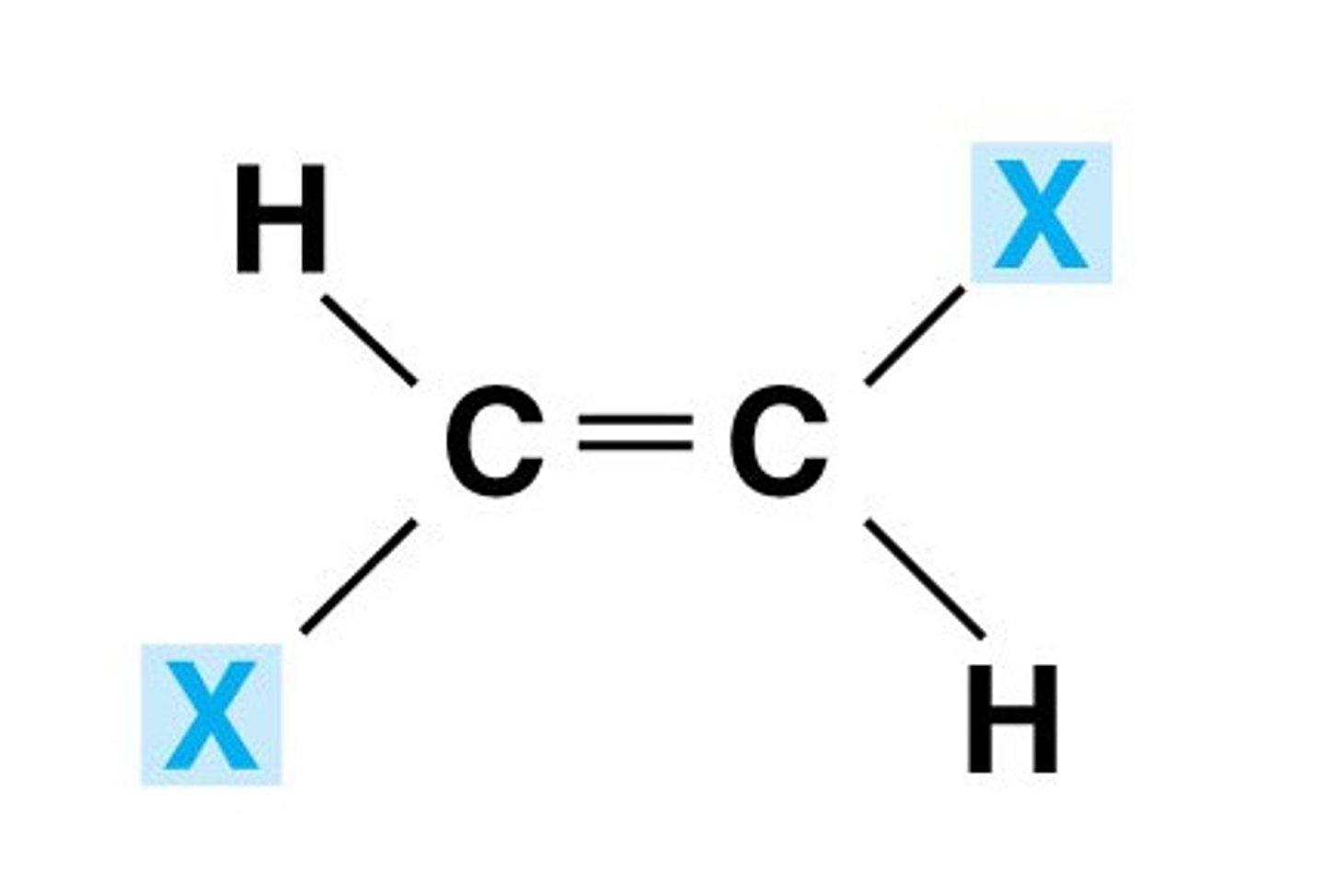
Jab of trans H alkene H
15 Hz
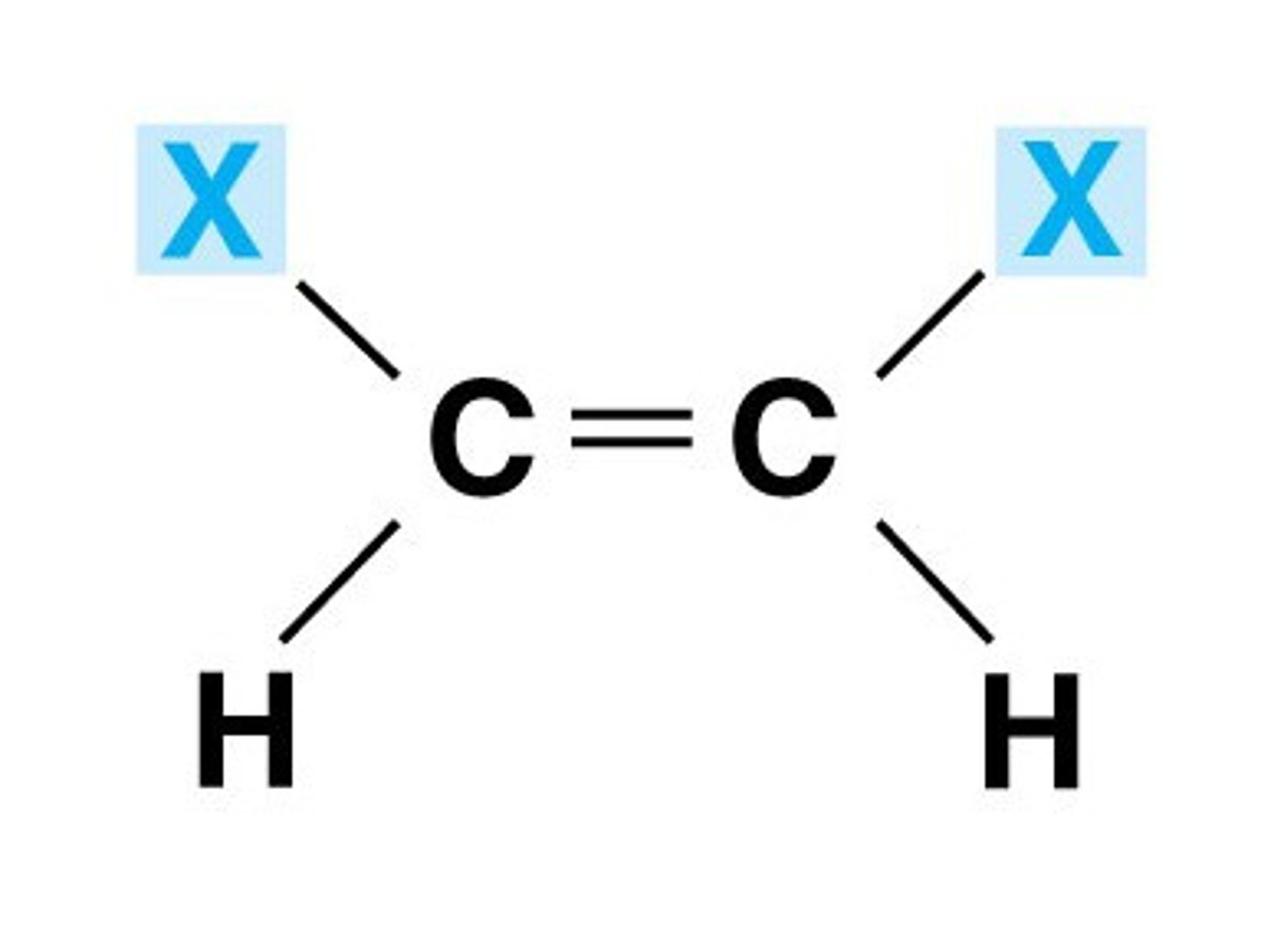
Jab of cis H alkene H
10 Hz
Jab of Geminal H on alkene
2 Hz
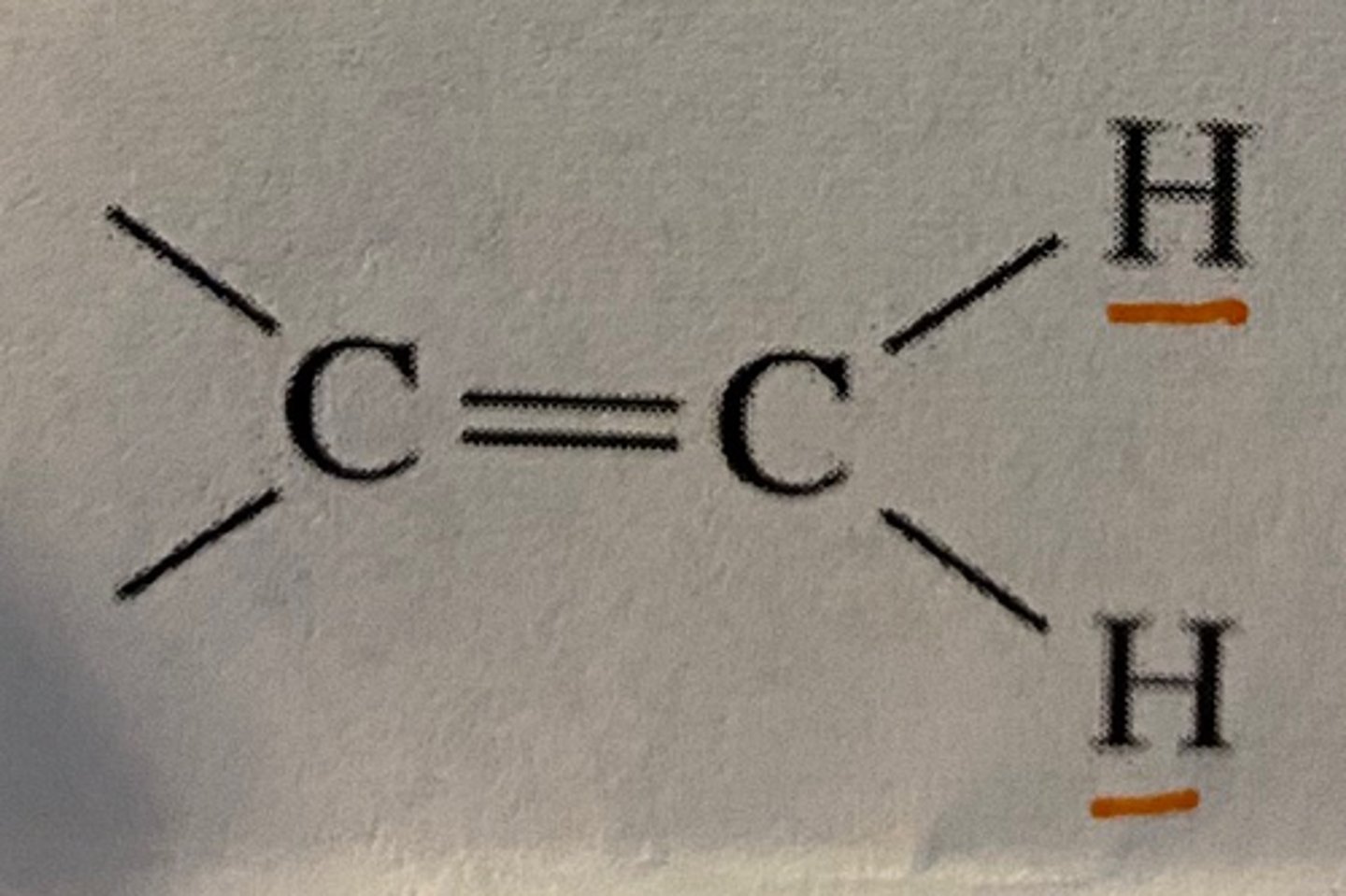
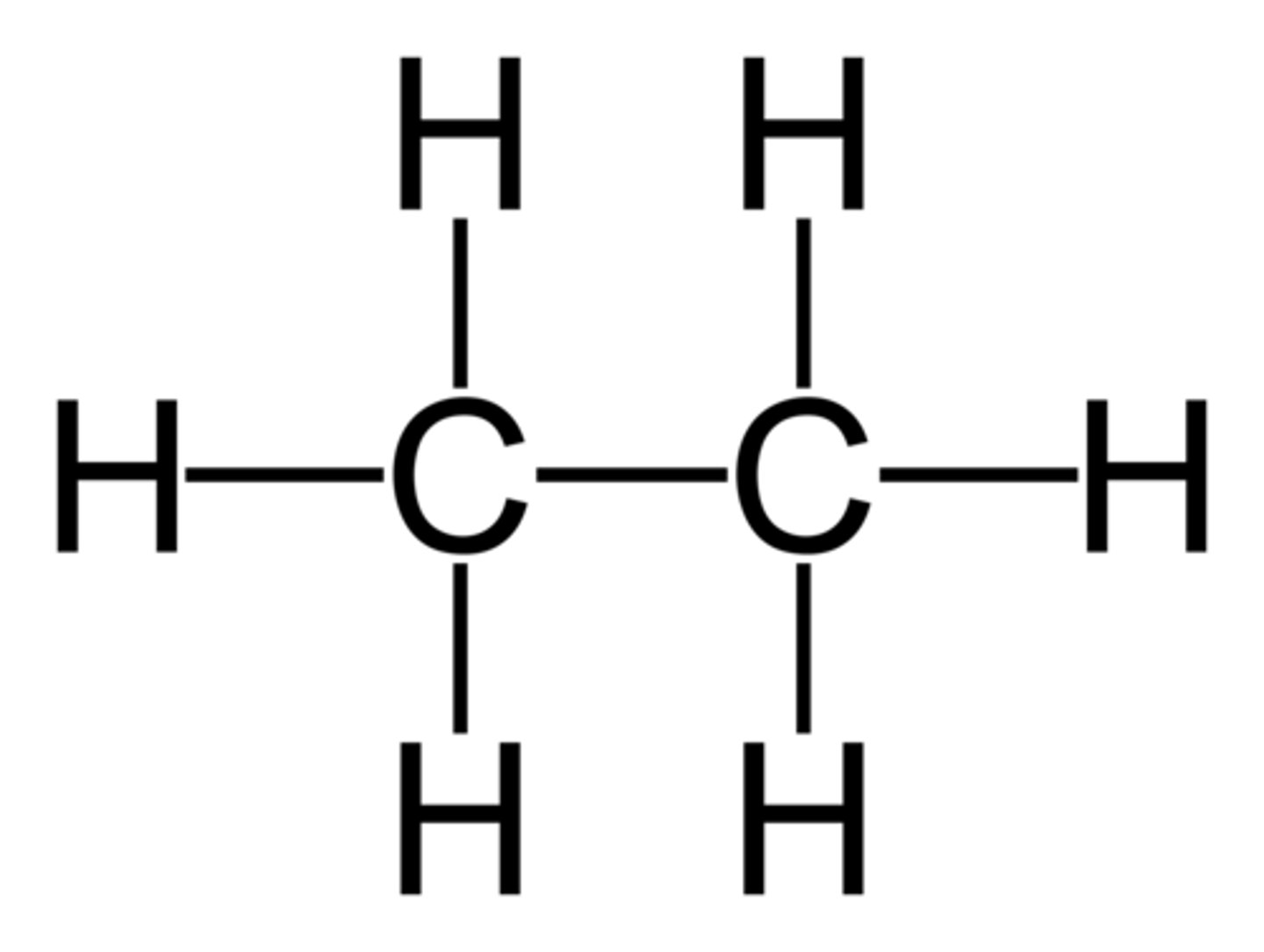
Jab of alkyl H on alkane HC-CH
7 Hz
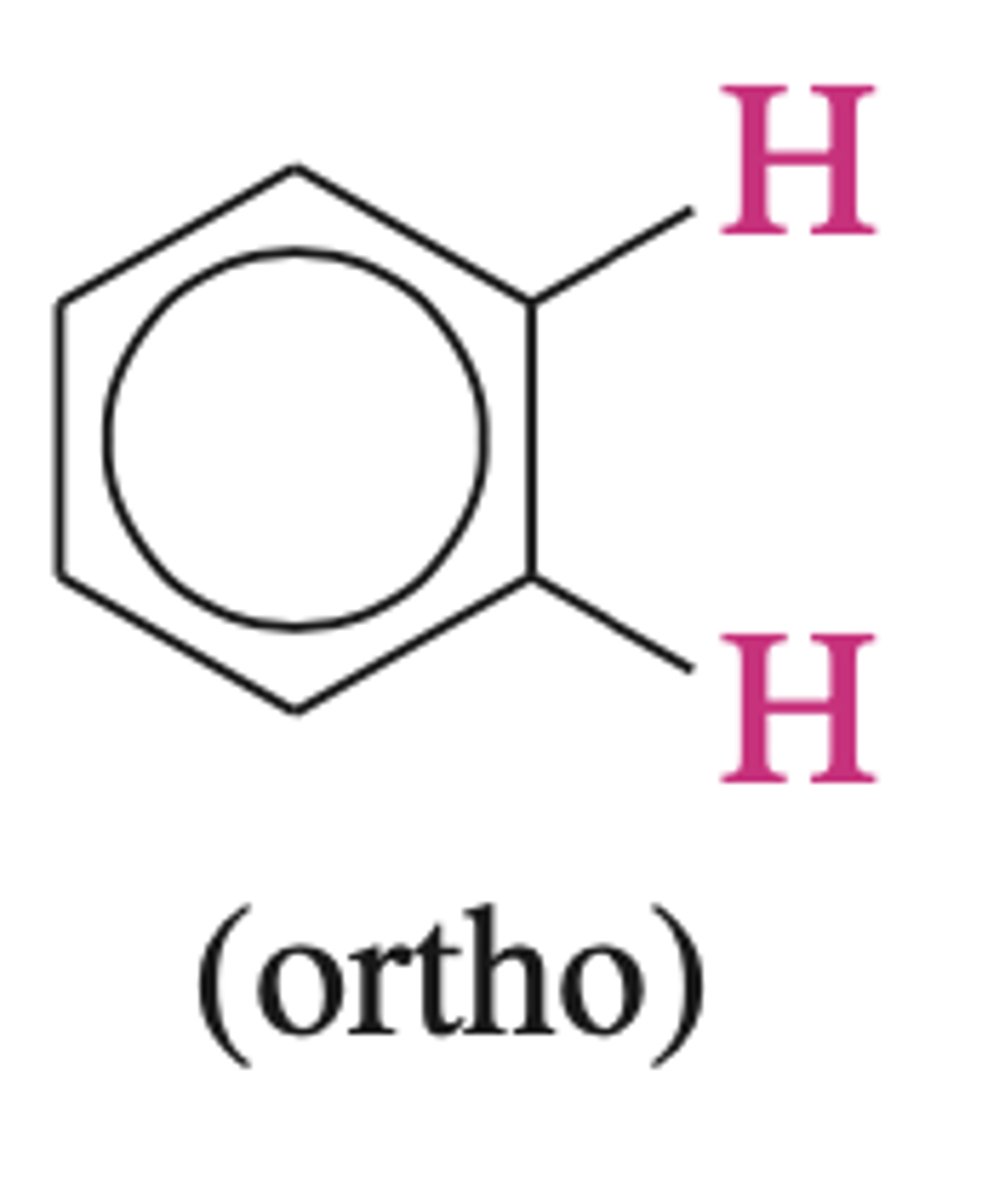
J ortho of ortho H
6.5 - 8.5 Hz
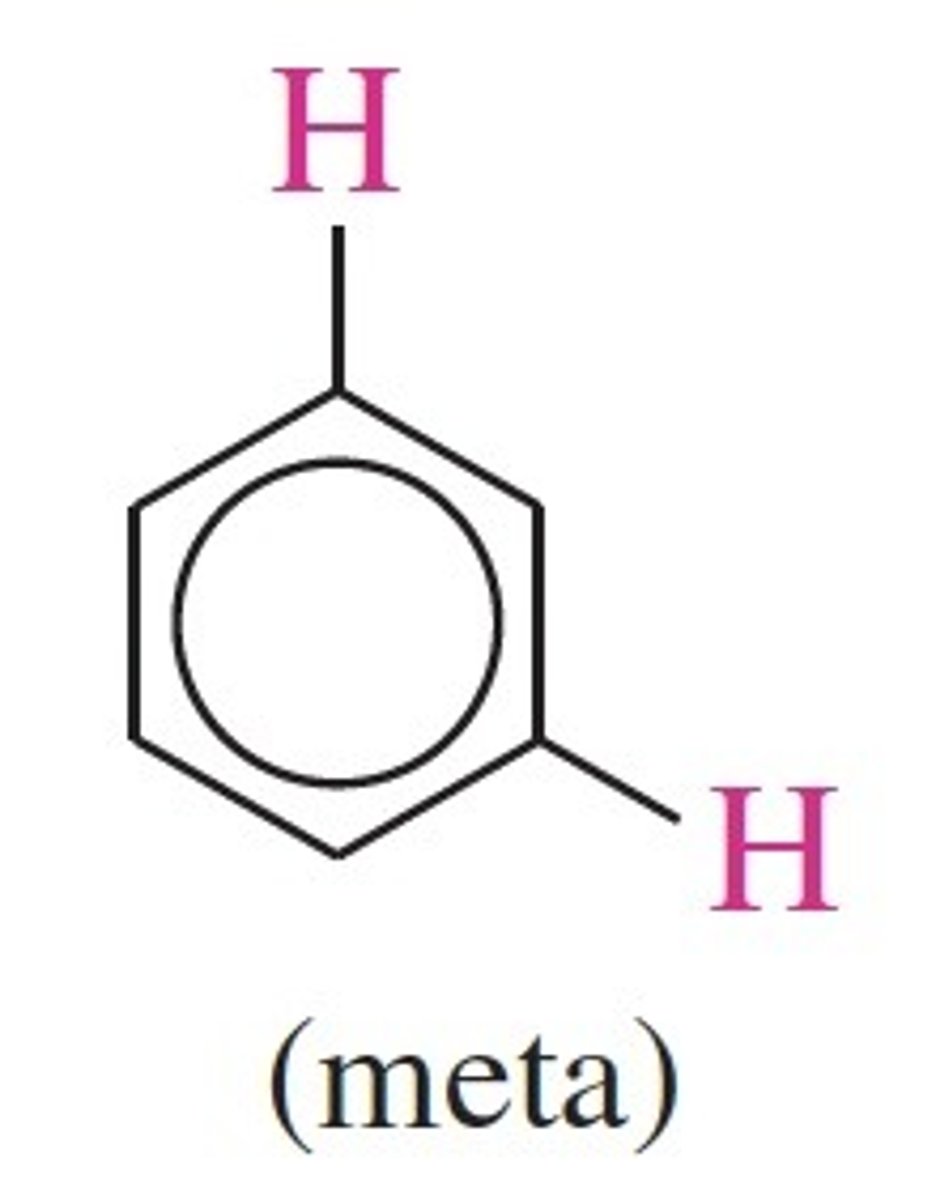
J meta of meta H
1 - 3 Hz
J para H
0 - 1 Hz
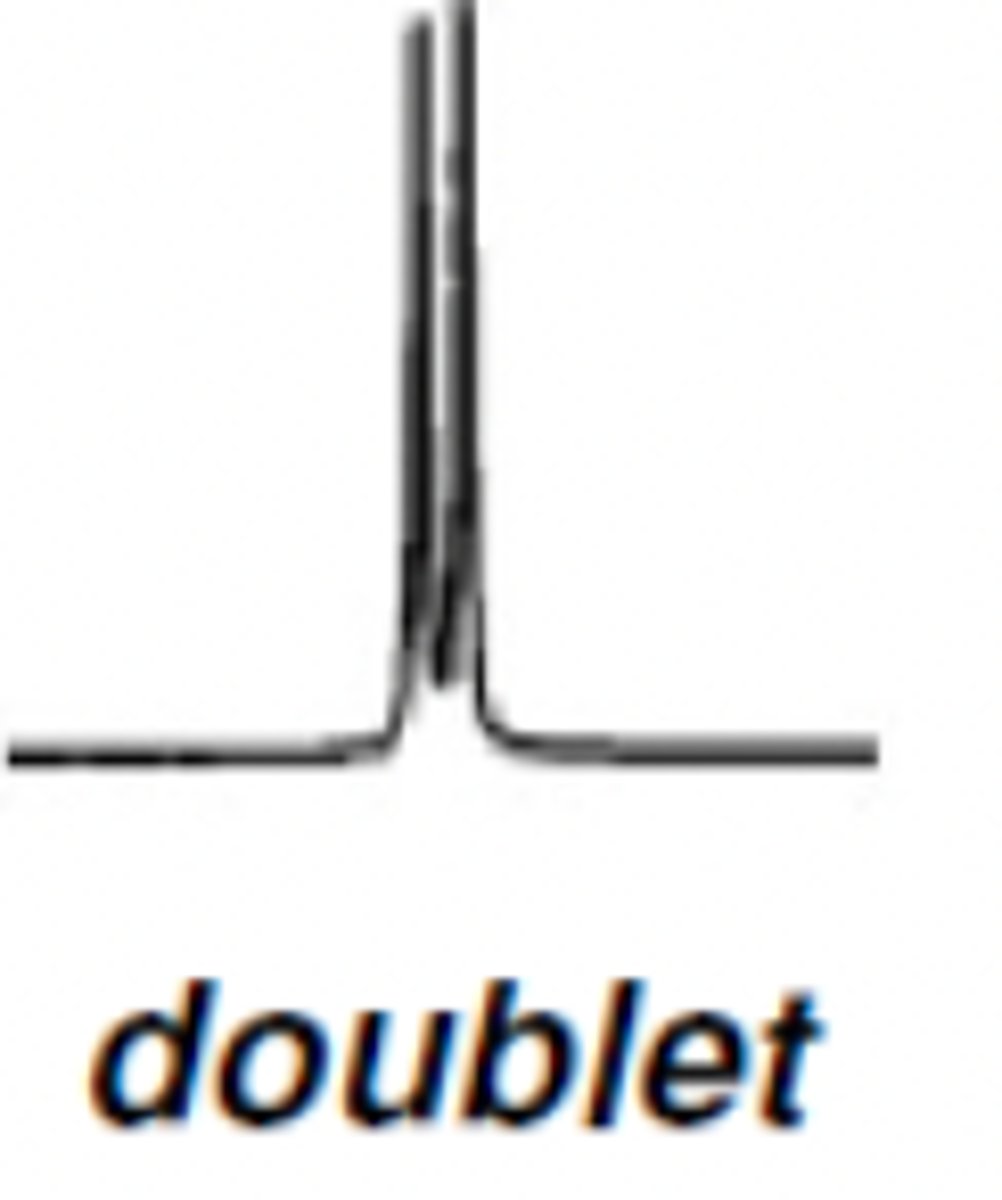
H NMR Doublet ratio
1:1
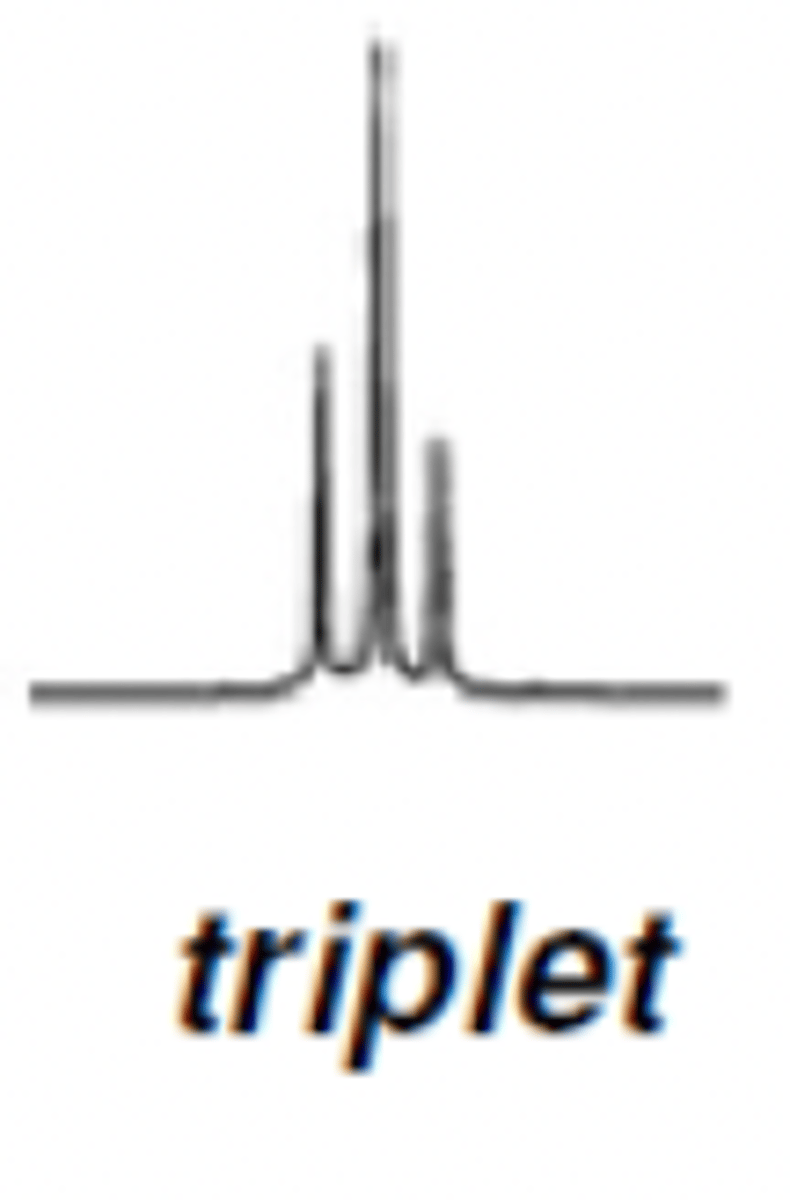
H NMR Triplet ratio
1:2:1
H NMR Quartet ratio
1:3:3:1
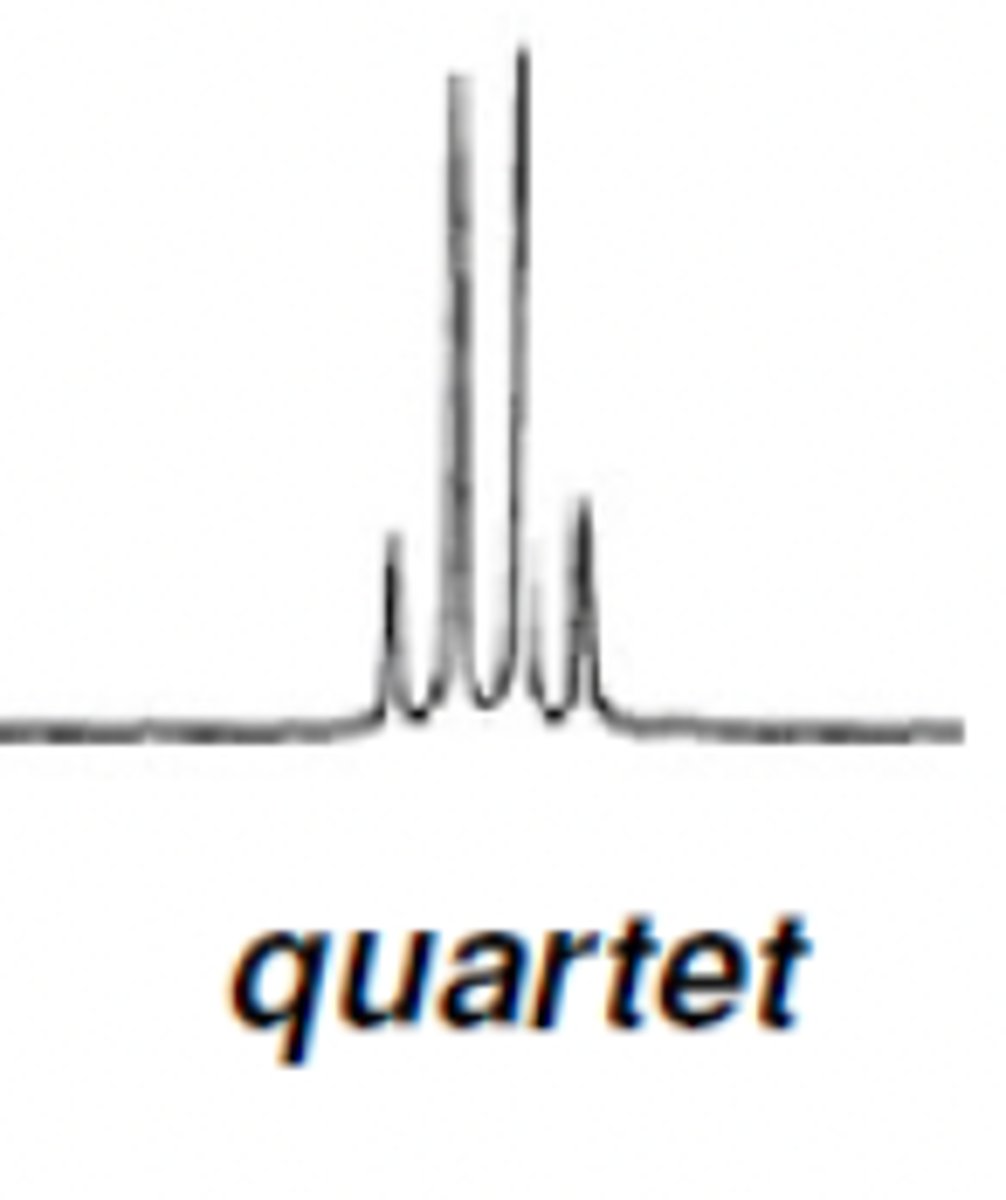
