Enthalpy changes
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Enthalpy change
The heat energy transferred during a reaction at constant pressure
Exothermic energy profile
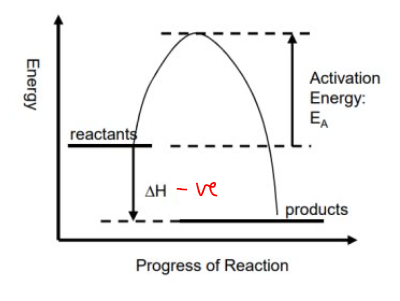
How can you tell an energy profile is exothermic?
The products are at a lower energy than the reactants
Examples of Exothermic Reactions
Combustion
Respiration
Neutralisation always -57kJ
Endothermic Energy Profile
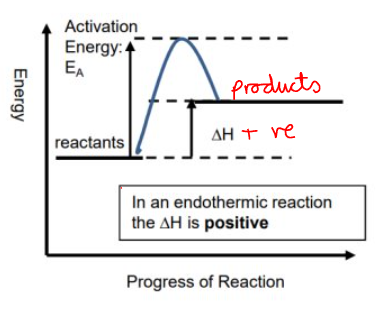
How can you tell an energy profile is endothermic ?
The products are at a higher energy than the reactants
Examples of Endothermic reactions
Thermal decomposition
Photosynthesis
Activation energy
The minimum energy required for a reaction to take place
Standard enthalpy change of a reaction
The enthalpy change for a given reaction in the molar quantities shown in the equation
Can be either exothermic or endothermic
Standard enthalpy change of combustion
The enthalpy change when one mole of a substance completely combusts at 298K and 100KPa
Exothermic reaction
Standard enthalpy change of formation
The enthalpy change when 1 mol of a compound is formed from its elements at 298K and 100KPa
Can be either exothermic or endothermic
Standard enthalpy change of neutralisation
The enthalpy change when 1 mol of H20 is formed from a reaction of H+ and OH- at 298K and 100KPa
H+ + OH- → H20
Always exothermic
Calorimetry
q=mcΔt
q= Energy Change
m= Mass of solution
ΔT= Change in temperature
ΔH = ± q/n
ΔH= Enthalpy Change
q= Energy change in kJ
n= Moles of reactant
Combustion Calorimetry: What acts as the surroundings?
The water
Combustion Calorimetry: What acts as the chemical system?
The fuel burning
Why is the value for ΔH values from experiments different from the data book values
Heat loss to surroundings
Incomplete combustion can occur
How to reduce sources of error in combustion calorimetry
Ensure a ventilated room
Move beaker closer to the flame
Copper beaker instead of glass beaker
Add a lid to the cover
Cover the wick
Calorimetry with a reaction in solution: What acts as the surroundings?
The solution
Calorimetry with a reaction in solution: What acts as the chemical system
The chemicals reacting or dissolving
Sources of error in calorimetry reaction in solution
Heat loss/gain to/from surroundings
Water evaporates from the beaker
Incomplete reaction
Concentration of solution is not the same as water
How to minimise the errors in calorimetry reaction in solution
Use a polystyrene cup
Use a lid to prevent loss/gain
Ensure standard conditions
Predicting temperature changes with different quantities
ΔH stays the same
If n is doubled, q is doubled
Volume is doubled/ Volume is the same
So ΔT stays the same/ ΔT is doubled
ΔH stays the same
If n is halved, q is halved
Volume is halved
ΔT stays the same
Average bond enthalpy
The enthalpy change for the breaking of one mole of bonds in gaseous molecules
How does the bond enthalpy indicate the strength if the bond?
The more positive the bond enthalpy the larger the amount of energy needed to break the bond and so the stronger the bond
Why are bond enthalpies endothermic
Bond breaking requires energy, it is endothermic
Bond making releases energy, it is exothermic
Why is a reaction exothermic?
The energy required to break the bonds is less than the energy released when making bonds
Why is a reaction endothermic?
The energy required to break the bonds is more than the energy released when making bonds
Bond enthalpy calculation
ΔrH= ΣΔBeH (bonds broken) - ΣΔBeH (bonds formed)
Why is it not always possible to measure enthalpy changes of reactions directly from experiments?
A very high activation energy
A very slow rate of reaction
Occurrence of side reactions
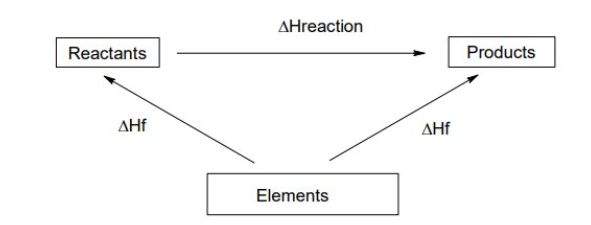
Enthalpy of combustion cycle
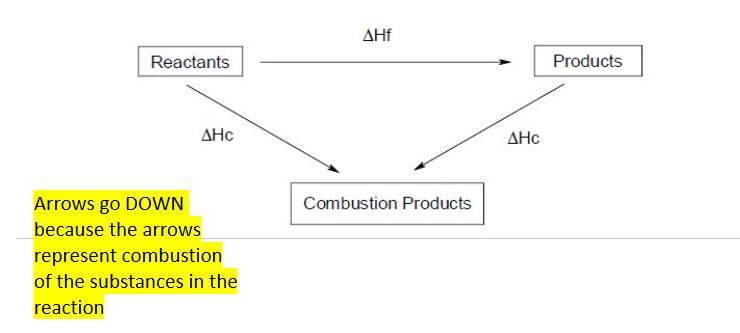
Enthalpy of formation cycle