IBDP Biology
1/162
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
163 Terms
Discuss possible exceptions to cell theory (4)
- skeletal muscle fibres have many nuclei
---cells always function as autonomous units
- aseptate fungal hyphae have a continuous cytoplasm
---living structures are composed of discrete cells
- giant algae (unicellular) grow to very large sizes
---larger organisms are always made up of many microscopic cells
State the functions of one named extracellular component (4)
plant cell wall:
- strengthens/supports cell
- maintains shape of cells
- prevents pathogen entry
Explain the importance of surface area:volume ratio (6)
- large surface area:vol needed
- as cells increase in size ratio is reduced (volume increases faster than surface area)
- surface area needs to be large enough to absorb nutrients/oxygen/substances needed
- needs to be large enough to excrete/pass out waste products
- optimum diffusion is large surface area and small volume
- cells divide when they reach a certain size
Outline differentiation of cells in a multicellular organism (6)
- differentiation is development in different/specific ways
- cells carry out specialised functions/become specialised
- some genes are expressed/switched on but not others
- regulated mainly during transcription
- chemicals determine how a cell develops
- becomes more efficient
- group of differentiated cells = tissue
Describe the importance of stem cells in differentiation (4)
- stem cells are undifferentiated cells
- stem cells retain capacity to divide and differentiate along different pathways into specialised cells
- differentiation involves expressing some genes but not others
- stem cells can be used to repair/replace tissue/heal wounds
Describe stem cells and how they are used in medicine (stargardt's) (5)
- stem cells are undifferentiated and retain capacity to divide
Stargardt's:
- genetic degeneration of photoreceptor cells in retina
- human embryonic stem cells treated to differentiate into retinal cells
- retinal cells injected into area
- they attach and become functional - improves vision
Describe stem cells and how they are used in medicine (leukemia) (6)
- stem cells are undifferentiated and retain capacity to divide
Leukemia:
- cancer of blood/bone marrow
- hematopoeic stem cells harvested from the bone marrow
- chemotherapy/radiotherapy used to destroy diseased WBCs
- HSCs transplanted back into bone marrow
- differentiate to become WBCS and replace diseased ones
Draw a labelled diagram to show the ultrastructure of E.coli (6)
- cell wall
- plasma membrane
- cytoplasm
- flagellum (embedded in cell wall)
- 70S ribosomes
- nucleoid/naked DNA
Compare adult and embryonic stem cells (5)