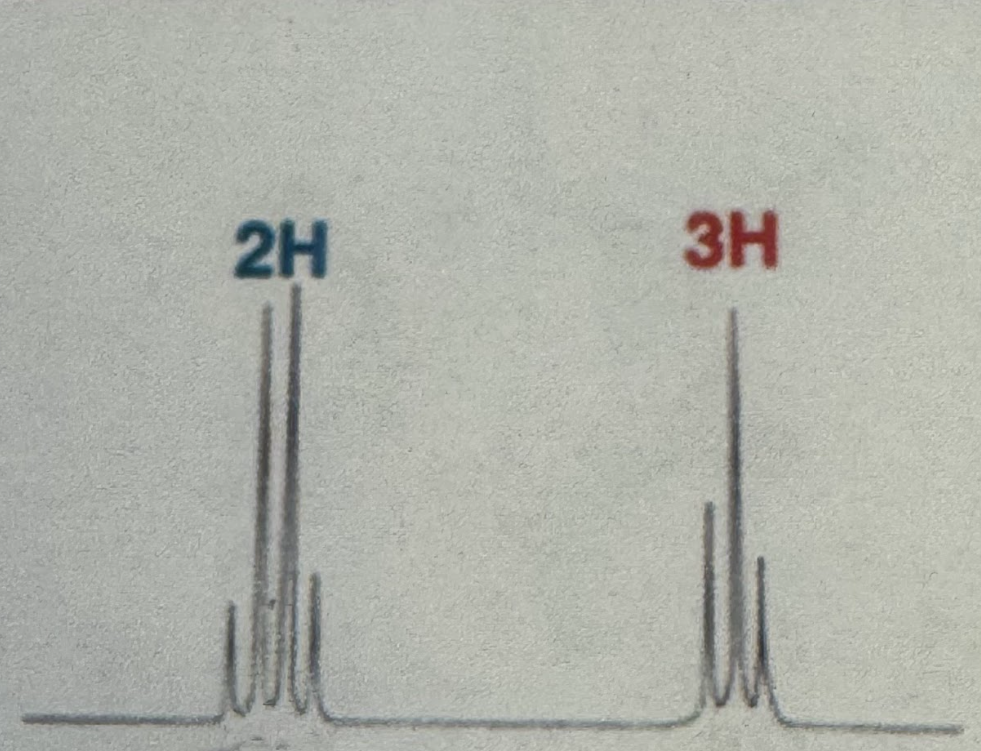
Quartet (2H) and triplet (3H)
Ethyl Splitting pattern (H NMR)
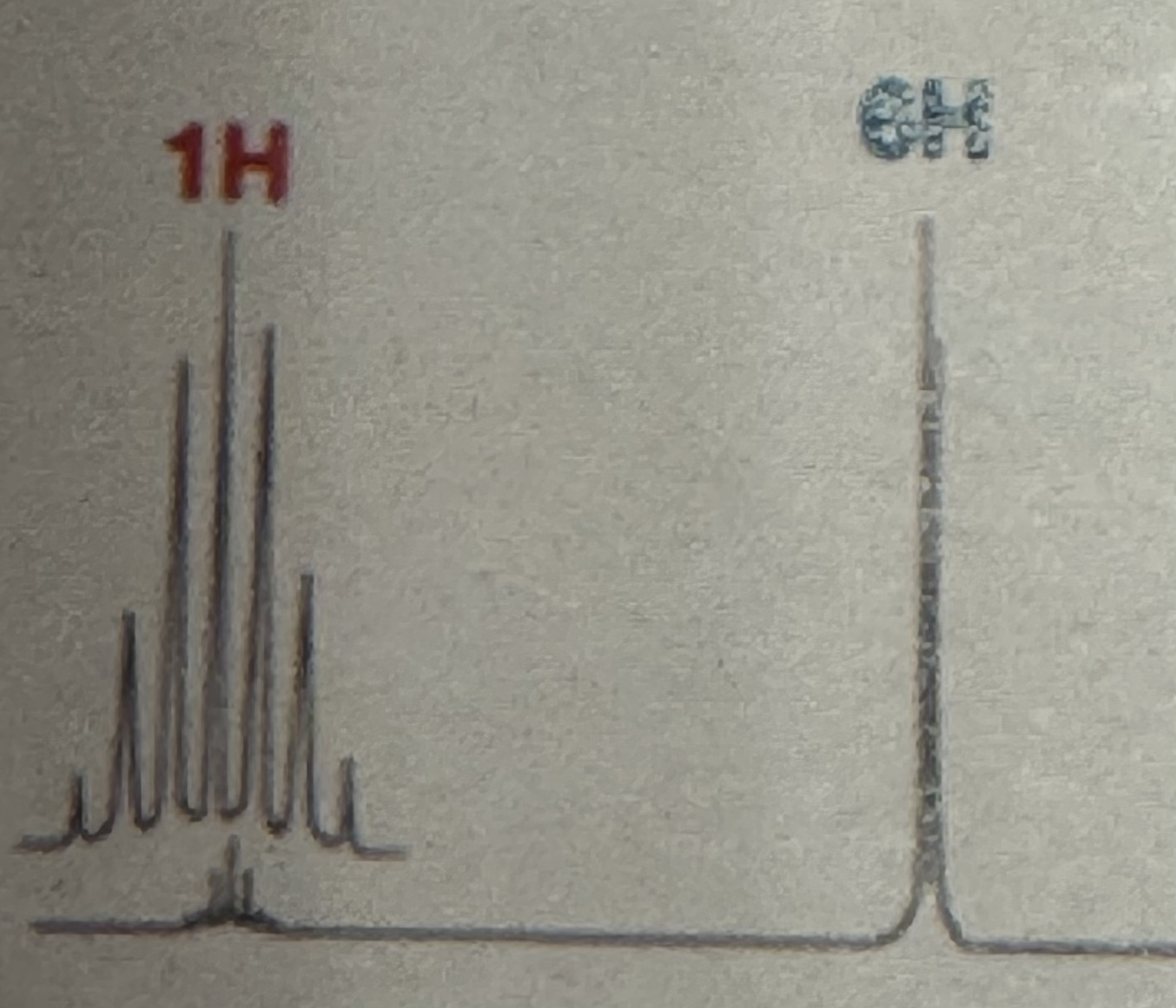
Tiny Septet (1H) and doublet (6H)
Isopropyl splitting pattern (H NMR)
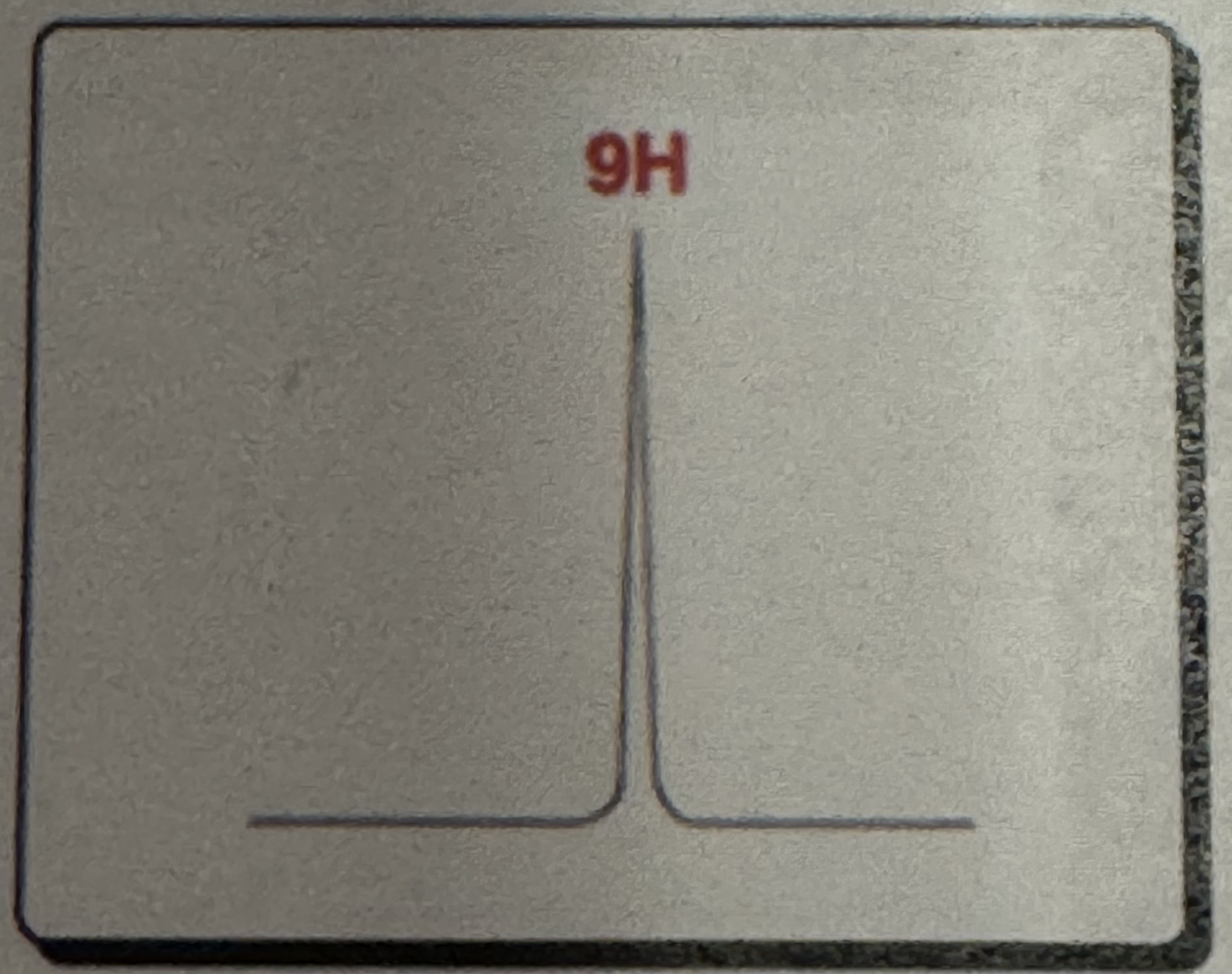
Tall singlet (9H)
Tert-Butyl Splitting pattern (H NMR)
General splitting rules
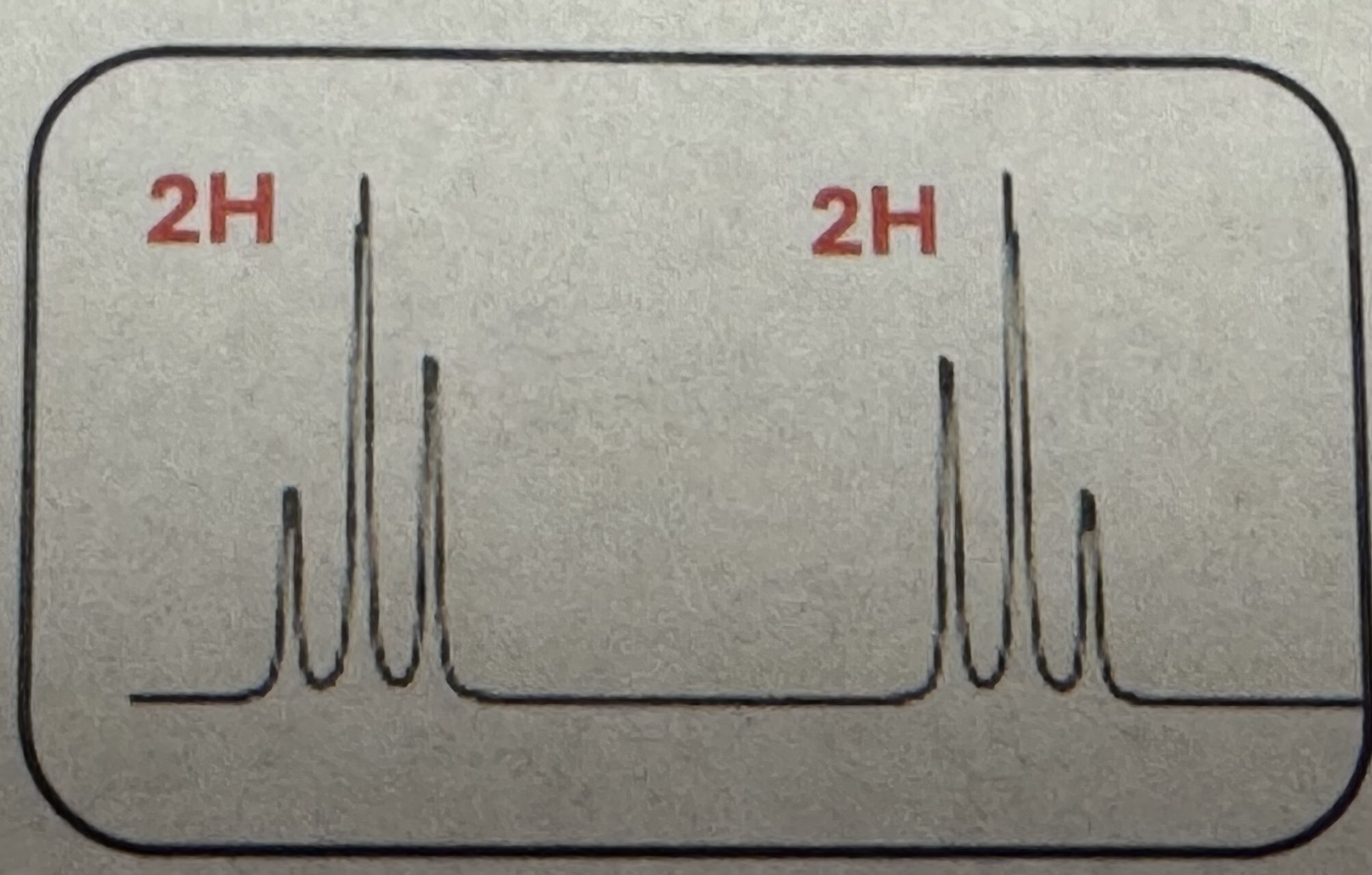
Triplet (2H) and Triplet (2H)
-CH2CH2- splitting pattern (H NMR)
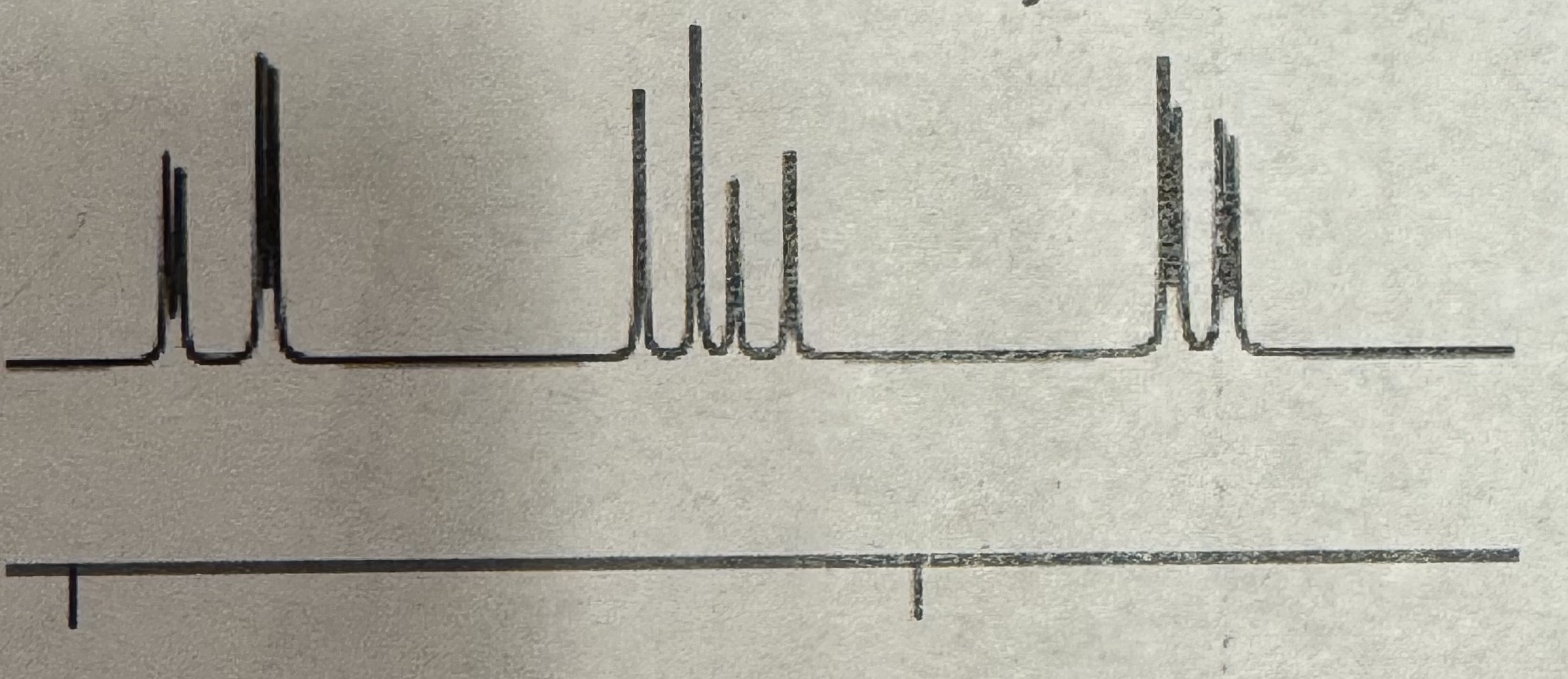
Doublet of Doublet and quartet and doublet of a doublet
Vinylic splitting pattern (H NMR)
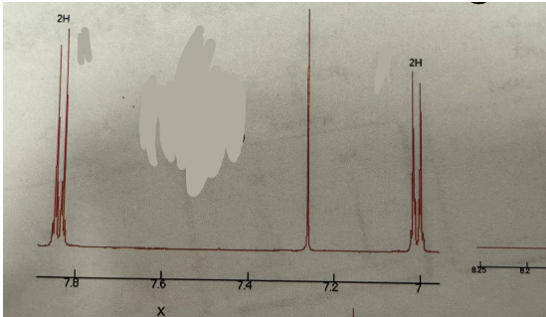
Doublet (2H) and doublet (2H)
Para splitting pattern (H NMR)
