Lab Techniques- Chromatography
5.2 Chromatography
Basic features that are present in all types of chromatography:
All are used to separate mixtures of compounds (whether its for identification or for purification)
involves a mobile phase and a stationary phase
Analyte: the substance of interest that will be separated by chromatography
Eluent: a solvent that carries the analyte
Thin Layer Chromatography (TLC)
Compounds are separated based on differing polarities
Frequently used in OChem labs
Used for separating very small amounts of material
fast separation
TLC is a solid-liquid partitioning technique where the mobile liquid phase ascends a thin layer of absorbent (typically silica) that is coated on a glass plate
absorbent acts as a polar stationary phase
sample (~1 microliter of sample) is spotted near the base of plate (approx. 1cm from the bottom)
plate is then placed upright in a sealed container with a shallow layer of solvent
capillary action draw the solvent up the plate (in a process called developing or running)
Separation of compounds is due to polarity
more polar molecules travel at a slower rate
less polar molecules travel at a faster rate
Visualization of TLC can be through colors or in the process of iodine vapor or other chemical staining techniques
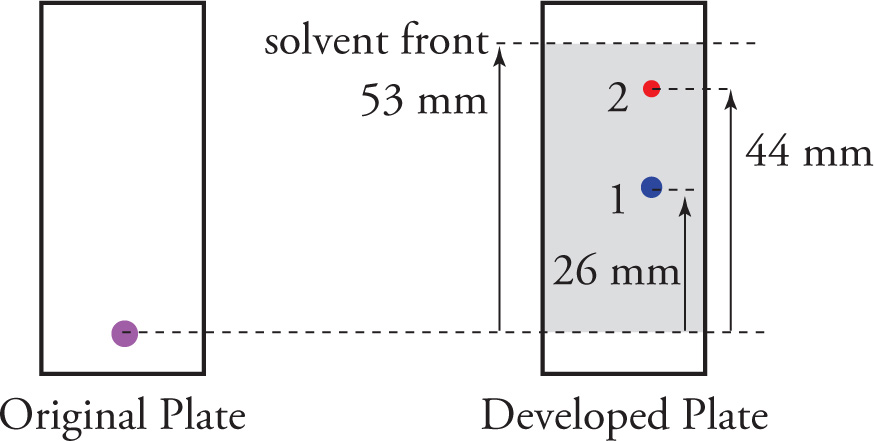
The distance molecules traveled can be measured through Rf (ratio to front) values
Calculated by dividing the distance traveled by an individual component by the distance traveled by the solvent front
Rf is always positive but is never greater than 1
Column (Flash) Chromatography
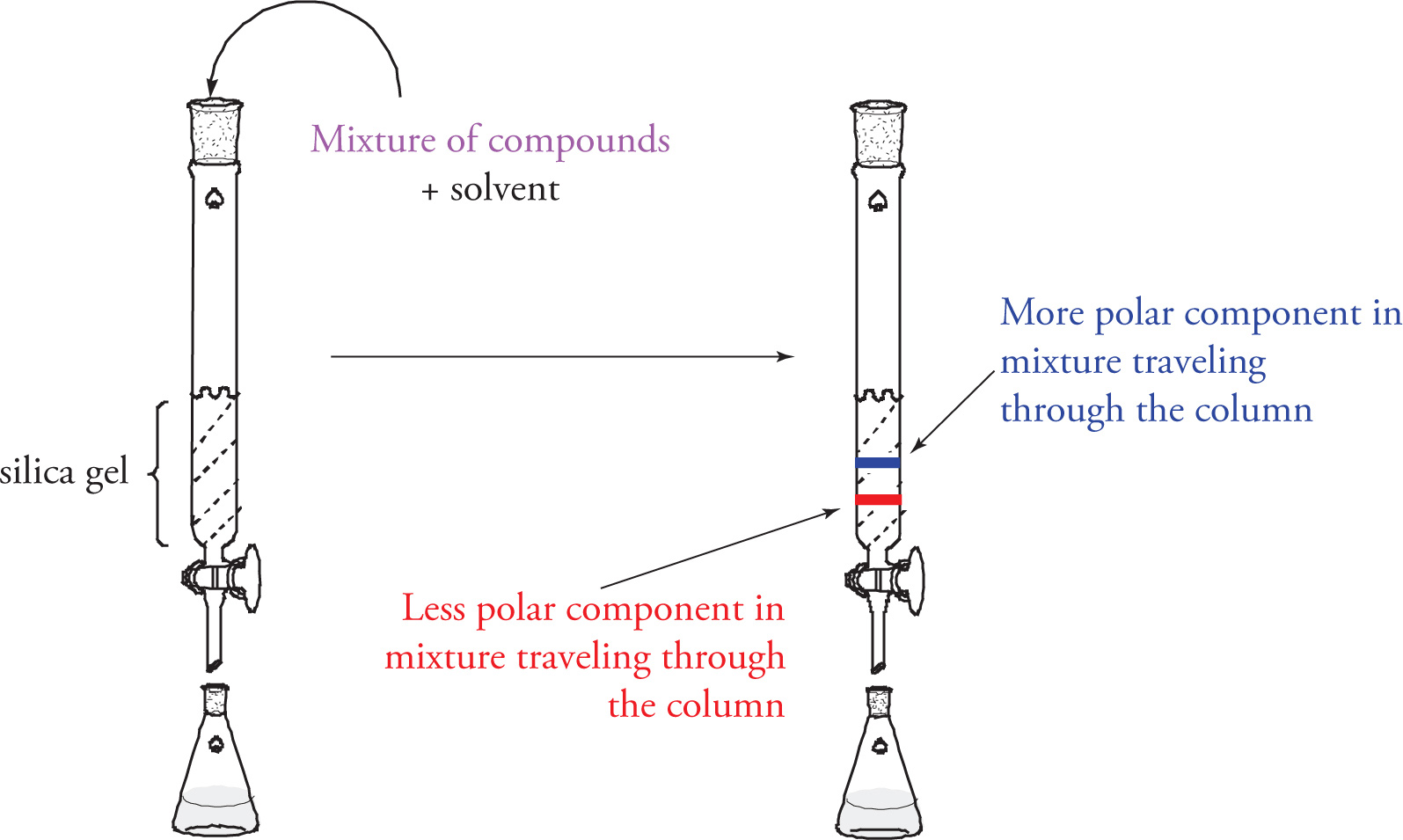
Uses same principles as TLC to isolate bulk compounds
Column is filled with silica gel and is saturated with chosen organic solvents and mixture of compounds at the top of the column