embryonic:
/ almost unlimited in growth potential
/ less chance of genetic damage
X more risk of becoming tumor
X chance of rejection (diff from recipient)
X ethics - extraction kills embryo
adult:
/ less chance of developing tumors
/ extraction does not kill adult
/ no rejection issues
X difficult to obtain (few + deep)
X less growth potential
X limited capacity to differentiate
Function of cell wall
Protect against mechanical and hypertonic stress
Function of plasma membrane
phospholipid layer controls movement of materials in/out
Function of nucleoid region
storage of genetic info (genetic info) and DNA replication
Function of ribosomes
site of protein synthesis
Function of flagella
movement and sexual conjugation
Function of cytoplasm
where metabolic reactions occur
How do prokaryotic cells divide (4)
Binary Fission:
- single circular chromosome replicated
- 2 copies move to opposite ends
- division of cytoplasm = genetically identical daughter cells
- CYTOKINESIS
Draw a labelled diagram of a eukaryotic cell (6)
- golgi apparatus
- free 80S ribosomes
- mitochondrion
- lysosomes
- centrioles
- rough ER
- vesicles
- nucleus
- vacuole
- plasma membrane
Function of golgi apparatus
sorting and transporting proteins
Describe the process of tRNA activation (4)
- specific tRNA activating enzyme recognises specific amino acid
- enzyme binds ATP to amino acid
- specific tRNA molecule recruited
- tRNA binds to amino acid (AMP released)
- ATP phosphorylation creates high energy bond - transferred to RNA
- provides energy for peptide bond to form during translation
Function of lysosomes
digestion/metabolism of materials ingested by endocytosis using digestive enzymes
Function of centrioles
move chromosomes during cell division
Function of rough ER
protein synthesis and transport
Function of nucleus
controls cell activities/DNA replication/DNA transcription/mitosis
Function of vacuole
Digestion, storage, waste disposal, water balance
Function of vesicle
Small vacuoles used to transport materials inside cell
Explain the advantages of compartmentalization (5)
= membrane bound organelles
1. Efficiency of metabolism (enzymes/substrates more concentrated)
2. localised conditions (e.g. pH maintained at ideal)
3. toxic substances isolated (e.g. digestive enzymes in lysosome)
4. numbers/locations of organelles can be changed
Distinguish between the structure of plant and animal cells (6)
plant cells:
- have cells walls
- have chloroplasts
- large central vacuole
- store starch
animal cells:
- have centrioles
- have cholesterol in membrane
Compare prokaryotic cells and eukaryotic cells (5)
Size: p = small, e = large
compartmentalized: p = no, e = yes
ribosomes: p = 70s, e = 80s
