Lab Techniques- Chromatography
0.0(0)
Studied by 0 peopleCard Sorting
1/23
There's no tags or description
Looks like no tags are added yet.
Last updated 5:27 PM on 9/1/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
1
New cards
What are the basic features present within all types of chomatography?
1. All are use to separate mixtures of compounds
2. Involves a mobile phase and a stationary phase
2
New cards
There are 7 chromatography techniques that are on the MCAT
1. Thin layer (TLC)
2. Column/Flash
3. Ion Exchange
4. High Performance Liquid (HPLC)
5. Size Exclusion
6. Affinity
7. Gas
3
New cards
Analyte
the substance of interest that will be separated by chromatography
4
New cards
Eluent
a solvent that carries the analyte
5
New cards
What is TLC typically used for?
Used for separating compounds based on differing polarities
6
New cards
Why is TLC frequently used in OChem labs?
Fast separation
Used for separating very small amounts of material
Used for separating very small amounts of material
7
New cards
What is the mobile and stationary phase of TLC?
Mobile: liquid (compound)
Stationary: absorbent- typically silica
Stationary: absorbent- typically silica
8
New cards
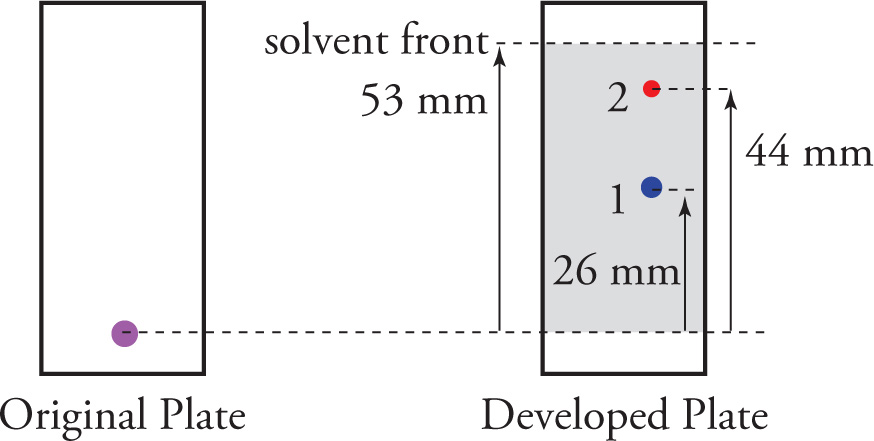
How does TLC work?
\-silica is coated on a glass plate and the plate is placed upright in a sealed container with a shallow layer of solvent
\-sample is spotted near the base of the plate (1cm from the bottom)
\-capillary action draws the solvent up the plate
* more polar molecules travel slower than less polar
\-distance of molecules traveled is measured through Rf values (original spot location/distance traveled by individual component)
