AOS2
0.0(0)
Studied by 3 peopleCard Sorting
1/11
Earn XP
Description and Tags
Last updated 6:29 AM on 4/24/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
1
New cards
How is crude oil formed
Crude oil is formed through the effects of heat and pressure on dead animals, plants and microorganisms trapped in the Earth’s crust, buried beneath sediment over millions of years
2
New cards
Alkane general formula
C(n)H(2n+2)
3
New cards
Alkane structure
\
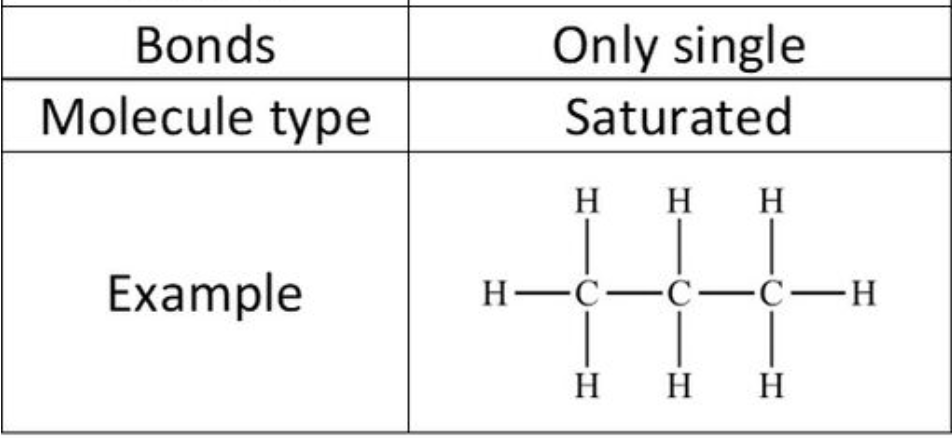
4
New cards
Alkene general formula
C(n)H(2n)
5
New cards
Alkene structure
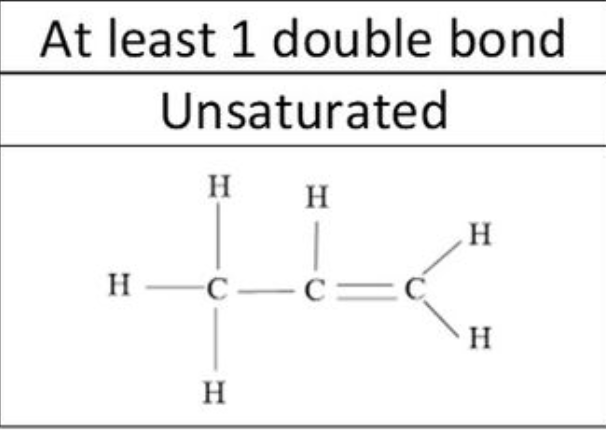
6
New cards
Branched alkanes have…
Have hydrocarbon groups attached to the parent chain
* AKA alkyl groups
* Suffix ‘-yl’
* AKA alkyl groups
* Suffix ‘-yl’
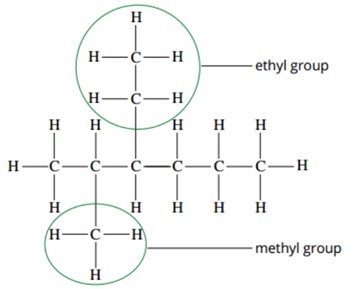
7
New cards
Structural Isomers
Molecules with the **same molecular formula** but a **different structural formula**
* The more atoms in the molecule the more possible isomers there are
* The more atoms in the molecule the more possible isomers there are
8
New cards
Unsaturated hydrocarbons
They do not contain the maximum amount of hydrogen
9
New cards
**Functional groups**
Atom or a group of atoms responsible for the characteristic chemical properties of a particular compound
10
New cards
Haloalkanes
### ==**general formula**==
**2(n)H(2n+1)X**, where X is a halogen atom
\
### ==**halo prefix**es==
chloro-, fluro-, bromo- or ido-
**2(n)H(2n+1)X**, where X is a halogen atom
\
### ==**halo prefix**es==
chloro-, fluro-, bromo- or ido-
11
New cards
Producing haloalkanes from alkanes
* Substitution reaction
* A hydrogen in an alkane is substituted for a halo group
* Halogen molecule is a reactant
* UV light is required for the reaction to occur
* A hydrogen in an alkane is substituted for a halo group
* Halogen molecule is a reactant
* UV light is required for the reaction to occur
12
New cards