dna structures: p = naked, no histones e = bound to histones
site of dna: p = nucleoid, e = nucleus
mitochondria: p = no, e = yes
Draw a labelled diagram to show the structure of the plasma membrane (5)
- phospholipid bilayer
- phospholipid (hydrophobic fatty acid tail, hydrophilic phosphate head)
- integral protein
- glycoprotein
- channel protein
- peripheral
- cholesterol
Explain how the structure and properties of phospholipids help to maintain the structure of cell membranes (9)
- hydrophobic tail/hydrophilic head
- form bilayer (hydrophilic exterior hydrophobic interior)
- held together by hydrophobic interactions
- layers stabilised by interactions of hydrophilic heads and surrounding water
- phospholipids can move about to increase fluidity
- allow for membrane fluidity
- fluidity helps be functionally stable
- fluidity important in making/breaking membranes (endo/exocytosis)
- layers restrict entry/exit of substances
Explain the role of cholesterol in the plasma membrane (4)
- component of plasma cell membranes
- positioned between phospholipids in the membrane
- reduces membrane fluidity and permeability to some solutes
- disrupts regular packing of hydrocarbon tails
- prevents crystallisation and solid behavior
- help membranes curve into concave shape (formation of vesicles)
Explain the role of vesicles in the transportation of materials within cells (8)
- vesicles are membrane bound packages
- formed by pinching off a piece from membrane (endocytosis)
- can carry proteins
- RER synthesizes proteins - proteins accumulate in ER - vesicles bud off RER
- vesicles transport to Golgi apparatus
- fuse to GA, process protein into final form
- bud off GA move to plasma membrane
- protein secreted by exocytosis
Describe the process of active transport (4)
- uses/requires ATP
- against concentration gradient
- uses protein
- protein goes through conformational change
- ATP is hydrolysed -> ADP
Outline the ways in which a substance can move passively across a membrane (5)
- diffusion is a method of passive transport
- channel proteins in facilitated diffusion allow hydrophilic molecules to cross
- movement from high to low conc gradient
- e.g. oxygen
- osmosis is diffusion of water through a membrane
- region of higher water potential to lower
Describe simple diffusion (2)
passive net movement of particles from area of high concentration to low concentration often through a partially permeable membrane
- non polar small particles (e.g. oxygen) diffuse easily
Describe facilitated diffusion (2)
Movement of large and polar molecules that can't get across the membrane via simple diffusion through transmembrane proteins
Describe osmosis (2)