excess solvent is periodically added to the column and the flow of solvent (along with separated compounds is collected at the bottom)
More polar molecules travel slower than less polar molecules
compounds can be expected to leave in order of least polar to most polar
Ion Exchange Chromatography
Used when materials need to be separated based on varying charge states
frequently used in the separation of mixtures of proteins
because at any given pH proteins within a mixture may exist protonated or deprotonated
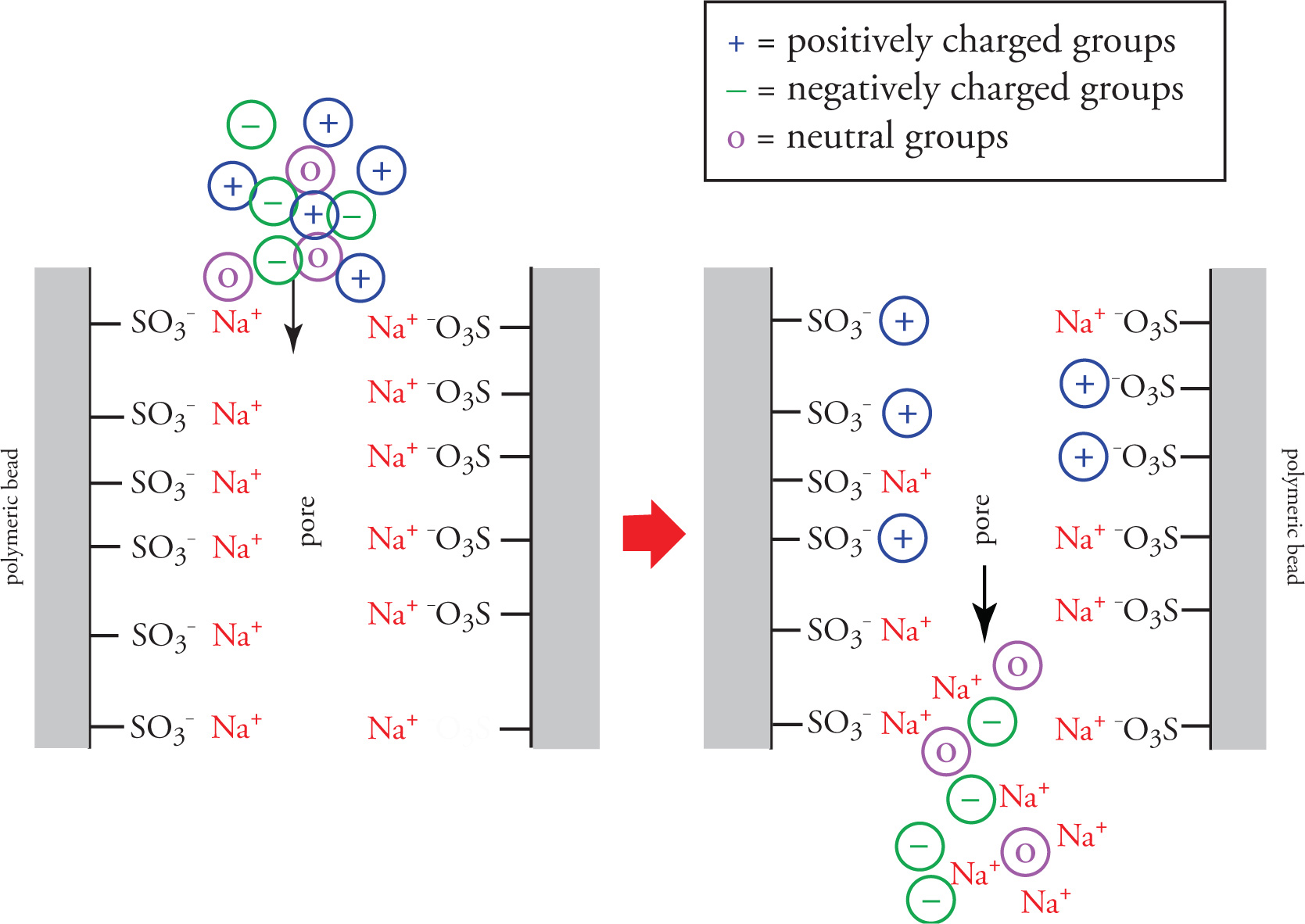
Solid stationary phase uses a polymeric resin functionalized with either positive or negatively charged moieties on a polymer surface
positive charged moieties in resin capture negative charged groups (making them move slower), allowing positive charged and neutral charged groups to pass freely
negatively charged moieties in resin capture positive charged groups (making them move slower), allowing negatively charged and neutral charged groups to pass freely.
