8 Energetics 1
1/18
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
19 Terms
What is enthalpy change?
The heat change change in a reaction at constant pressure, units kJmol⁻¹, symbol ∆H
What conditions is enthalpy change found in?
Standard conditions with elements in standard states:
100 kPa pressure
298K temp
Is the enthalpy change positive or negative for exothermic reactions?
Negative
Is the enthalpy change positive or negative for endothermic reactions?
Positive
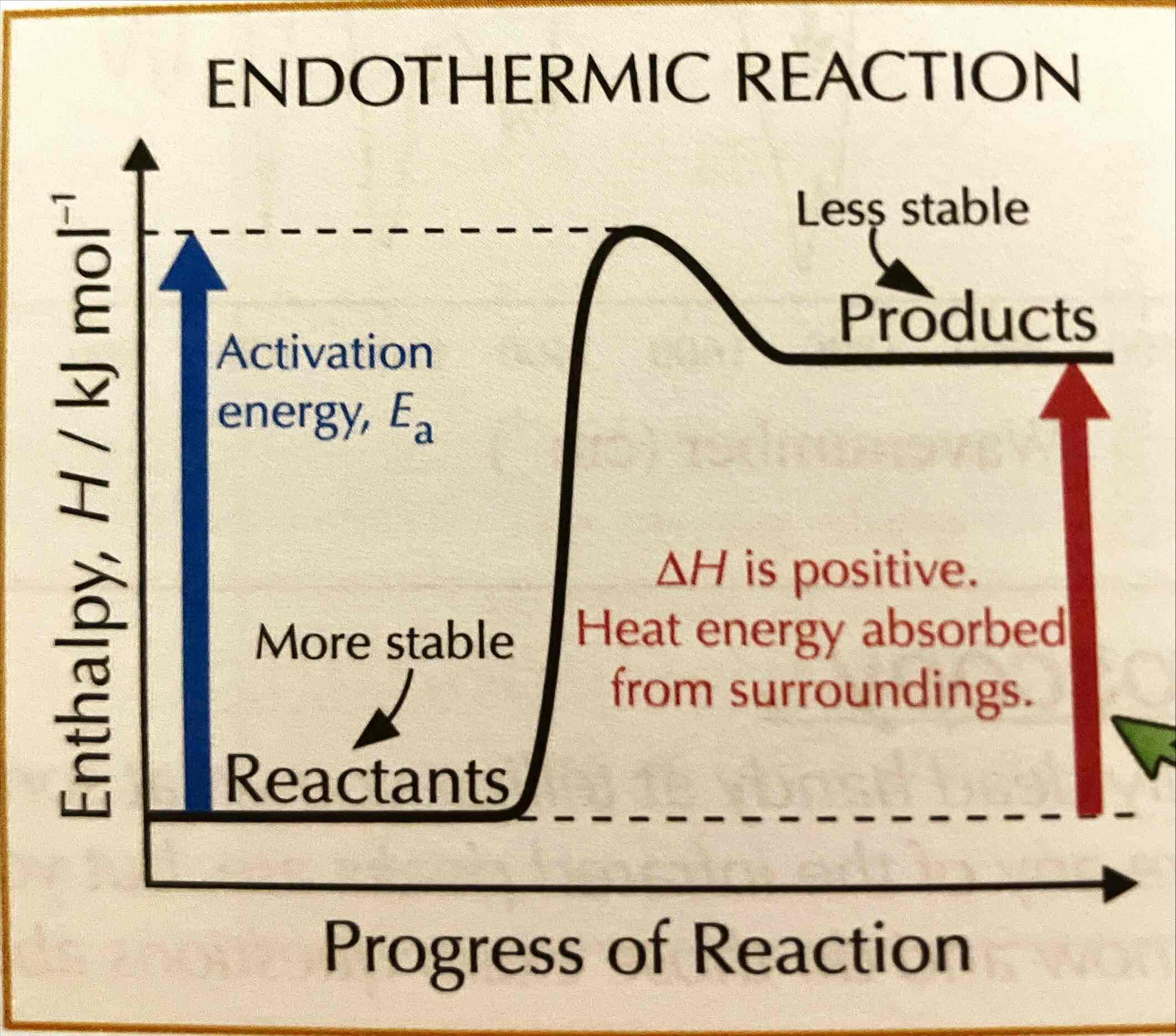
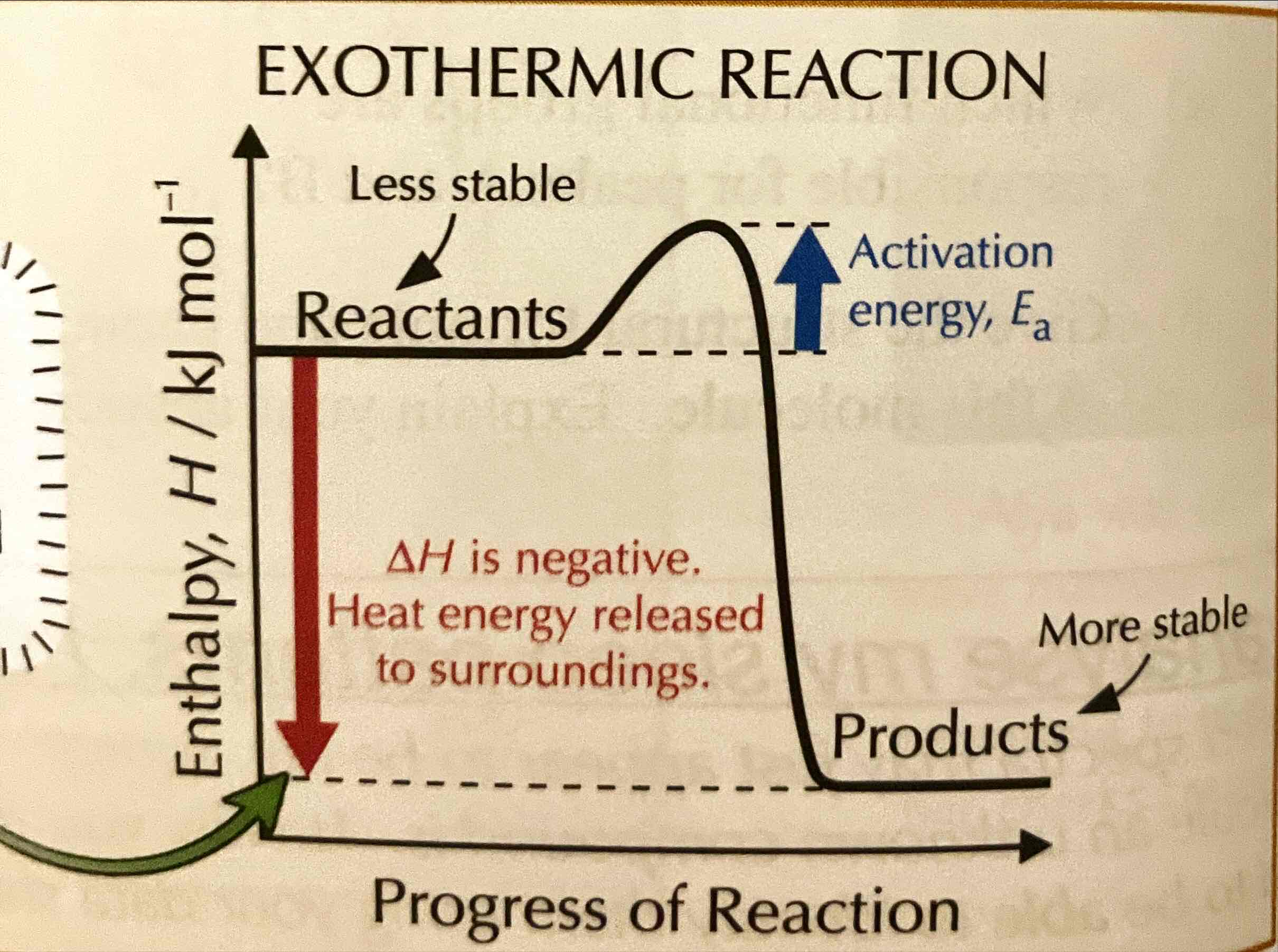
What is the relation between enthalpy and stability?
The less enthalpy a substance has, the more stable it is.
Draw the enthalpy level diagram of an exothermic reaction.