Passive net movement of water molecules from low solute conc. to area of high solute conc. through partially permeable membrane
(sometimes through protein aquaporin)
Distinguish between passive and active movements of materials across plasma membranes, using named examples (4)
passive: diffusion, osmosis, f diffusion
active: ion pumps, exocytosis, endocytosis, active transport
a = requires energy, p = does not
a = against conc g, p = down conc g
a = protein required, p = no protein required
a = e.g. root cell ion uptake, p = oxygen alveoli
Outline, with example, the process of exocytosis (5)
- vesicles carry materials to the plasma membrane
- vesicle fuses with plasma membrane
- by joining of phospholipid bilayers
- aided by fluidity of the membrane
- material released/expelled from cell
- membrane flattens
e.g. hormone secretion
- hormones released have an effect on another cell
Explain the reasons for cell division in living organisms (8)
- increase number of cells in an organism
- allow differentiation/cell specialization
- for greater efficiency
- and replace damaged/lost cells
e.g. binary fission
- asexual reproduction of unicellular organisms
- gamete/spore formation
- cells cannot grow beyond certain size
- sa:vol ratio becomes too small
- transport across membrane is too slow
Outline the processes that occur in a cell during interphase, including those needed for mitosis (4)
Interphase: active period in cell cycle when many metabolic reactions occur in nucleus and cytoplasm
- dna replication
- organelle duplication
- cell growth
- transcription/translation
- obtain nutrients
- respiration
Outline the 3 stages in interphase (3)
G1 - cell grows and preps for DNA replication
S - synthesis stage - DNA replicated
G2 - cell finishes growing and preps for cell division
Describe the cell cycle (8)
1. Prophase
- DNA supercoils and chromatin condense
- centrosomes move to opposite cell poles and form microtubule spindle fibers
- nuclear membrane breaks down
2. Metaphase
- microtubule spindle fibers connect to centromeres of each chromosome
- spindle fibres contract and chromosomes align along center of cell
3. Anaphase
- contraction causes sister chromatids to separate
- move to opposite poles of cells
4. Telophase
- spindle fibers dissolve
- chromosomes uncoil, decondense
- nuclear membranes reform
Describe process of cytokinesis (4)
process of cytoplasmic division
- mt filaments form ring around cell center
- constrict and form cleavage furrow
- furrow meets center and is pinched off
- 2 cells formed
Explain the role of cyclins (5)
- regulatory proteins that control progression of cell cycle
- bind to enzymes called cyclin-dependent kinases
- activate them and CDK attaches phosphate groups to other proteins in the cell