\-sample is spotted near the base of the plate (1cm from the bottom)
\-capillary action draws the solvent up the plate
* more polar molecules travel slower than less polar
\-distance of molecules traveled is measured through Rf values (original spot location/distance traveled by individual component)
9
New cards
What is Column/Flash Chromatography used for?
Used to isolate bulk compounds
10
New cards
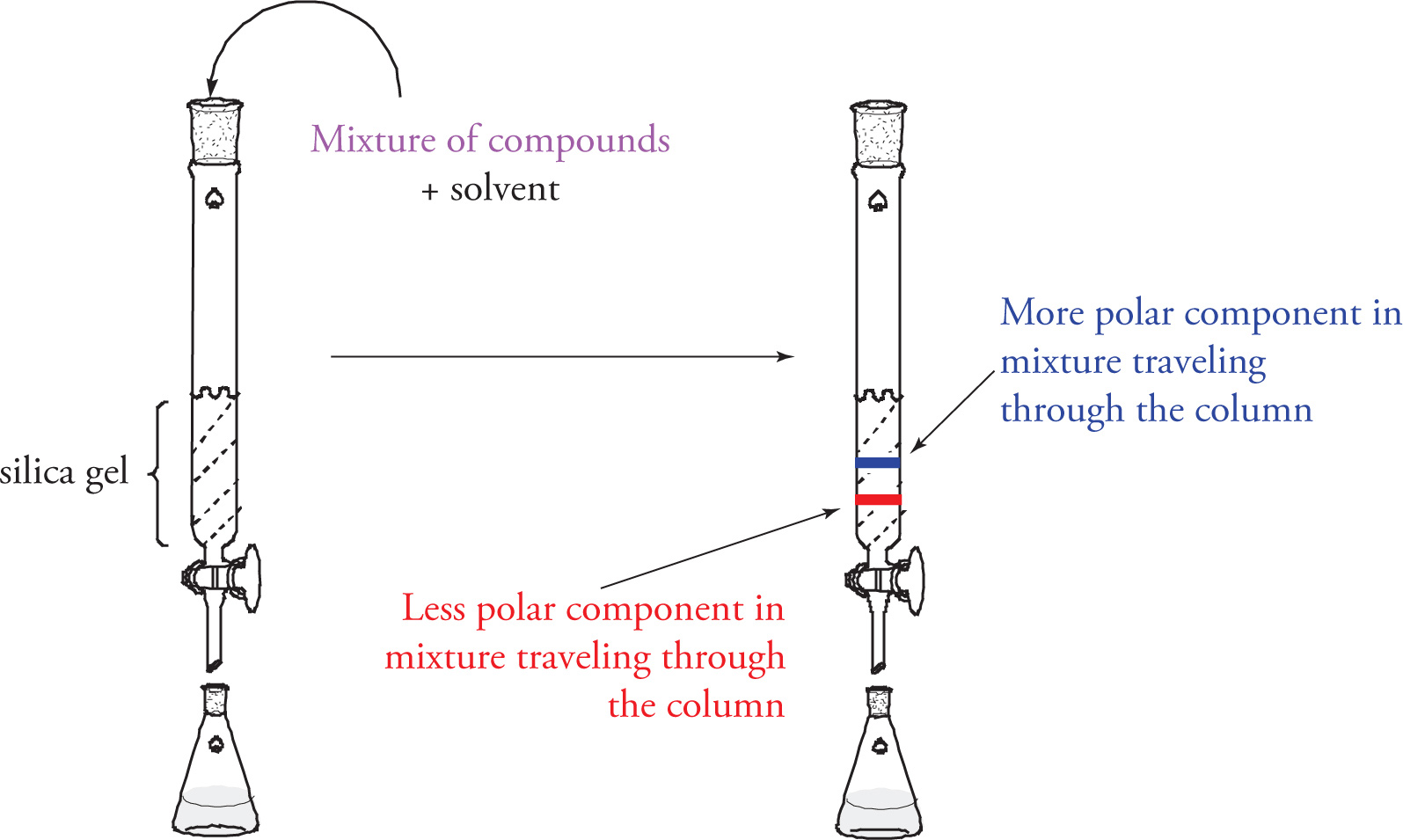
How does Flash/Column work?
A column is filled with a silica gel and is saturated with chosen organic solvents and mixture of compounds at the top of the column
Gravity brings the mixture down, more polar molecules travel slower than less polar
Compounds expected to leave in order of least polar to most polar
Gravity brings the mixture down, more polar molecules travel slower than less polar
Compounds expected to leave in order of least polar to most polar
11
New cards
What are the mobile and stationary phases of Flash?
Stationary: silica beads (polar)
Mobile: organic solvent/compound mixture
Mobile: organic solvent/compound mixture
12
New cards
What is Ion Exchange used for?
separation of materials based on varying charge states, typically in the separation of protein mixtures (cause proteins can either be protonated or deprotonated at specific pH)
13
New cards
2 types of Ion Exchange
Cation Exchange: uses negatively charged groups to attract positive charges
Anion Exchange: uses positively charged groups to attract negative charges
Anion Exchange: uses positively charged groups to attract negative charges
14
New cards
What is the mobile and stationary phase of Ion Exchange?