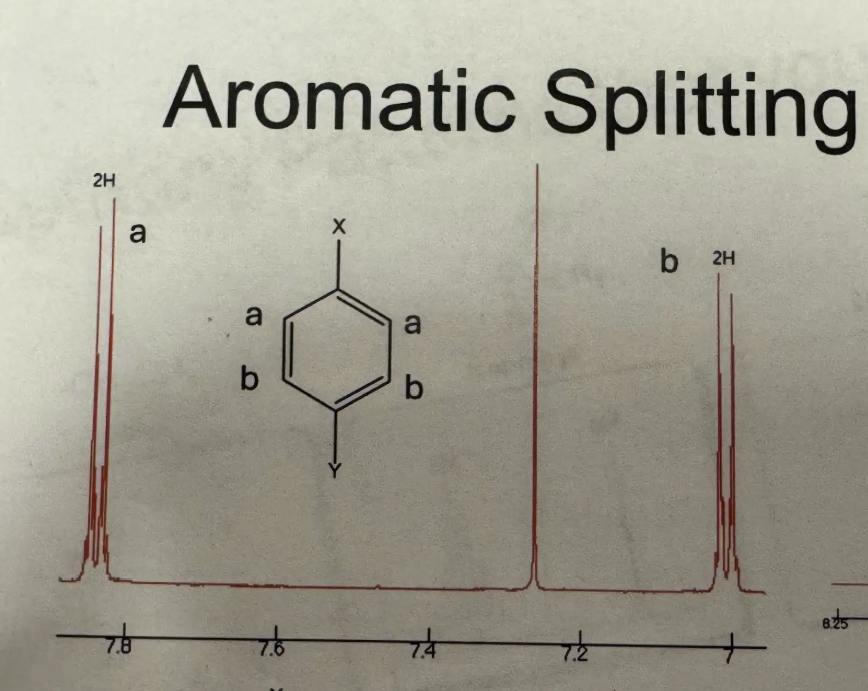
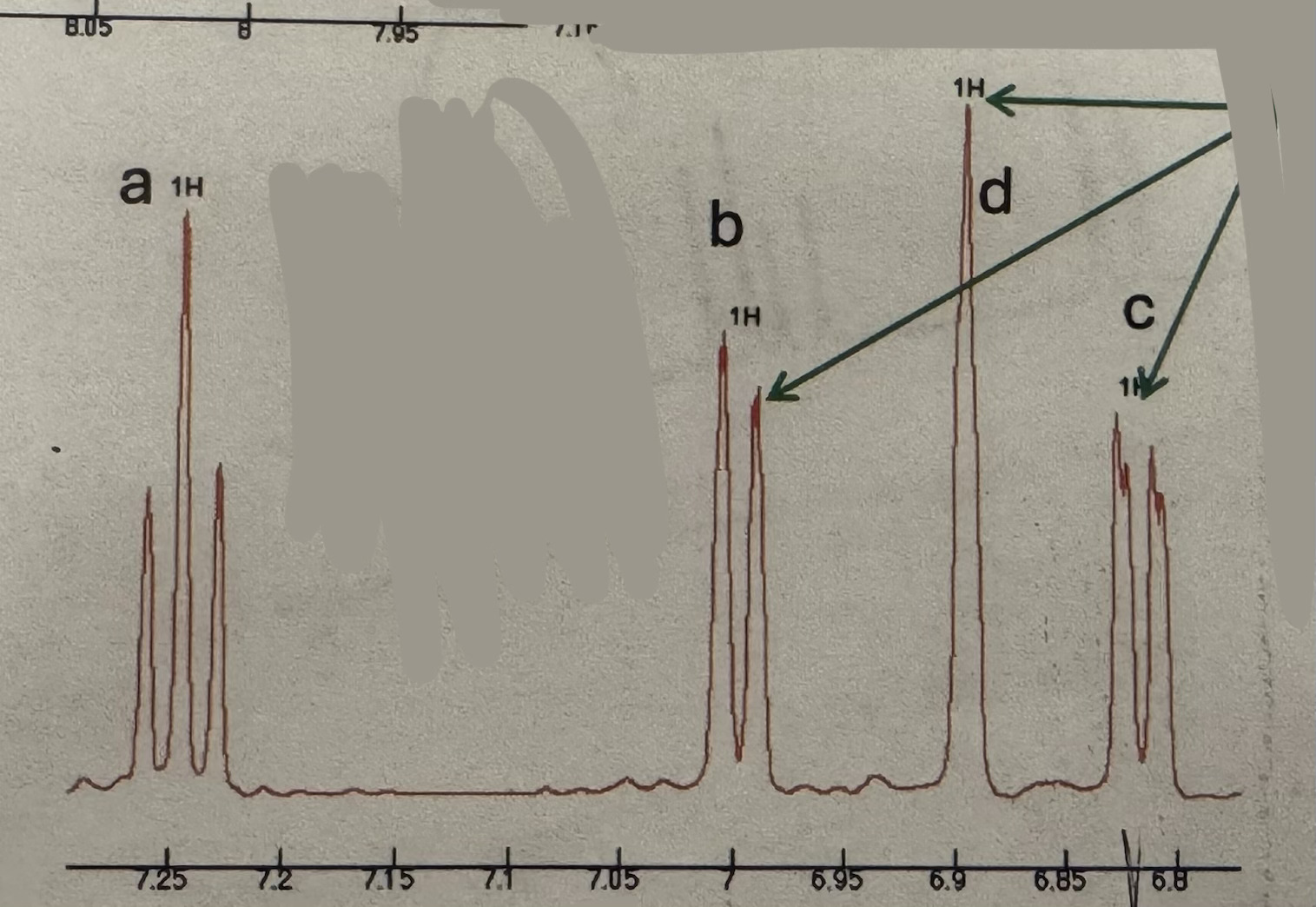
Triplet (1H ~7.25) and doublet (1H ~7) and singlet (1H ~6.9) and doublet (1H ~6.8)
Meta splitting pattern (H NMR)
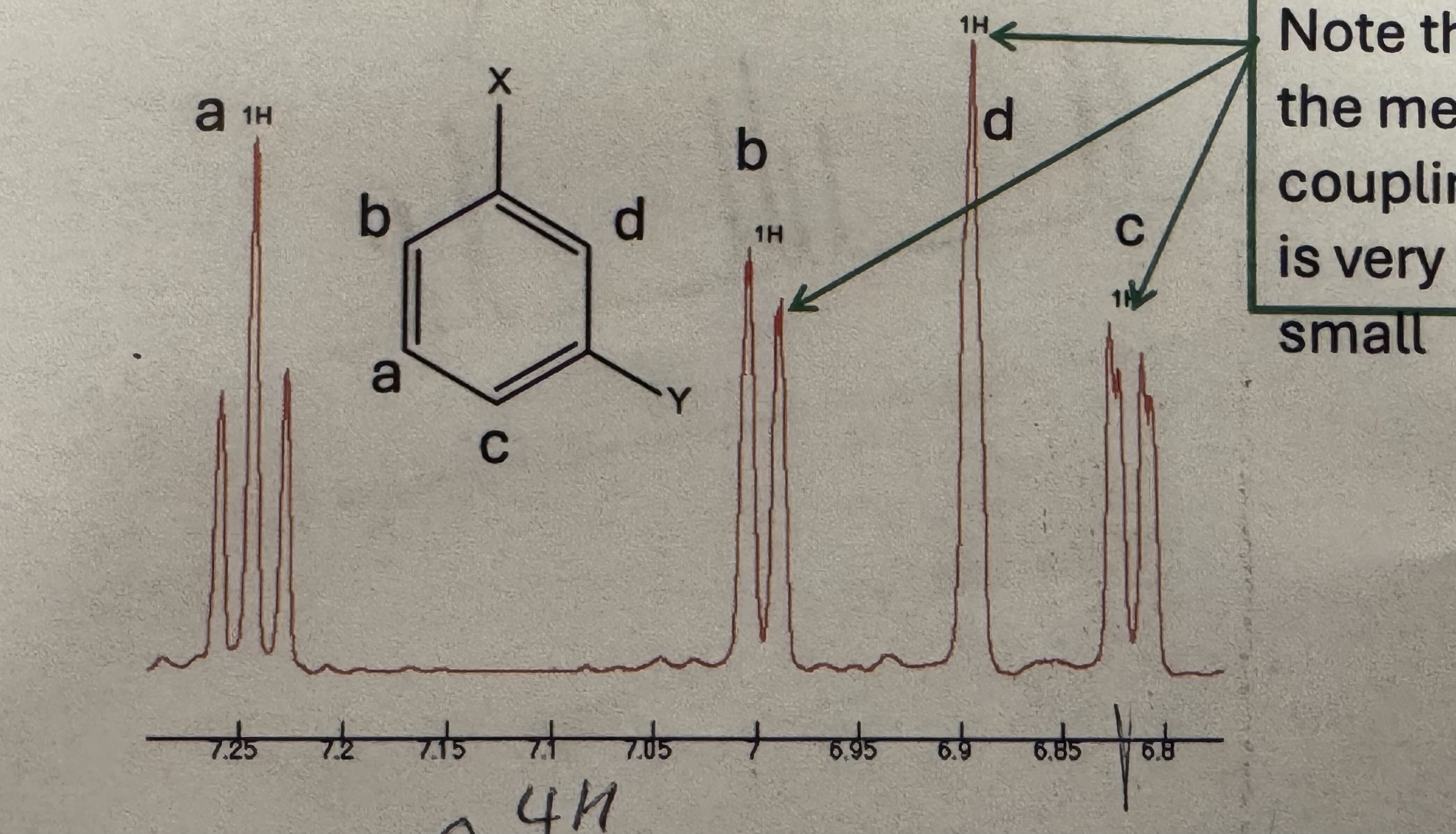
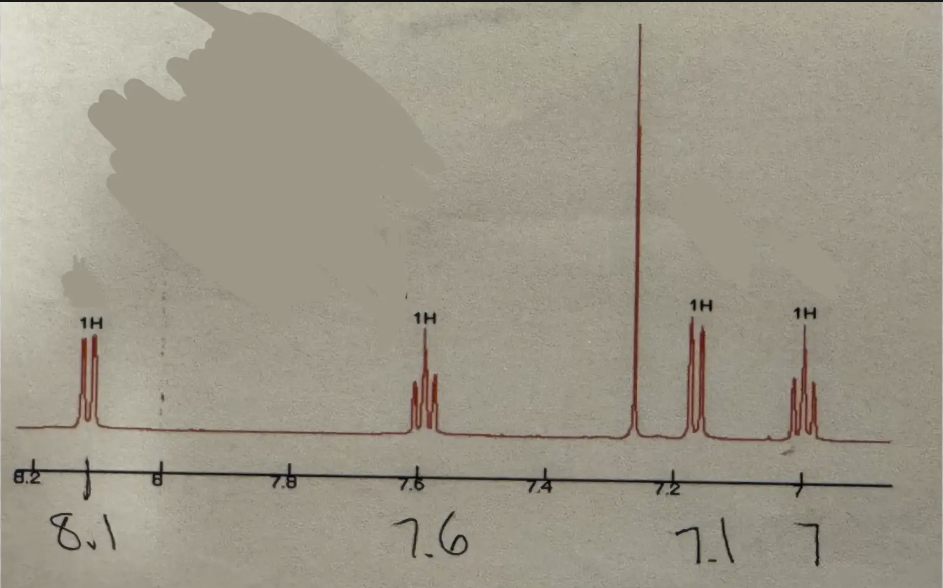
Doublet (1H ~8.1) and triplet (1H ~7.6) and doublet (1H ~7.1) and triplet (1H ~7)
Ortho splitting pattern (H NMR)
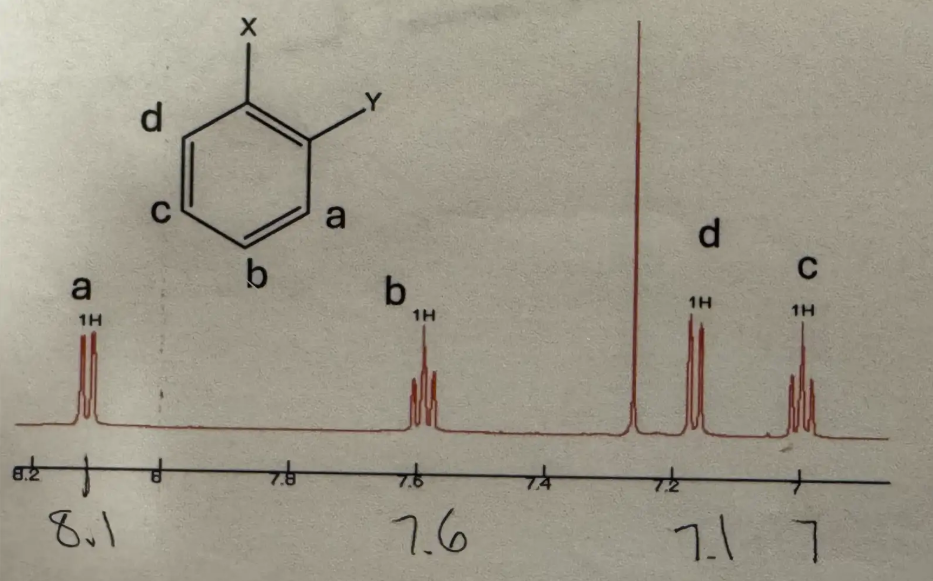
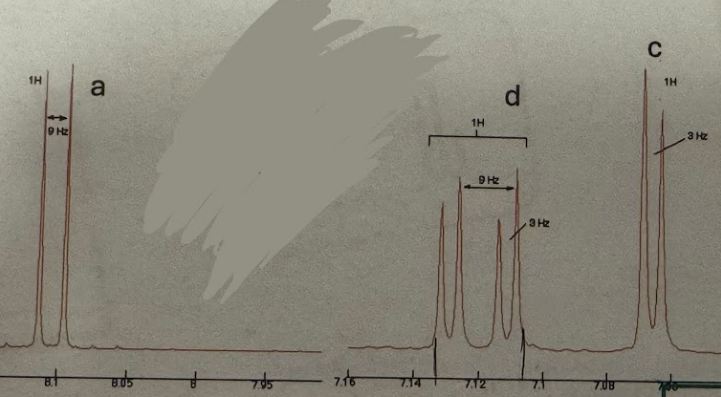
Doublet (1H ~8.1) and doublet of a doublet (1H ~7.13) and doublet (1H ~7.06)
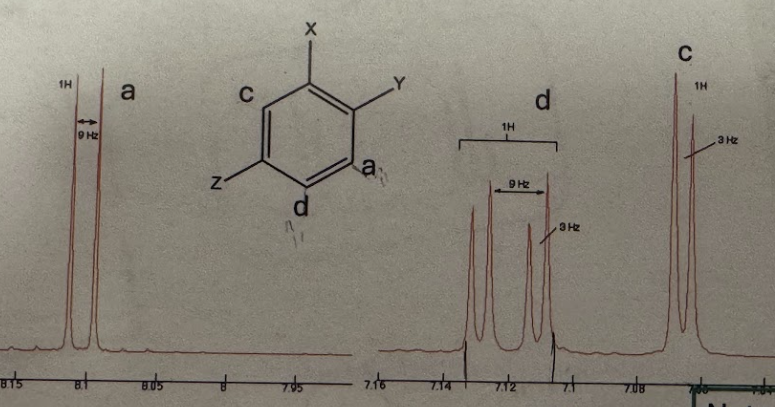
3 other substituents on benzene (H NMR)
If integrating: Carbon NMR Peak height equals what