- triggers other proteins to become active and carry out tasks in cell cycle
- 4 main cyclins -> have to reach certain concentration before progressing to next stage
Outline the evidence for endosymbiotic theory (4)
- organelles originated as symbioses between separate single-celled organisms
- contain own circular DNA (like prokaryotes)
- 70S ribosomes
- only produced by division of pre-existing mitochondria/chloroplasts
- transcribe DNA and use mRNA to synthesize own proteins
Outline the thermal, cohesive and solvent properties of water (5)
- high specific heat capacity
- large amount of heat causes small increase in temperature
- high latent heat of vaporization
- large amount of heat is required to vaporize
- cohesive (due to hydrogen bonds)
- good solvent (water molecules are polar)
Describe the significance of water to living organisms (5)
adhesion - allows plants to transport water up during transpiration
cohesion - allows high surface tension - insects can move on surface
good solvent - can dissolve substances for transportation in organisms
high LOV - good coolant
high SHC - thermally stable aquatic habitats
Describe the use of carbohydrates and lipids for energy storage in animals (5)
carbohydrate
- stored as glycogen in liver
- short-term energy storage
- more easily digested so energy released quickly
lipid
- stored as fat in animals
- long-term energy storage
- more energy per gram than carb
List three functions of lipids (3)
- energy storage
- insulation
- internal organ protection
- buoyancy
- component of cell membranes
- electrical insulation - myelin sheath
- hormones (steroid)
Describe the significance of polar and non-polar amino acids (5)
polar:
- hydrophilic
- can make hydrogen bonds
- found on surface of water-soluble protein
non-polar
- hydrophobic
- found in interior of water soluble proteins
- found in protein in interior of membrane
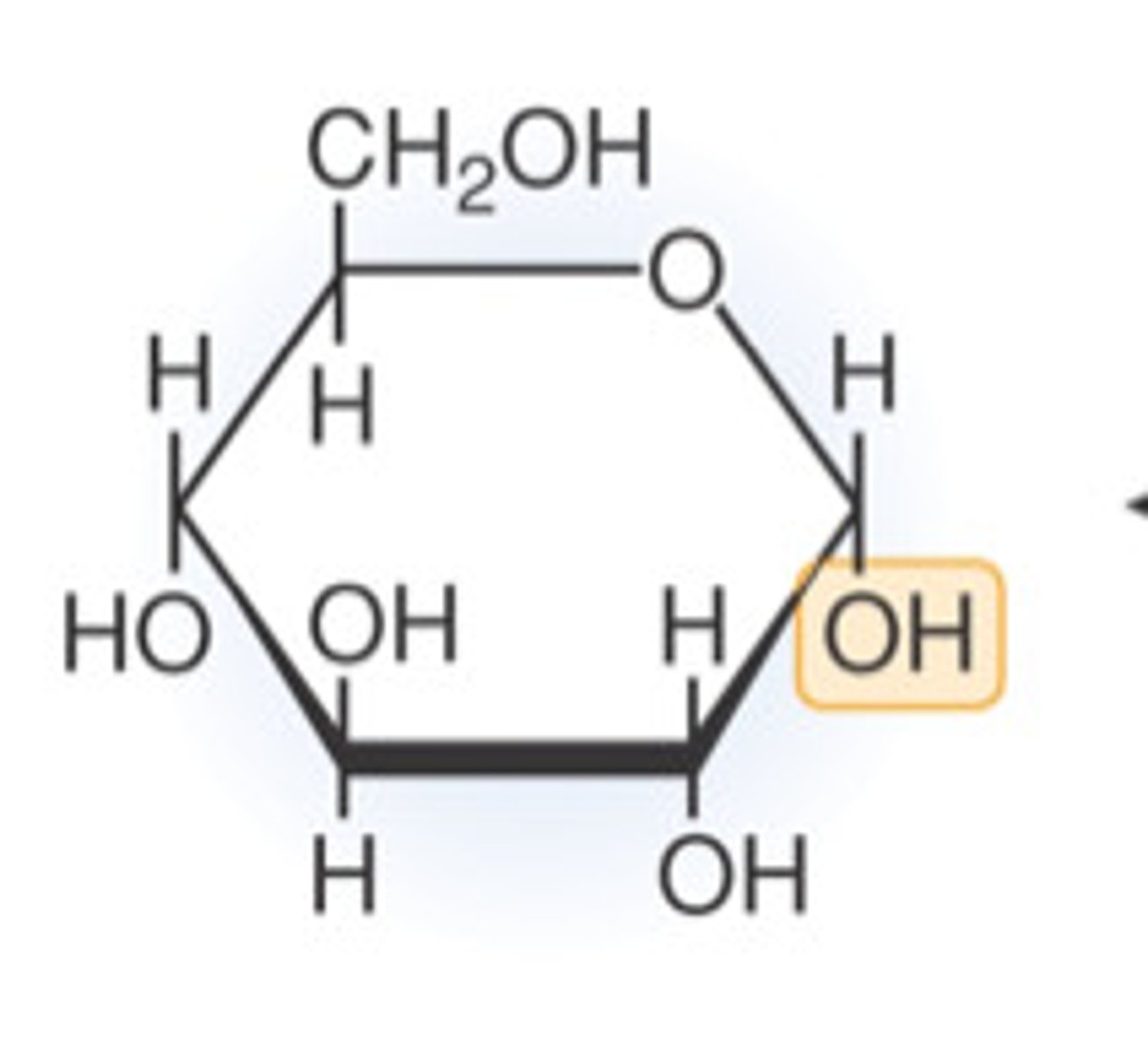
Draw and describe the structure of a glucose molecule (3)
C6H12O6
- 6-membered ring
- side chain
- hydroxyl group points _ _ ^ _
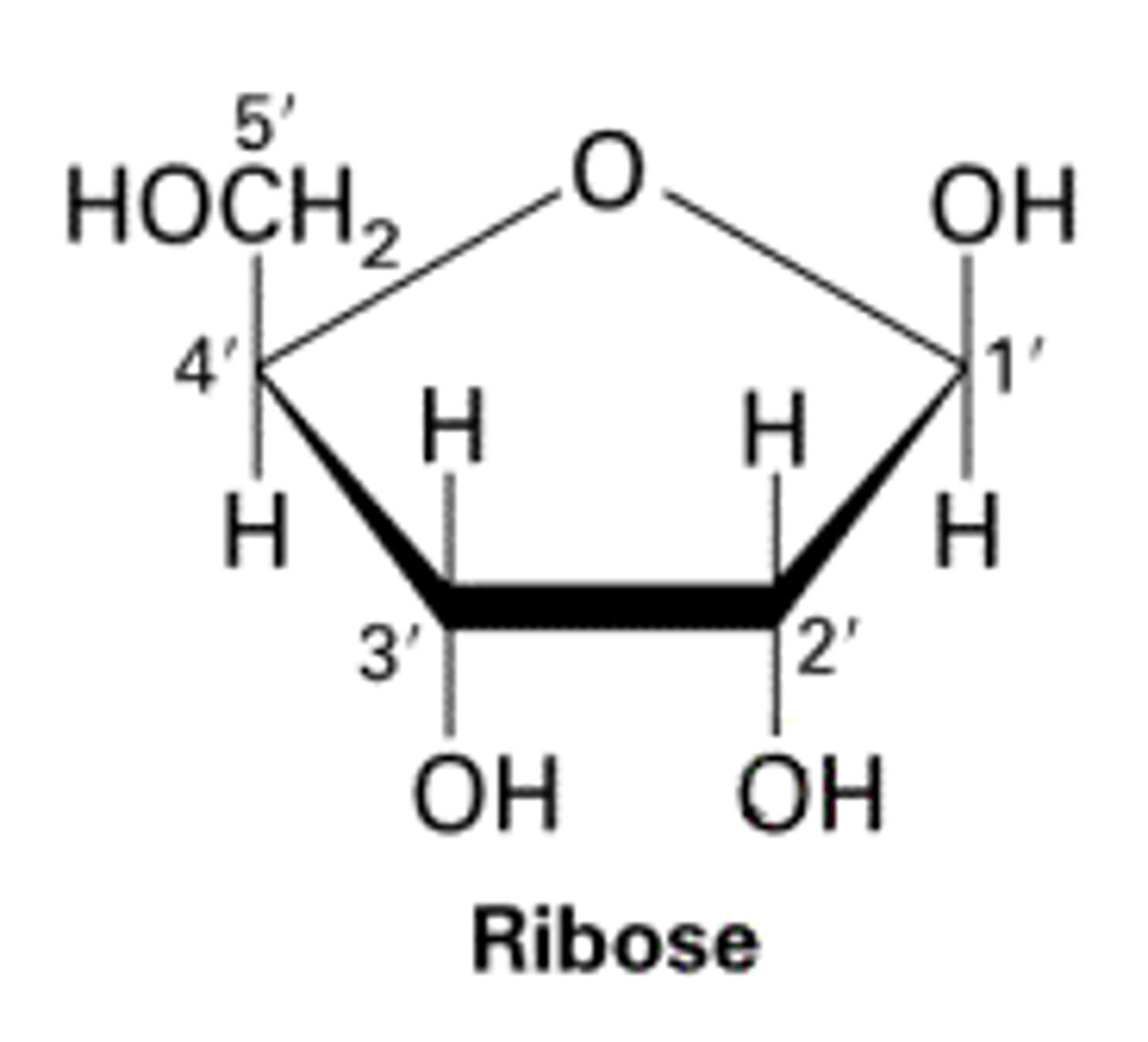
Draw and describe the structure of a ribose molecule (3)
C5H10O5
- 5 membered ring
- side chain
- ^ _ _ hydroxyl group
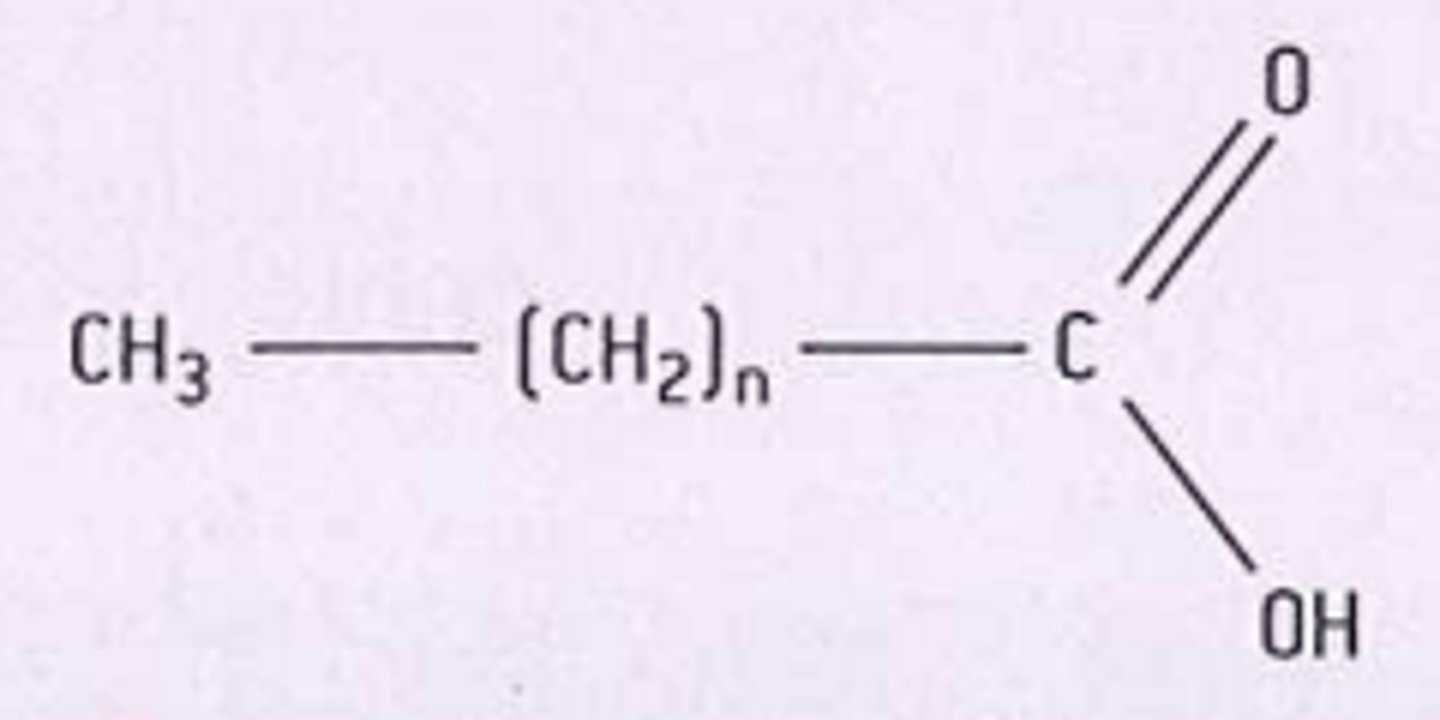
Draw and describe the structure of a saturated fatty acid (3)
- unbranched chain
- single bonds (saturated)
- no. carbon atoms = 14-20
- one end = carboxyl group
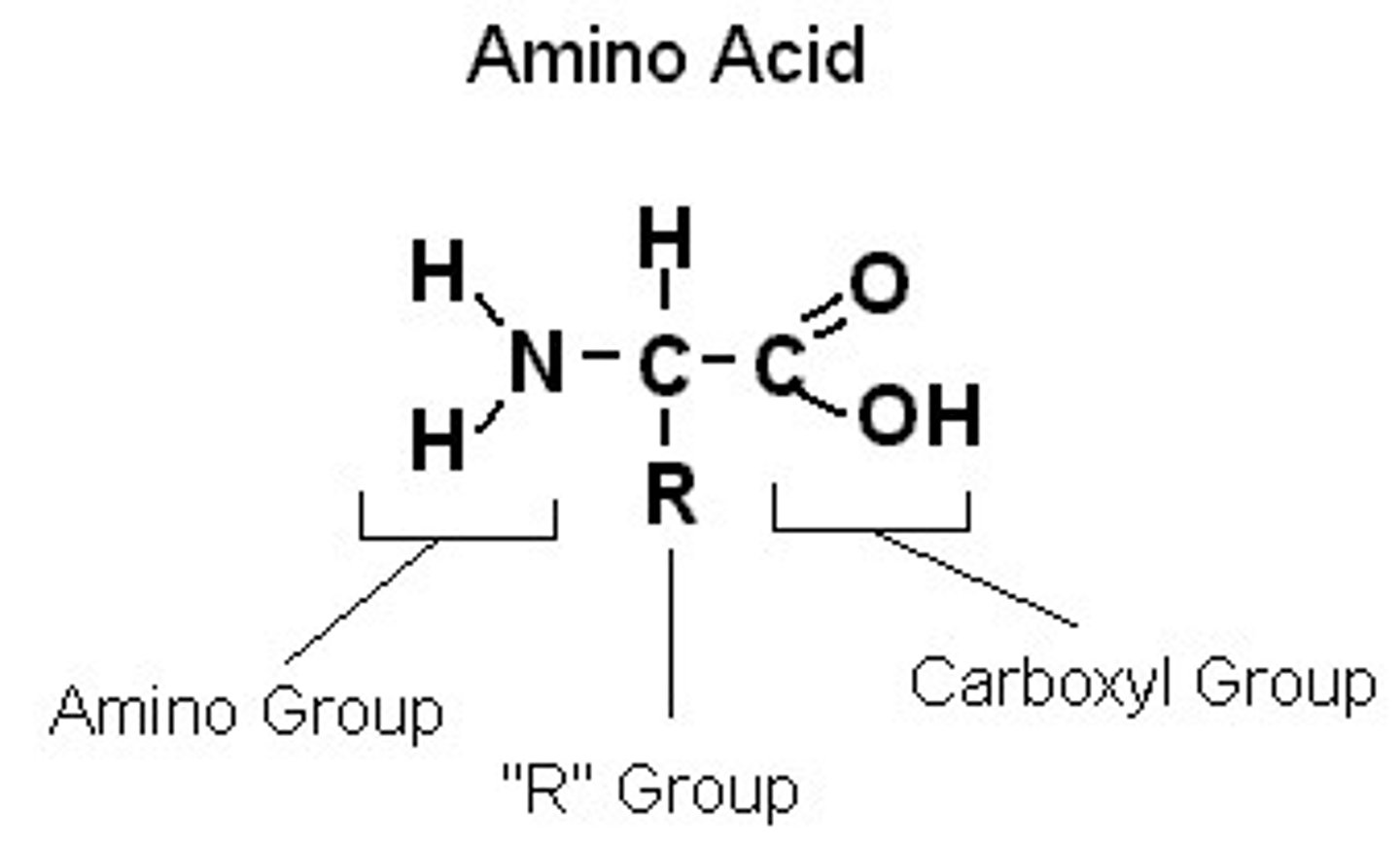
Draw and describe the structure of a generalised amino acid (3)
carbon bonded to:
- amine group
- carboxyl group
- hydrogen atom
- R group
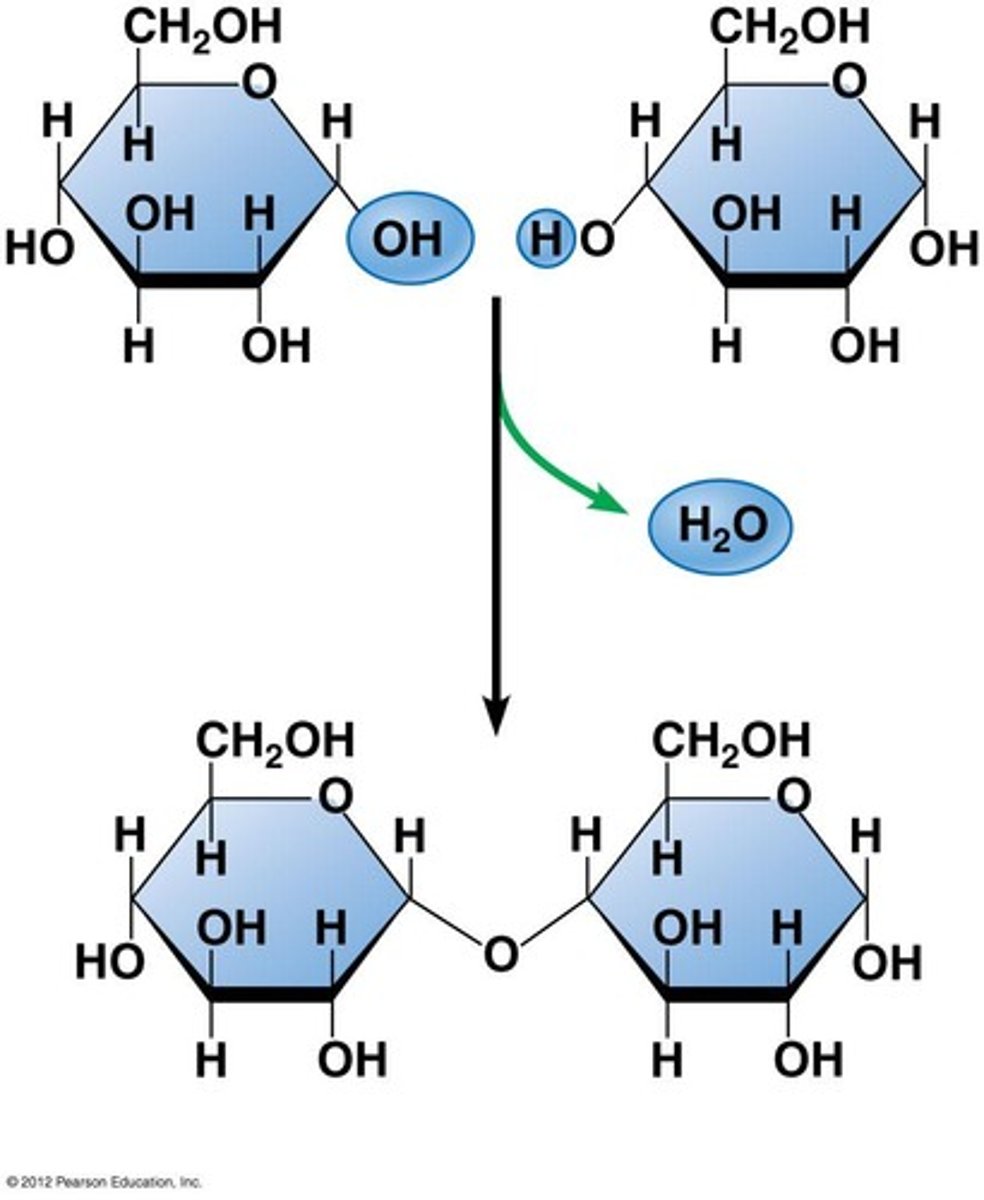
Draw and describe the formation of disaccharide (3)
glucose + glucose -> maltose + H2O