Stationary: solid polymeric resin with either positive or negatively charged moieties
Mobile: liquid solvent/mixture
Mobile: liquid solvent/mixture
15
New cards
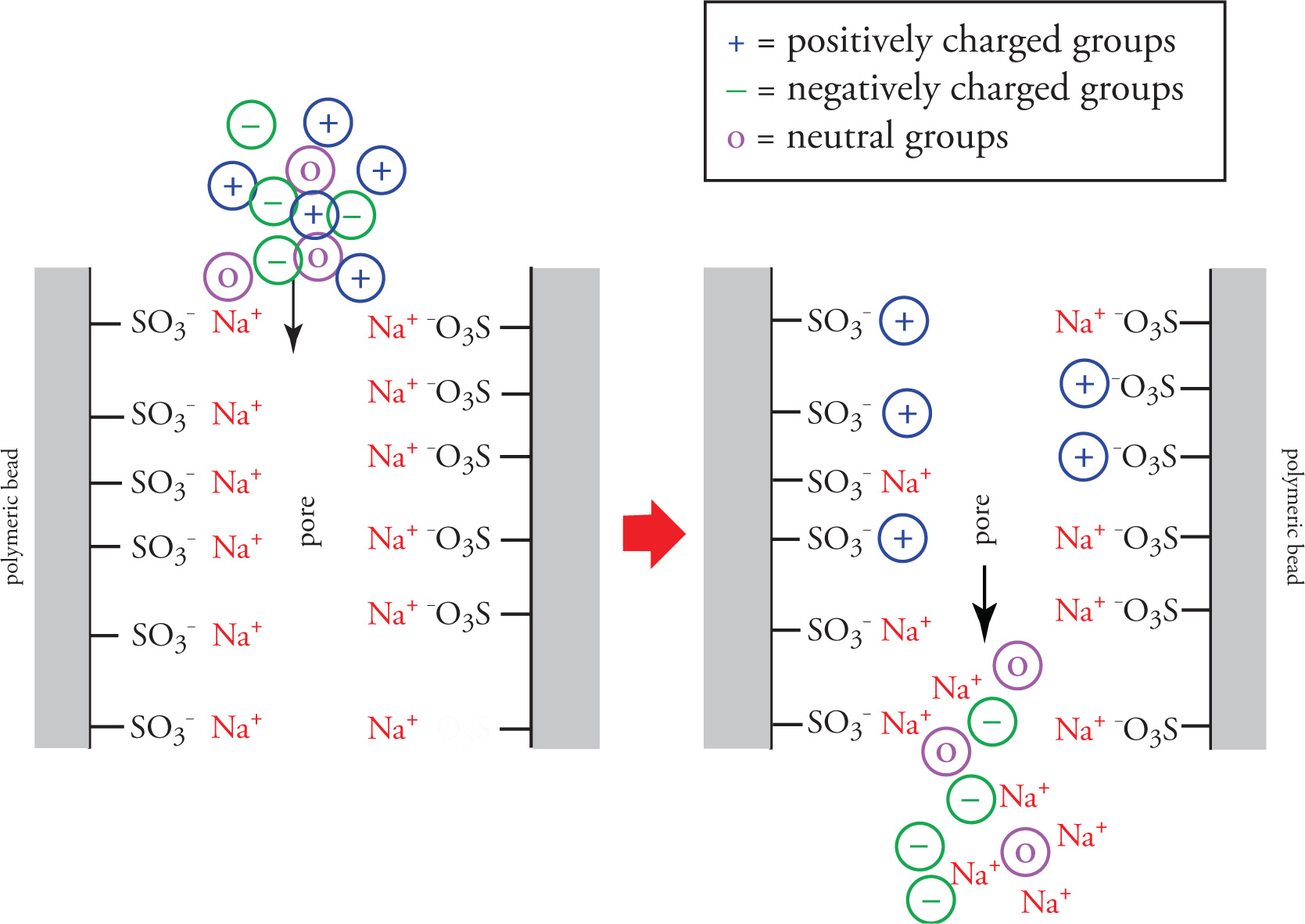
How does Ion Exchange work?
Solvent is fed through stationary phase
Neutral charged groups and groups with the SAME charge as the resin pass through freely
Groups with charges opposite of the moieties in resin move slower
Neutral charged groups and groups with the SAME charge as the resin pass through freely
Groups with charges opposite of the moieties in resin move slower
16
New cards
What is High Performance Liquid Chromatography (HPLC) used for?
Separating bulk compounds
17
New cards
Why is HPLC an improvement over Flash?
HPLC forces mobile phase under high pressures, which increases speed and efficiency of separation
18
New cards
Why is HPLC also called reverse phase HPLC?