roughly the number of carbons
IR
Shows functional groups
NMR
Shows molecular formula and arrangement of carbons and hydrogens
Mass spec
Shows molecular formula
IR Primary amine
Double fangs
IR Secondary amine
One fang one broken
M+ (Mass spec)
Molecular weight of compound
Broad signal in IR
H bonding present (concentrated solution)
Narrow signal in IR
Less H bonding present (dilute solution)
Hairy beard in IR + spike at 1700
Carboxylic
Mass spec, # of carbons equation
((M+1)/M)100 = X, X/1.1 = Number of carbons
Nitrogen present in mass spec
If the entire molecular weight is odd, it is possible you have an odd number of Nitrogens, and if the entire molecular weight is even it is possible you have an even number of nitrogens
Cl and Br in mass spec
(M + 2) peak
Cl ratio in mass spec
100:30
Br ratio in mass spec
100:100
MW of Carbon
12
MW of Oxygen
16
MW of Hydrogen
1
MW of Nitrogen
14
MW of Br
79
MW of Cl
35
Base peak is (Mass spec)
100 (the tallest peak) then everything else is relative to that (abundance)
Stability of a fragment (mass spec) is directly related to
The most stable is tertiary carbocation/radical to least stable is CH3+ carbocation/radical
(H NMR) The location of the full signals tell you ____
The types of protons
Downfield
Deshielded (Left)
Upfield
Shielded (Right)
The number of peaks in a signal