- via condensation reaction
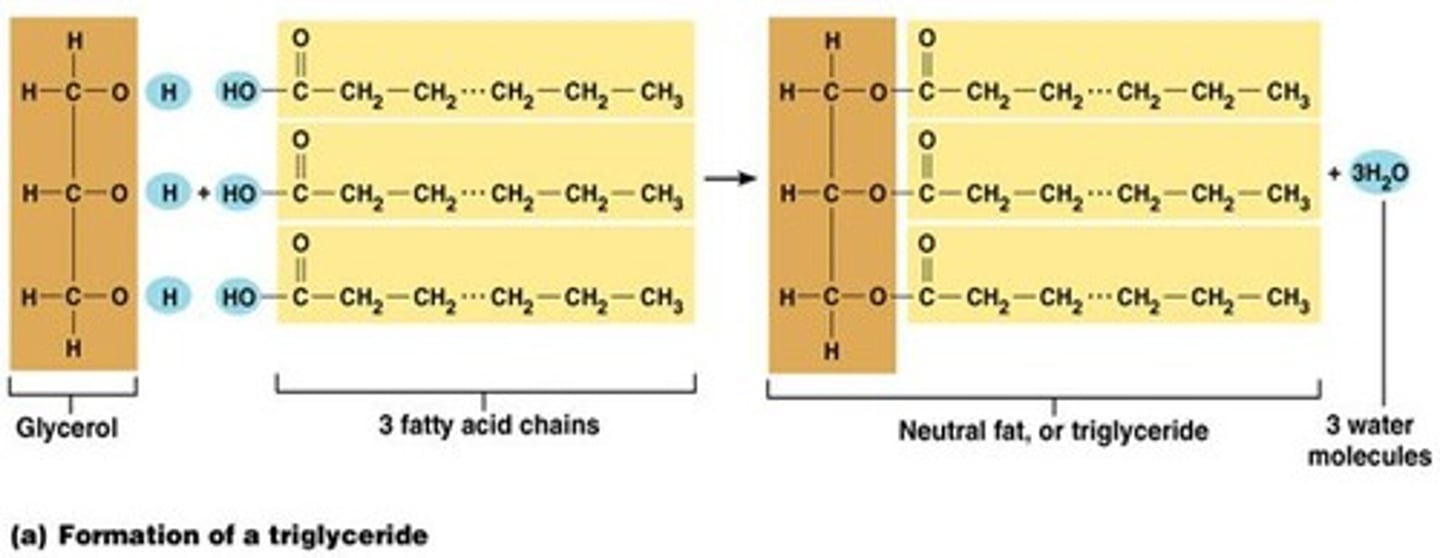
Draw and describe the formation of a triglyceride (6)
1 fatty acid + 3 glycerol = triglyceride (via condensation)
- held by ester bond
- composed of C, H, O
- non-polar hydrophobic molecule
- saturated if all are C-C
Outline the types of carbohydrates and their functions with examples (5)
Monosaccharides
- Energy source
e.g. glucose, galactose, fructose
Disaccharides
- transport form
e.g. lactose, sucrose, maltose
Polysaccharides
- storage form
e.g. cellulose, glycogen, starch
Outline the features of cellulose (5)
- beta-glucose subunit
- 1-4 glycosidic bond
- unbranched
- straight shape
- function = cell walls
Outline the features of amylose (5)
- alpha glucose subunit
- 1-4 glycosidic bond
- unbranched
- helix shape
function = energy storage
Outline the features of amylopectin (5)
- alpha glucose subunit
- 1-4 & 1-6 bond
- branched
- globular shape
function = energy storage
Outline features of glycogen (5)
- alpha glucose subunit
- 1-4 & 1-6 bond
- branched
- more compact shape than starch
- energy storage (humans)
Outline the difference between saturated and unsaturated fatty acids (3)
- fatty acids are long hydrocarbon chains
saturated = single bonds and linear, solid @ room temp
unsaturated = double bonds, bent, liquid @room temp
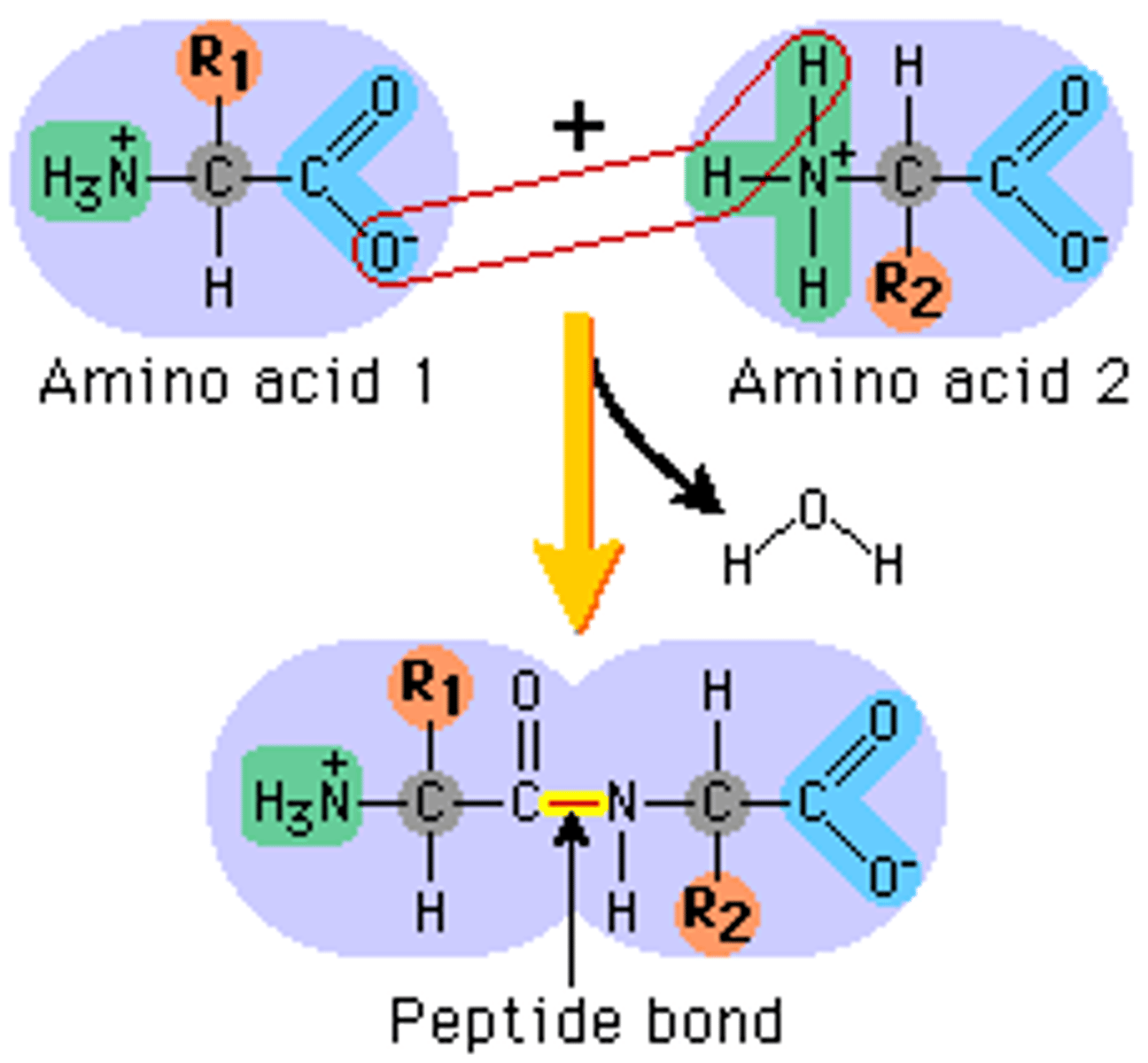
Outline the role of hydrolysis and condensation in the relationship between amino acids and dipeptides (4)
- condensation: water produced when 2 amino acids joined
- hydrolysis - water needed to break bond
Describe the structure of proteins (9)
- primary = chain of amino acids
- each position occupied by one of 20 diff amino acids
- linked by peptide bonds
- secondary = interaction between amino and carboxyl (N-H and C=O)
- weak hydrogen bond formed
- alpha helix/beta pleated sheet forms
- tertiary = folding of polypeptide
- stabilised by disulfide bridges/hydrogen/ionic bond
- quarternary = several polypeptide subunits join
- some combine with non-protein molecules
List 6 functions of proteins, giving examples of each (4)
STRUCTURE
- connective tissue of animals
e.g. collagen
HORMONES
- e.g. insulin
- hormone that triggers reduction in blood glucose levels
IMMUNITY
e.g. immunoglobin
- antibody that targets specific antigens
TRANSPORT
- nutrients and gases, e.g. hemoglobin
SENSATION
- receptors e.g. rhodopsin in eyes
- pigment that detects light
MOVEMENT
- muscle contraction (actin and myosin)
ENZYMES
- e.g. Rubisco
- enzyme catalysing CO2 fixation
Distinguish between cis and trans isomers (3)
cis: hydrogen atoms attached to C=C on same side
trans: hydrogen atoms attached to C=C on different side
- produced by hydrogenation (bad!)