HPLC contains a nonpolar stationary phase, which means polar molecules elute first instead of last
19
New cards
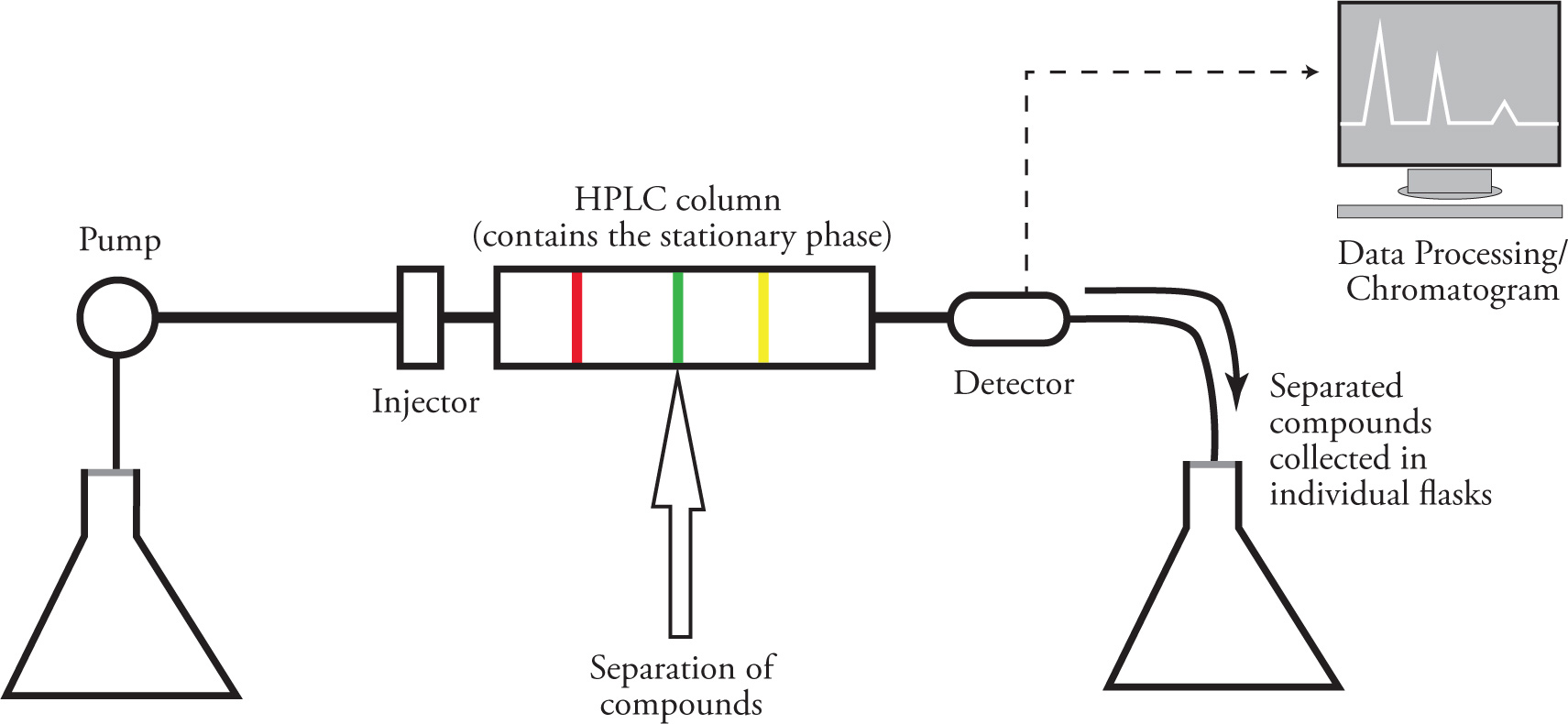
How does HPLC work?
Under high pressures, mobile phase is separated by different affinities of various compounds
Sample is injected and separates within an HPLC column based on affinities and is detected through a data processing program and a detector attached to the apparatus once eluent exits the column
eluent is collected after detected and components can be isolated if desired
more polar molecules elute first compared to lesser polar/non-polar molecules
Sample is injected and separates within an HPLC column based on affinities and is detected through a data processing program and a detector attached to the apparatus once eluent exits the column
eluent is collected after detected and components can be isolated if desired
more polar molecules elute first compared to lesser polar/non-polar molecules
20
New cards
Size Chromatography is used for what?