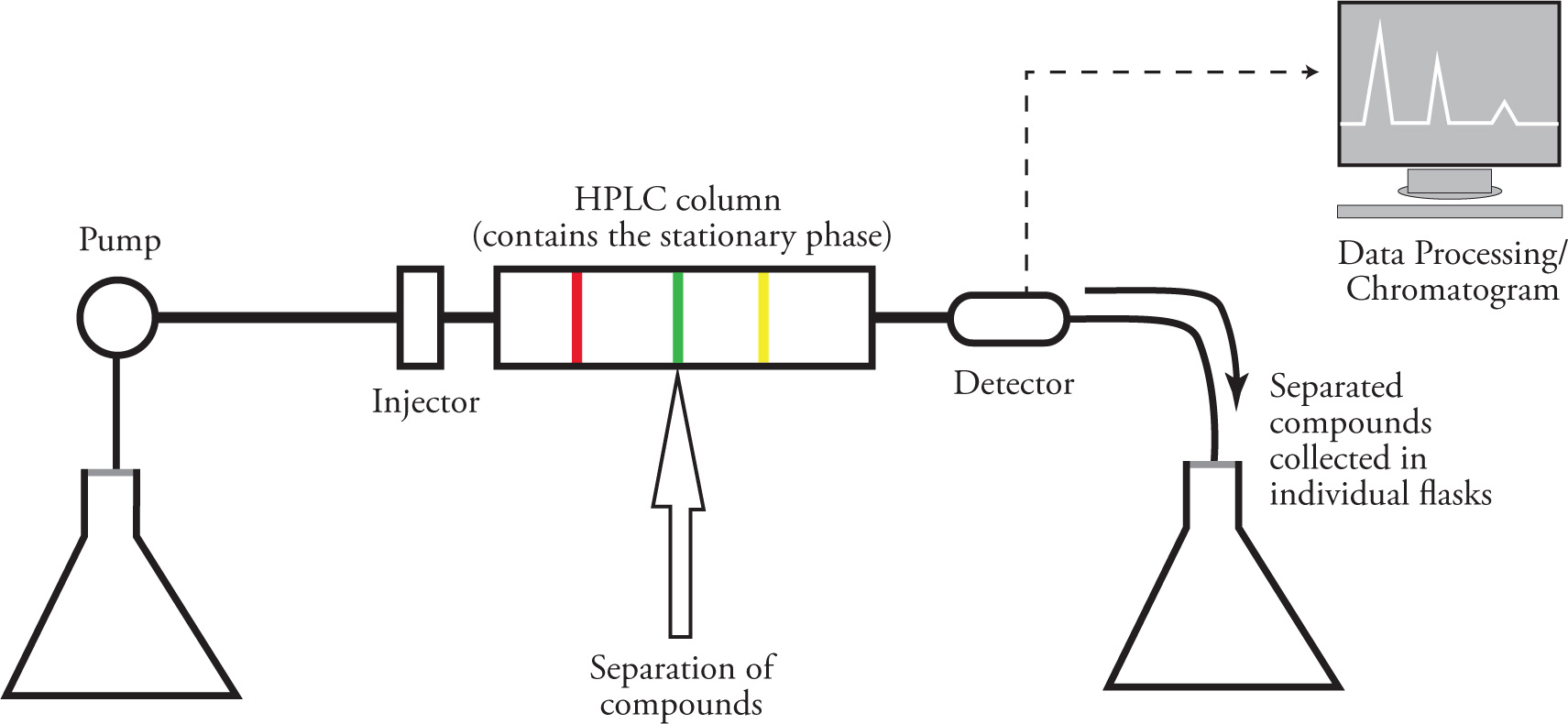
High Performance Liquid Chromatography (HPLC)
Takes advantage of differing affinities of various compounds for a mobile phase or stationary phase
Improvement over Flash Chromatography
because HPLC forces mobile phase under high pressures, which increases speed and efficiency of separation
Pumping unit is where pressurization of the mobile phase first occurs and the sample to be separated is solubilized and injected by a syringe
the mobile phase carries the sample to the column to be separated and detected when they exit the column
eluent is collected after detection and the components can be isolated after evaporation of solvent if desired
Most HPLC contains a nonpolar stationary phase
more polar compounds travel faster in HPLC than nonpolar
why HPLC is also called reverse phase HPLC
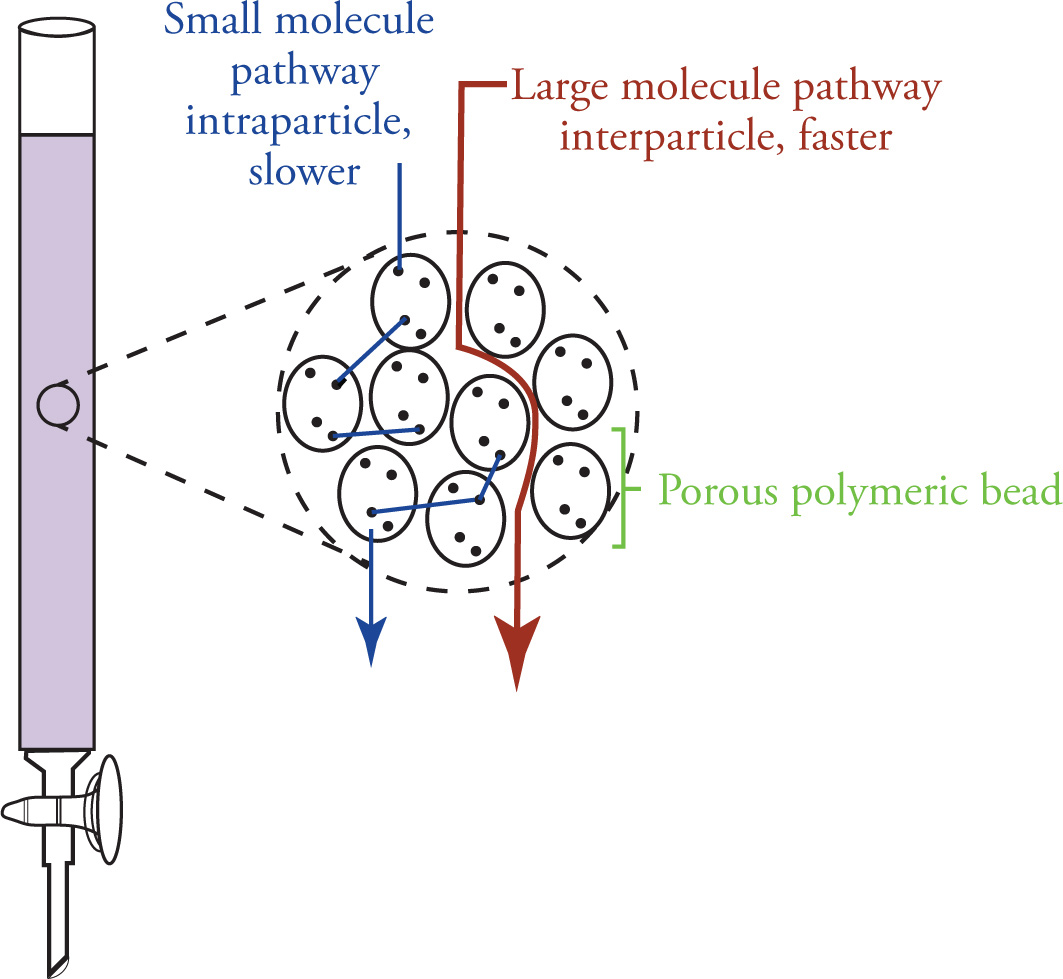