Distinguish between fibrous and globular proteins with reference to one example of each (6)
- f = strand/sheets, g = ronuded
- f = usually insoluble, g = usually soluble
- g = more sensitive to change in pH
- f = have structural roles
- g = function roles (e.g. transport)
- f = e.g. collagen
= g = e.g. hemoglobin
Explain 3 reasons for converting lactose to glucose and galactose during food processing (3)
- allows people who are lactose intolerant to consume milk products
- galactose/glucose taste sweeter than lactose - reduce need for extra sweetener
- more soluble (smoother texture, reduce crystallization in ice cream)
- bacteria ferments them quicker - faster yogurt production time
Outline how enzymes catalyse reactions (7)
- increase rate of chemical reaction
- without being changed itself
- lowers activation energy
- due to kinetic energy and random collisons
- substrate joins with enzyme at active site
- substrate is complementary shape to active site
- forms enzyme substrate complex
- active site/enzyme is specific for particular substrate
-
Explain the effect of pH on enzyme activity (3)
- enzymes have optimal pH
- lower activity below and above optimum (graph)
- too high/low can denature enzyme
- change shape of active site/alter tertiary structure
- substrate cannot bind to active site - ESC cannot form
Compare the induced fit model of enzyme activity with the lock and key model (4)
- both models substrate binds to active site
- both - ESC forms
- L = substrate fits exactly, I = fit is not exact
- L = active site does not change shape, I = changes shape
- L = binding reduces EA, I = substrate reduces EA
- I explains competitive inhibition (L does not)
Outline the advantages of using immobilized enzymes (4)
an enzyme fixed to a static surface to improve efficiency of a catalysed reaction
- easily separated from products of reaction
- enzymes can be recycled
- increases enzymes stability (less prone to pH/temp change)
- substrates can be exposed to higher enzyme conc = faster reaction rate
Explain, using one example, the effect of a competitive inhibitor on enzyme activity (6)
- has similar shape/structure to substrate
- fits to active site
- substrate cannot bind as long as inhibitor is bound
- only 1 active site per enzyme molecule
- inhibitor and substrate compete for active site
- high substrate concentrations can overcome inhibitor
e.g. ethanol and alcohol dehydrogenase
Explain, using one example, the effect of a non-competitive inhibitor on enzyme activity (6)
- inhibitor has different shape to substrate
- binds to allosteric site
- causes conformational change on active site
- substrate cannot bind with enzyme
- substrate cannot prevent binding even at high conc
e.g. cyanide + cytochrome oxidase
Discuss the solubility of proteins in water (4)
- depends on what amino acids/R groups are present
- smaller proteins are more soluble than big ones
- proteins with many polar R groups are more soluble
- globular proteins are more soluble than fibrous
- affected by ph/temp
- denaturation make proteins insoluble
Outline enzyme-substrate specificity (5)
- active site of enzyme binds to specific substrate
- shape of substrate and active site complement
- lock and key
- chemical properties of substrate and enzyme attract
- induced fit
- active site is not rigid - substrate can induce slight changes in shape
- causes weakening of bonds in substrate to lower EA
Explain how proteins act as enzymes, including control by feedback inhibition in allosteric enzymes (9)
- enzymes are globular proteins
- there is active site
- substrate binds to active site
- shape of substrate and active site is changed
- bonds in substrate weakened
- activation energy reduced
- in feedback inhibition end product binds to enzyme
- end product is substance produced in last/later stage of a pathway
- inhibitor product binds to allosteric site
- causes active site to change shape
- substrate no longer fits active site
- higher conc of end product lowers enzyme activity
- whole pathway is inhibited
- prevents build up of intermediates
Define trophic level
- feeding level for an organism in a food chain
Compare the ways in which autotrophic, hetertrophic and saprotrophic organisms obtain energy (6)
- autotrophs use external energy source e.g. light
- heterotrophs obtain energy from other organisms
- usually ingest/consume food
- saprotrophs obtain energy from non-living matter/dead organisms
- saprotrophs digest organic matter extracellularly
Describe the relationship between the rise in concentration of atmospheric CO2 and the enhanced greenhouse effect (5)
- CO2 is a greenhouse gas
- increase in CO2 enhances greenhouse effect
- earth receives short wave radiation from sun
- reradiated from Earth as longer wave radiation
- CO2 absorbs longer wave radiation
- global warming happened same time as CO2 increase
- CO2 conc correlated positively with global temperature
Distinguish between unique and highly repetitive sequences in nuclear DNA (5)
U: occur once in genome, H: occur many times
U: long base sequences, H: short base sequences
U: may be genes, H: not genes
U: translated/coding sequences, H: never translated
U: small differences between individuals, H: can vary greatly
U: exons, H: introns
U: smaller genome prop., H: larger genome prop
- repetitive sequences used for profiling
Draw a labelled diagram of the structure of DNA (5)
- 4 nucleotides (one labelled)
- base, phosphate and deoxyribose labelled
- correct carbons labelled
- backbone labelled (covalent bond, 3-5 end)
- 2 base pairs linked by hydrogen bonds drawn as dotted lines
- 2 H bonds between A and T
- 3 H bonds between G and C
- antiparallel orientation shown
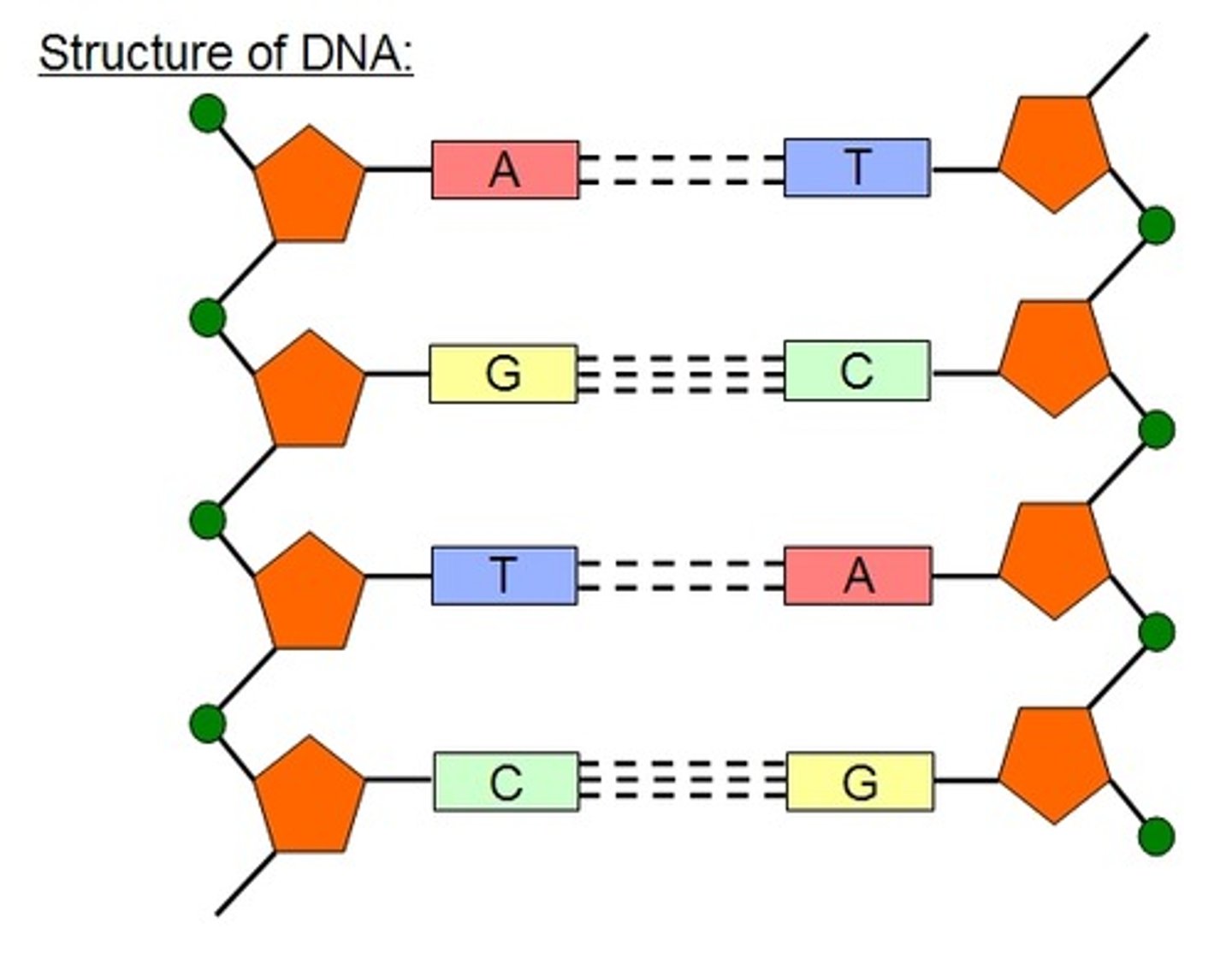
Explain the structure of the DNA double helix, including its subunits and the way in which they are bonded together (8)
- subunits are nucleotides
- one base, one deoxyribose and one phosphate in each nucleotide