To separated bulk materials based on molecular size
21
New cards
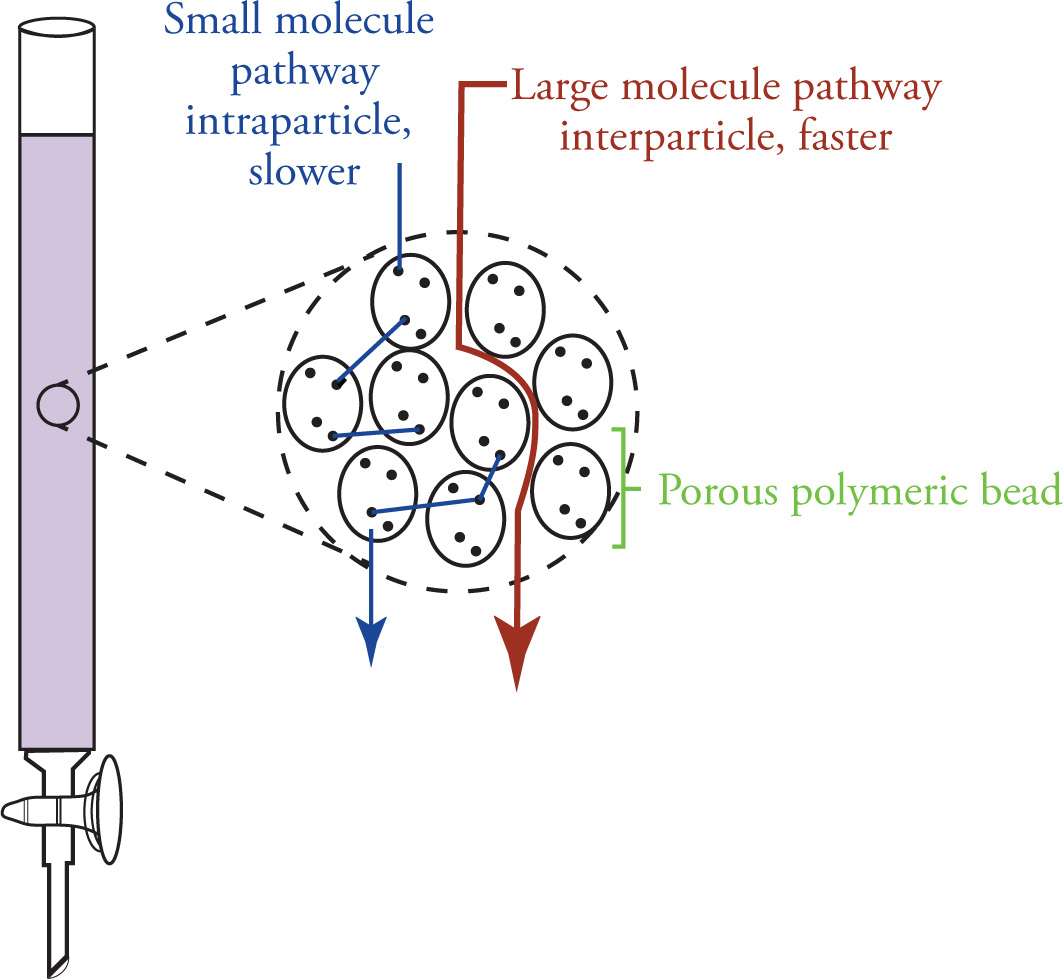
How does Size Exclusion work?
A stationary phase employs chemically inert porous polymer of beads that allows for permeation of small molecules
Beads are packed within a column and solvent is added to the column
Larger molecules elute first because they pass through between beads
Smaller molecules elute last because they travel through the porous beads, thus slowing them down
Beads are packed within a column and solvent is added to the column
Larger molecules elute first because they pass through between beads
Smaller molecules elute last because they travel through the porous beads, thus slowing them down
22
New cards
What is Affinity Chromatography used for?
To purify proteins or nucleic acids from biochemical mixtures (i.e. growth media, blood, cell lysates)
Works on highly specific interactions between macromolecules (antibody, affinity tags, or magnetic bead interactions)
Works on highly specific interactions between macromolecules (antibody, affinity tags, or magnetic bead interactions)
23
New cards
How does Affinity Chromatography work?
With Antibody interactions:
1. Sample is mixed with antibodies that are specific to be against the protein of interest
2. Once antibodies bind to the protein of interest, protein beads are added that are specific to the antibody to create a bead,antibody,protein complex
3. Separation is achieved through centrifuging and the supernatant is removed to isolate the complex
With magnetic beads
1. magnetic beads can bind to target protein
2. magnet is used to collect bead and target protein complex (doesn’t need a centrifuge)
Affinity tags can be used instead of antibodies (affinity tags are created through recombinant technology- popular tag is His a.a tag)
\
\
1. Sample is mixed with antibodies that are specific to be against the protein of interest
2. Once antibodies bind to the protein of interest, protein beads are added that are specific to the antibody to create a bead,antibody,protein complex
3. Separation is achieved through centrifuging and the supernatant is removed to isolate the complex
With magnetic beads
1. magnetic beads can bind to target protein
2. magnet is used to collect bead and target protein complex (doesn’t need a centrifuge)
Affinity tags can be used instead of antibodies (affinity tags are created through recombinant technology- popular tag is His a.a tag)
\
\
24
New cards
What affects GC?
Intramolecular hydrogen bonding and hydrocarbon length