Size Exclusion Chromatography
Used to separate bulk materials based on molecular size
Uses a column where stationary phase is packed in
stationary phase employs chemically inert porous polymer beads and allow for the permeation of small molecules
frequently used for the separation of large polymers from small oligomeric fragments or the separation of full proteins from smaller peptide chains
Relatively speedy elution (compared to silica chromatography)
Not effective at separating different compounds of similar sizes
larger molecules elute first (since they travel interparticle instead of intraparticle)
Affinity Chromatography
commonly used to purify proteins or nucleic acids from complex biochemical mixtures (growth media, blood, cell lysates)
based on highly specific interactions between macromolecules
target molecules is trapped on stationary phase, which is then washed to remove the unwanted components of the mixture
target protein is then eluted off the solid phase in a highly purified state
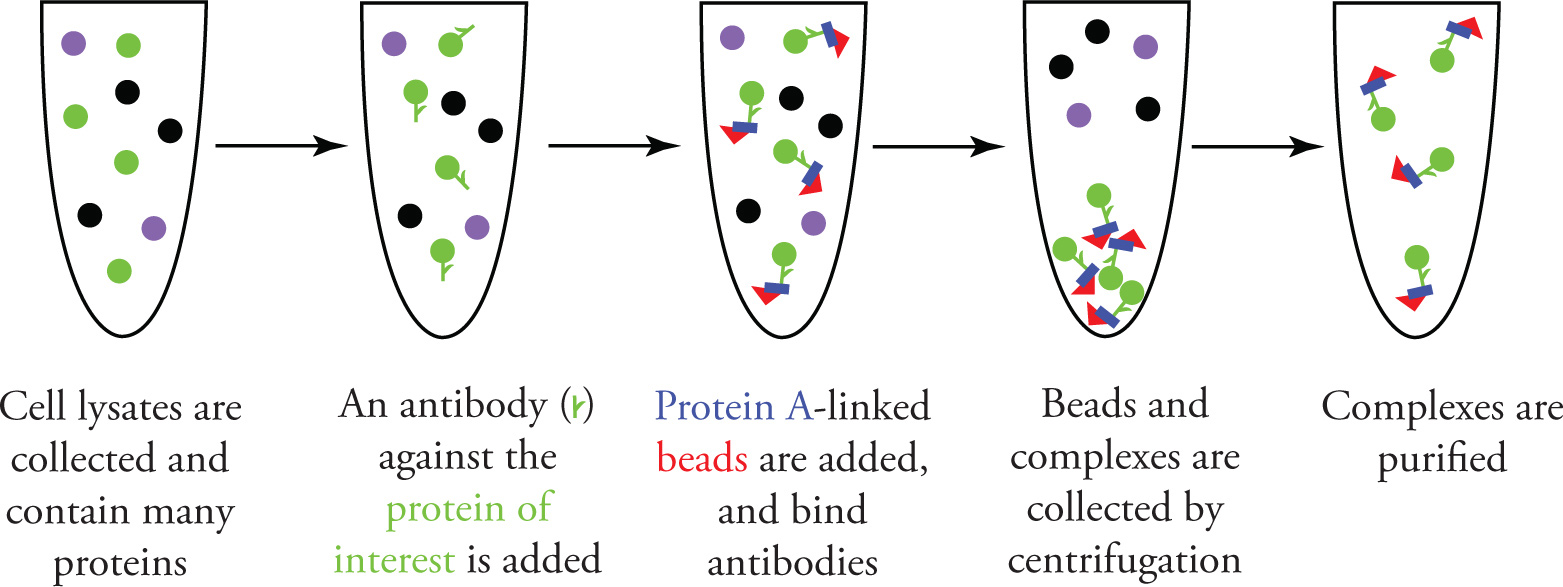
Isolation of protein involves antibody interactions
antibodies are added so that the protein can bind to the antibodies
beads and microbe-derived proteins are then added to bind to the antibodies to create a bead-antibody-microbe protein-and target protein complex
target is then isolated after centrifuging and decanting the supernatant
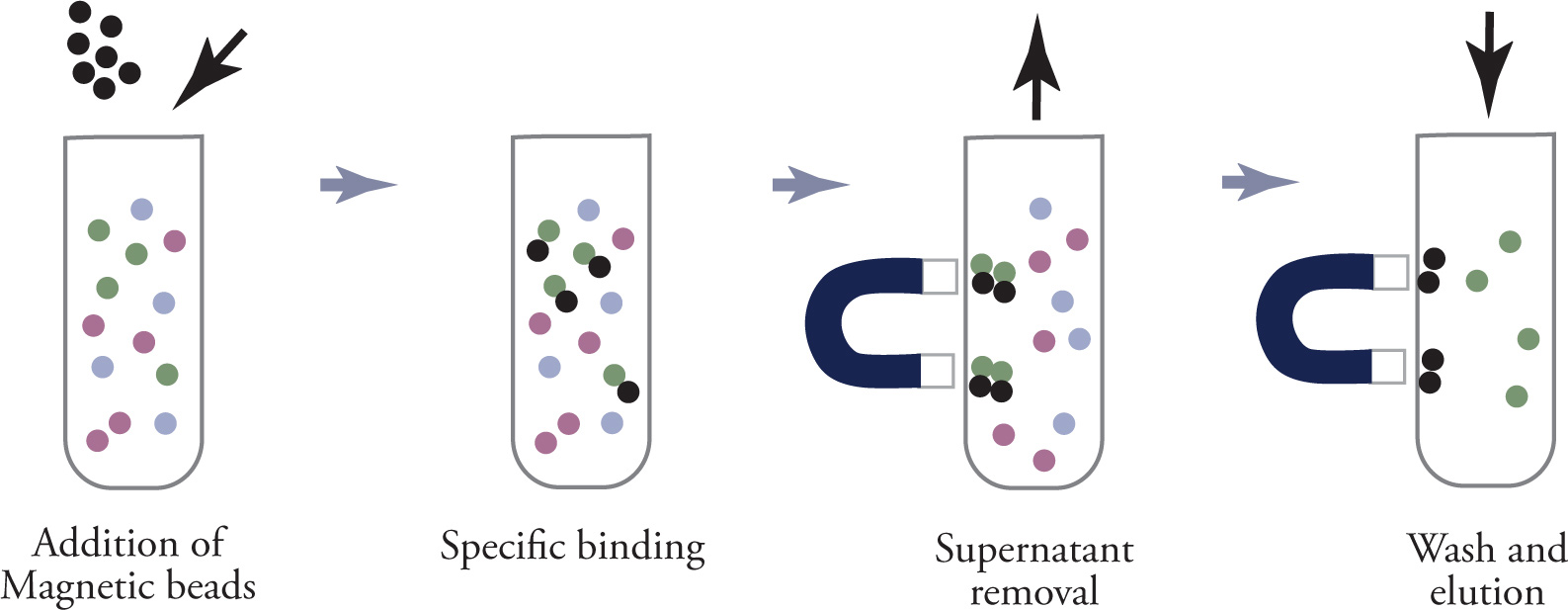
magnetic beads can also be used in place of centrifuging
magnetic beads can bind to target protein
magnet is used to collect bead and target protein complex
Affinity tags can be used (created through recombinant technology) instead of antibodies
popular tag is His tag
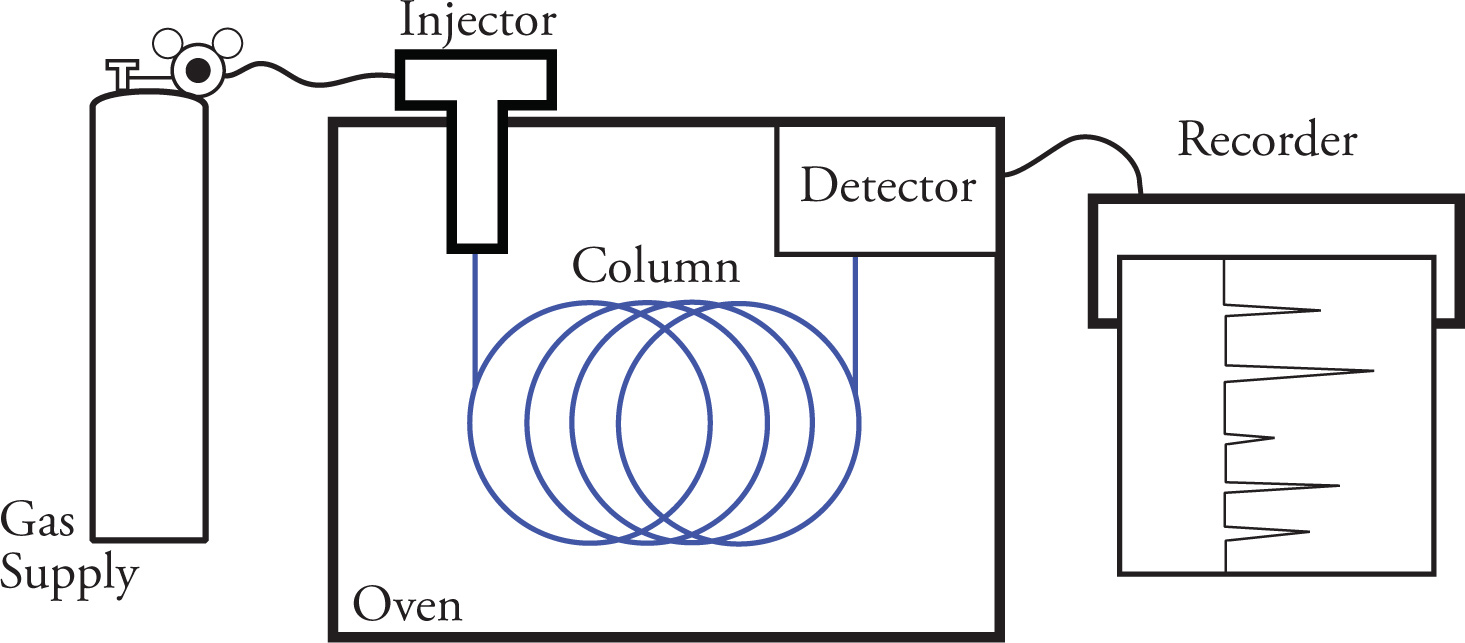
Gas Chromatography (GC)
form of column chromatography in which partitioning of components to be separated takes place between a mobile gas phase and a stationary liquid phase
separation based of various volatilities (boiling points)
Sample is loaded into a syringe and injected into a device through a rubber septum
sample is then vaporized by a heater in the injection port and carried along by a stream of inert gas (typically helium)
sample gas is moved into a column composed of particles that are coated with liquid absorbent
each component of sample interacts differently with the absorbent based of relative volatilities
less volatile components speed more time dissolved in liquid stationary phase
more volatile components spend less time in stationary phase
upon exiting the column, components are burned and results in ions that are detected and recorded to determine number of components and their relative amounts
smaller hydrocarbons have the lowest boiling point
intramolecular hydrogen bonding decreases melting and boiling points