Number of neighboring protons (even symmetrical) + 1
Area under the full Signal
Tells you the ratio of the types of protons
Area under the full signal is also called
integration
Deshielding -- Shielding effects of different halogens (NMR)
F Cl Br I CH4
Deshielding -- Shielding effects of amount of Cl (NMR)
CCl4 CHCl3 CH2Cl2 CH3Cl CH4
Proximity to halogen (NMR) Deshielding -- Shielding
Alpha Beta(1/5) Gamma
Chemical shift of O in ester (not carbonyl) (NMR)
+ 3.0 ppm
Chemical shift of OH or OR (NMR)
+ 2.5 ppm
Chemical shift of carbonyl O in ketone, aldehyde, ester, or carboxylic acid (NMR)
+ 1 ppm
Chemical shift of aryl H (NMR)
+ ~6-8 ppm
Chemical shift of methyl H (NMR)
+ ~0.9
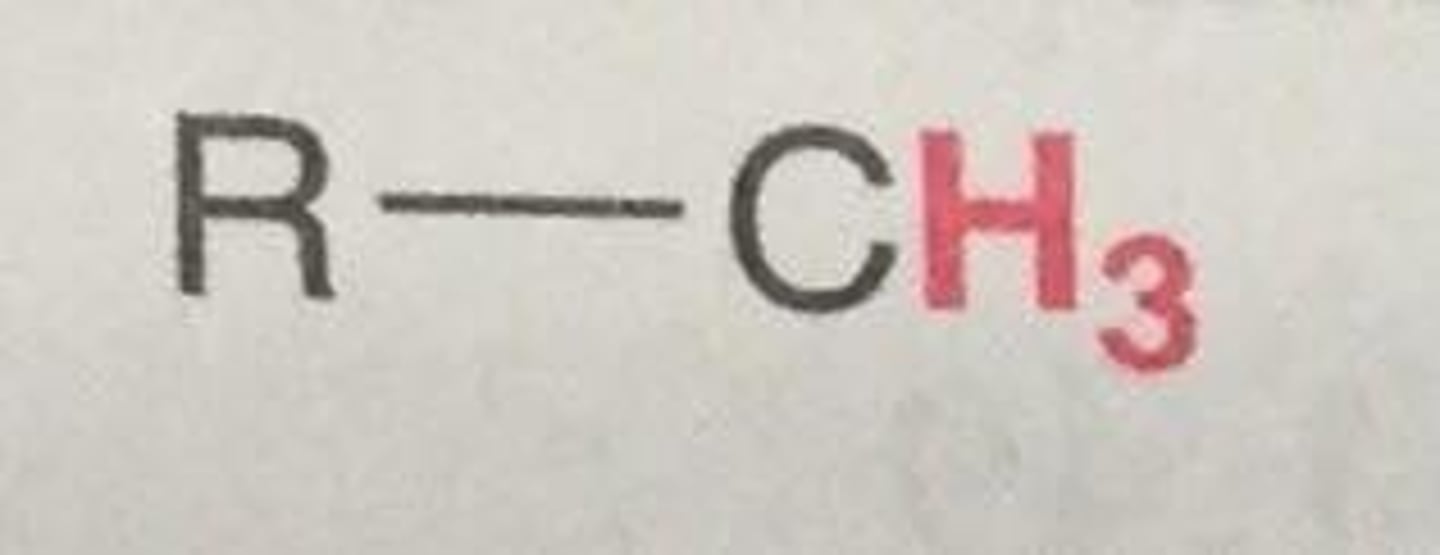
Chemical shift of methylene H (NMR)
+ ~1.2
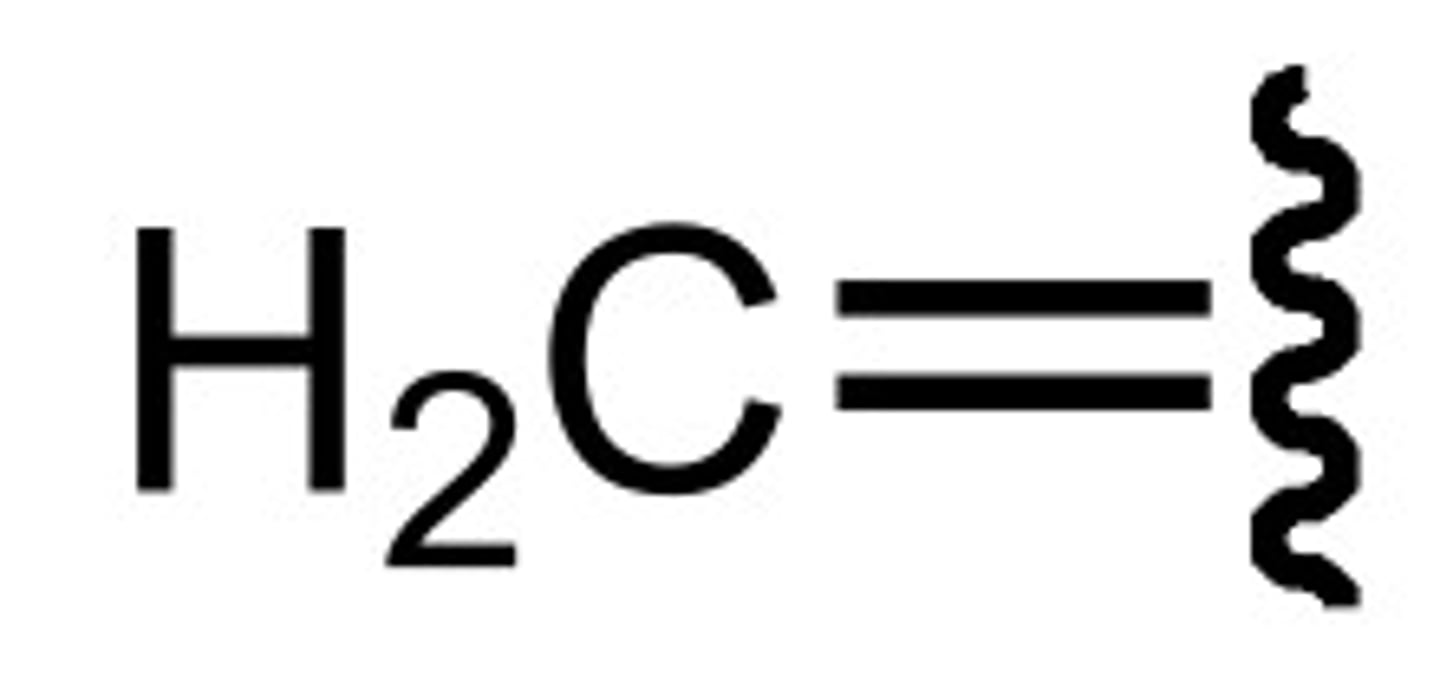
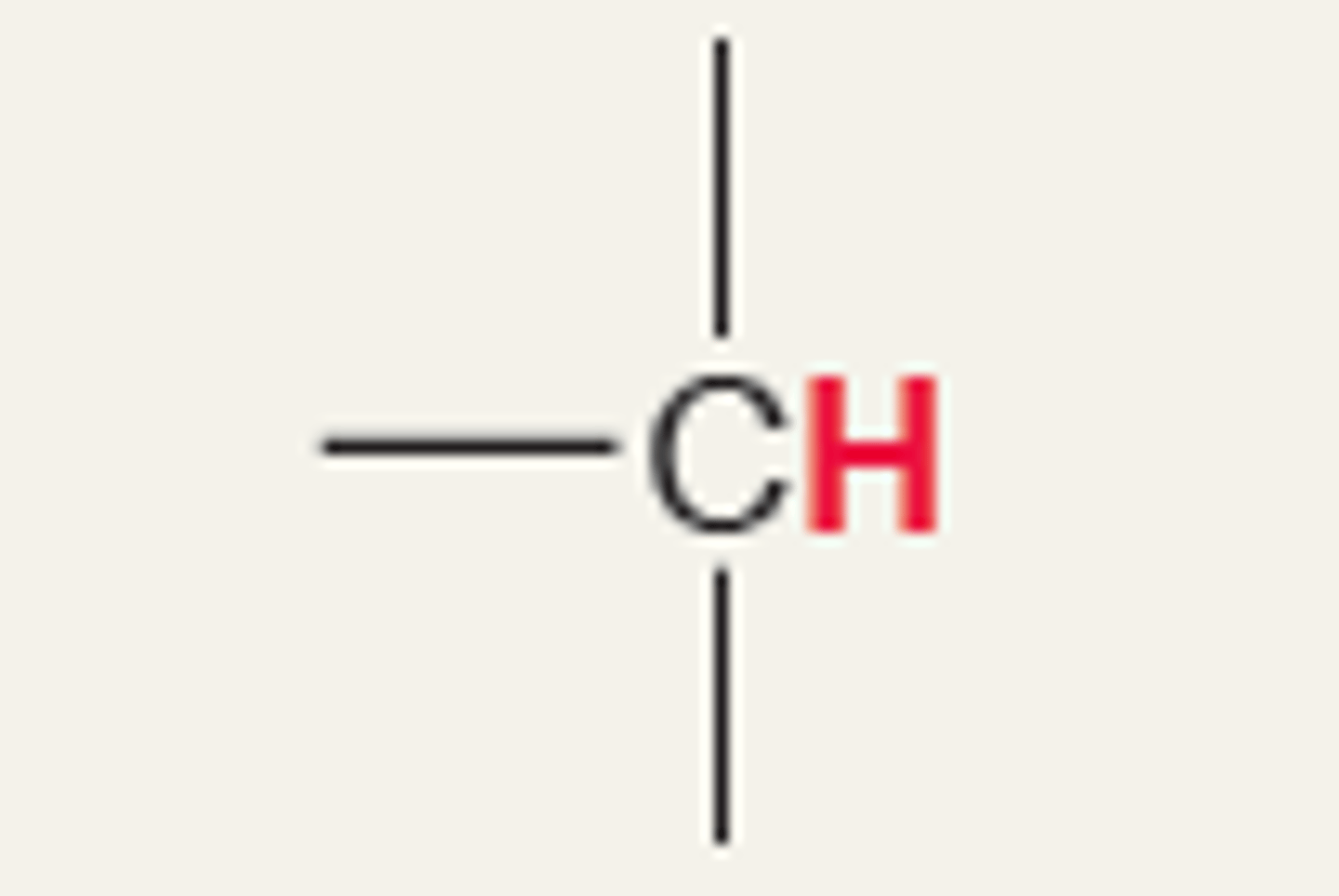
Chemical shift of methine H (NMR)
+ ~1.7
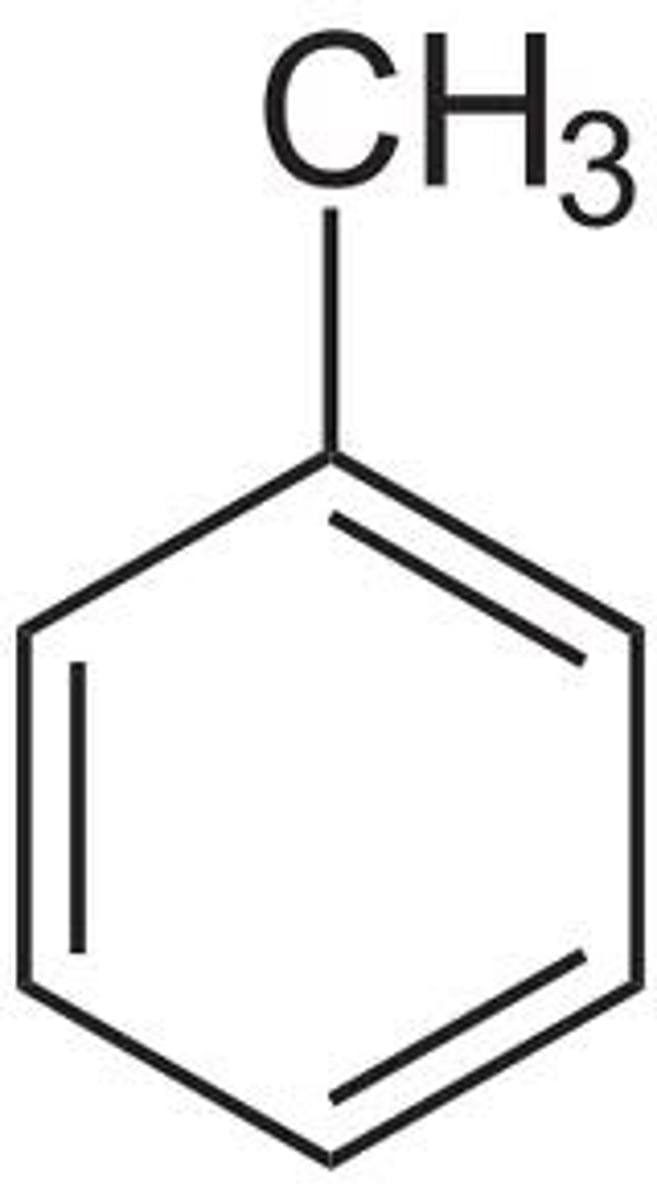
Chemical shift of aromatic methyl H (NMR)
+ ~2.5
To get the ratio of C or H in NMR
Find the smallest line (not C=O or aromatic) and that is 1C (NOT ONE CARBON BUT RATIO 1)
To get the exact number per full signal from ratio
Add all the ratio numbers, then divide each individual ratio by the total, and then multiply by the number of H from the formula (from mass spec)
Mass spec the biggest number peak is
molecular weight (but but there is also M+2 and M+1 so its not technically the biggest number)
1 neighbor to Hydrogen (H NMR)
1 split = Doublet
2 neighbor to Hydrogen (H NMR)
2 split = Triplet
3 neighbor to Hydrogen (H NMR)
3 split = quartet
Doublet of doublet
Two types of neighbors (1 each) in different chemical settings
Doublet of triplet
Two types of neighbors (1 on higher constant, 2 on lower constant) in different chemical settings
__ of a ___ (NMR)
Higher constant of a lower constant
Chemical shift of aldehyde (H NMR)
+ ~10
(Exchanges) in NMR
H connected to Oxygen or Nitrogen
Mass spec: If there's no Cl and no Br....(with a lot of MW left)
NO2 is your next best bet