- deoxyribose linked to C1, phosphate to C5
- 4 different bases (adenine, thymine, cytosine, guanine)
- covalent phosphodiester bonds
- 2 strands of nucleotides linked together
- base to base
- A-T, C-G
- hydrogen bonds between bases
- antiparallel strands
- double helix
Outline the structure of the nucleosome in eukaryotic chromosomes (4)
- contain histones
- 8 histone molecules form a cluster
- DNA strand wound around histones
- wound twice around each nucleosome
- histone molecule holds nucleosome in place
State a role for each of the 4 different named enzymes in DNA replication (6)
HELICASE
- unwinds DNA
DNA polymerase III
- adds nucleotides in 5' to 3' direction extending existing strand
RNA PRIMASE
- synthesizes short RNA primer on DNA (initiation point for DNA pol III)
DNA polymerase I
- replaces RNA primer with DNA nucleotides
DNA ligase
- joins Okazaki fragments on lagging strand (joins sugar-phosphate backbones with phosphodiester bond)
Explain the process of DNA replication (8)
- occurs during S phase of interphase in prep for mitosis
- semi-conservative
- unwinding of double helix by helicase at replication origin
- hydrogen bonds between 2 strands are broken
- each strand of parent DNA used as template of synthesis
- continuous on leading strand but not on lagging strand
- leads to formation of Okazaki fragments on lagging strand
- synthesis from 5-3' direction
- RNA primase synthesizes RNA primer on parent DNA
- DNA pol III adds nucleotides to 3' end
- added according to complementary base pairing
- adenine-thymine, cytosine - guanine
- DNA pol I removes RNA primers and replaces with DNA nucleotides
- DNA ligase joins Okazaki fragments
Describe how the process of DNA replication depends on the structure of DNA (9)
- dna molecule is double stranded
- hydrogen bonds linking two strands can be broken
- DNA can be split into two strands by helicase
- backbones are linked by covalent bonds - so strands do not break and base sequence is conserved
- base pairing is complementary
- A=T and C=G
- 2 original strands carry same info
- 2 new strands have same base sequence as 2 original
- base nucleotides added in 5-3' diretion
- strands have opposite polarity (antiparallel)
Outline what is meant by semi-conservative replication (3)
DNA replication is semi-conservative and depends on complementary base pairing
- 2 DNA molecules, 1 original and 1 newly synthesized
- ensures strands are identical in base sequence to replicated parent molecule
Describe the genetic code (6)
- composed of mRNA base triplets called codons
- 64 codons
- each codes for the addition of an amino acid to a polypeptide chain
- genetic code is degenerate
- i.e. more than one codon can code for a particular amino acid
- genetic code is universal
- i.e. same in all organisms
- AUG is the start codon
- some codons code for the end of translation
Discuss the relationship between genes and polypeptides (5)
- originally assumed that one gene = one polypeptide = one mRNA
- many more proteins made than genes available
- some genes do not code for polypeptide
- some code for tRNA/rRNA
- some regulate gene expression
- polypeptides may be altered before becoming functional proteins
Explain briefly the advantages and disadvantages of the universality of the genetic code to humans (4)
- genetic material can be transferred between species/humans
- one species can use useful gene from another
- transgenic crop plants/livestock can be produced
- bacteria/yeast can be genetically engineered (useful)
X viruses can invade cells and take over genetic apparatus
X viruses cause disease
Compare the processes of DNA replication and transcription (9)
SIMILARITIES
- unwinding of helix
- separating 2 strands
- breaking hydrogen bonds between bases
- complementary base pairing involved
- C-G pairing
- 5'-3' direction
- linking of nucleotides
- start signal required
DIFF:
- R = DNA nucleotides, T = RNA molecules
- R = deoxyribose nucleotide, T = ribose nucleotide
- R = A-T, T = A-U
- R = both copied, T = 1 copied
- diff start signal
- R = 2 DNA molecules, T = mRNA
Distinguish between RNA and DNA (3)
- DNA is double stranded while RNA is single stranded
- DNA contains deoxyribose, RNA contains ribose
- Thymine in DNA, Uracil in RNA
- one form of DNA (double helix) but several forms of RNA (mRNA, tRNA, rRNA)
Describe the roles of mRNA, tRNA and ribosomes in translation (6)
- mRNA contains genetic code
- ribosomes have large subunit and small subunit
- tRNA with amino acid attached
- tRNA with anticodon
- mRNA held by ribosome
- 2 tRNA molecules attached to mRNA on ribosome
- peptide bond forms between amino acids on tRNA
- polypeptide forms
- continues until stop codon is reached
- polypeptide is released
Define transcription
The synthesis of mRNA copied from the DNA base sequence by RNA polymerase
Outline the structure of tRNA (5)
- one chain of RNA nucleotides
- has site for attaching amino acid
- at 3' end
- has anticodon
- of 3 unpaired bases forming a loop
- double stranded sections formed by base pairing
- has cloverleaf shape
- 3 loops
Outline the structure of ribosome (4)
- large subunit and small subunit
- mRNA binding site on small subunit
- 3 tRNA binding sites (E, P, A)
- made of protein and ribosomal RNA
Outline the use of Taq DNA polymerase (5)
- used to produce multiple copies of DNA rapidly by polymerase chain reaction
1. Denaturation - heating to 95 C to separate 2 strands
2. Annealing - sample cooled to 54C, primers designate sequence to be copied
3. Elongation - heated to 75 C, Taq polymerase can function optimally (extends nucleotide chain from primers)
4. cycle repeated
Outline the role of tandem repeats in DNA profiling (4)
variable number tandem repeat = short nucleotide sequence that shows variations between individuals in terms of no. of times sequence is repeated
- restriction enzymes used to cut DNA between tandem repeats
- electrophoresis to calculate length of tandem repeat
- profiling!