Draw the enthalpy level diagram for an endothermic reaction.

Draw the reaction profile for an endothermic reaction.
Draw the reaction profile for an exothermic reaction.
What is the standard enthalpy change of reaction?
∆rH⁰- the enthalpy change when the reaction occurs in the molar quantities shown in the chemical equation under standard conditions.
What is the standard enthalpy change of formation?
∆fH⁰- the enthalpy change when one mole of a compound is formed from its elements in their standard states, under standard conditions.
What is the standard enthalpy change of combustion?
∆cH⁰- the enthalpy change when one mole of a substance is completely burnt in oxygen, under standard conditions.
What is the standard enthalpy change of neutralisation?
∆neutH⁰- the enthalpy change when an acid and an alkali react together, under standard conditions, to form one mole of water.
What do you need to calculate the enthalpy change of combustion?
A calorimeter.
What are the variables in q=mc∆T?
q= energy lost or gained in joules.
m= mass of water in the calorimeter or solution in insulated container in grams.
c= specific heat capacity of water
T= change in temperature of the water or solution in K
In a lab experiment, 1.16 g of an organic liquid fluid was completely burnt in oxygen. The heat formed during this combustion raised the temperature of 100g of water from 17.5C to 80C. Calculate the standard enthalpy of combustion. The Mr is 58.0.
Calculate amount of heat given out: q=mc∆T.
= 100 × 4.8 x (80-17.5) = 26125J = 26.125 kJ
Find out how many moles were used to produce this heat: n = mass/Mr.
1.16/58= 0.02 mol of fuel
Find the heat produced by one mole of fuel:
26.125/ 0.02 = 1310kJmol⁻¹
Make it negative as combustion is exothermic.
The standard enthalpy change of combustion is -1310kJmol⁻¹
What does Hess’s Law state?
The total enthalpy change of a reaction is always the same, no matter which route is taken. Eg.:

How can you use the graph of a thermometric titration to find the max temperature rise? …
Draw two lines of best fit for when the temperature is increasing and decreasing, both of which do not include the highest value. The max temperature rise is where they meet, which should be above the highest value.
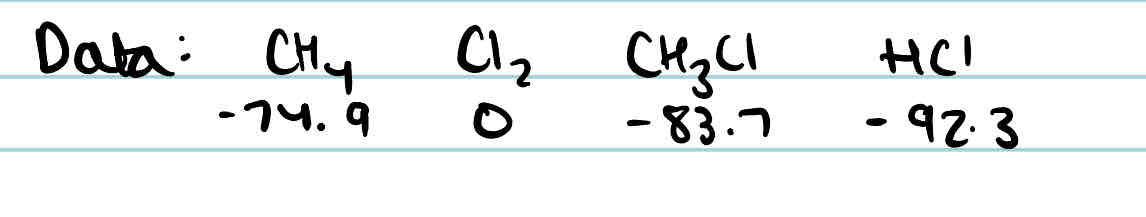
Given this data, find the standard enthalpy change of this reaction using standard enthalpy change of formation:
Cl₂ + CH₄ → CH₃Cl + HCl