Molecular Orbitals
0.0(0)
Card Sorting
1/27
Earn XP
Description and Tags
SQA Advanced Higher Chemistry 2023
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
28 Terms
1
New cards
When do molecular orbitals form?
When atomic orbitals combine.
2
New cards
What is the number of molecular orbitals formed equal to?
The number of atomic orbitals that combine.
3
New cards
What does the combination of two atomic orbitals result in?
The formation of a bonding molecular orbital and an antibonding orbital.
4
New cards
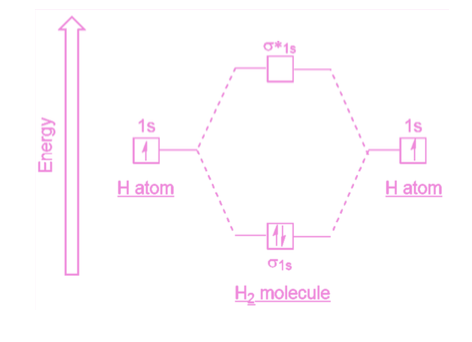
How many electrons can be held in a molecular orbital? And how are these electrons added?
Each molecular orbital can hold a maximum of two electrons. Electrons are added to orbitals in order of increasing energy.
5
New cards
Describe the bonding molecular orbital.
The bonding molecular orbital encompasses both nuclei. It is where the electrons spend most of their time between the two nuclei.

6
New cards
What is the basis of bonding between atoms?
The attraction of the positively charged nuclei and the negatively charged electrons in the bonding molecular orbital.
7
New cards
What determines the type of bonding?
The symmetry and position of the bonding orbitals between atoms.
8
New cards
How is the bonding molecular orbital in a pure (non-polar) covalent bond?
It is symmetrical about the midpoint between two atoms (where the bonding electrons are predicted to be found).

9
New cards
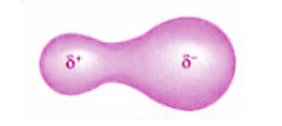
How is the bonding molecular orbital in a polar covalent bond?
It is asymmetric about the midpoint between two atoms. The atom with the greater value for electronegativity has the greater share of the bonding electrons.
10
New cards
How is the bonding molecular orbital in an ionic compound?
It is an extreme case of asymmetry, with the bonding molecular orbitals being almost entirely located around just one atom, resulting in the formation of ions.

11
New cards
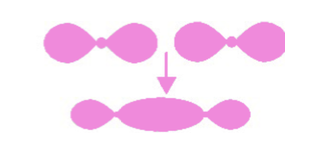
Describe sigma bonds.
Molecular orbitals that form by end-on overlap of atomic orbitals along the axis of the covalent bond are called sigma (σ) molecular orbitals or sigma bonds.
12
New cards
Describe pi bonds.
Molecular orbitals that form by side-on overlap of parallel atomic orbitals that lie perpendicular to the axis of the covalent bond are called pi (π) molecular orbitals or pi bonds. These arise where atoms make multiple bonds.

13
New cards
Compare the strengths of sigma and pi bonds.
End to end overlap is more efficient than side on overlap and therefore σ bonds are stronger than π bonds.
14
New cards
What is hybridisation?
Hybridisation is the process of mixing atomic orbitals within an atom to generate a set of new atomic orbitals called hybrid orbitals.
15
New cards
Describe the hybrid orbitals.
Degenerate.

16
New cards
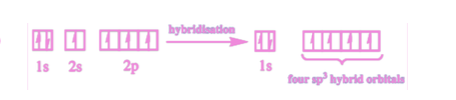
Describe sp³ hybridisation in alkanes.
In alkanes, the 2s orbital and the three 2p orbitals of carbon hybridise to form four degenerate sp3 hybrid orbitals. These adopt a tetrahedral arrangement (to minimise repulsion).
The sp3 hybrid orbitals overlap end-on with other atomic orbitals to form σ bonds.
The sp3 hybrid orbitals overlap end-on with other atomic orbitals to form σ bonds.
17
New cards
Describe sp² hybridisation in alkenes.
The bonding in alkenes can be described in terms of sp2 hybridisation. The 2s orbital and two of the 2p orbitals hybridise to form three degenerate sp2 hybrid orbitals.These adopt a trigonal planar arrangement.
The hybrid sp2 orbitals overlap end-on to form σ bonds. The remaining 2p orbital on each carbon atom of the double bond is unhybridised and lies perpendicular to the axis of the σ bond. The unhybridised p orbitals overlap side-on to form π bonds.
The hybrid sp2 orbitals overlap end-on to form σ bonds. The remaining 2p orbital on each carbon atom of the double bond is unhybridised and lies perpendicular to the axis of the σ bond. The unhybridised p orbitals overlap side-on to form π bonds.
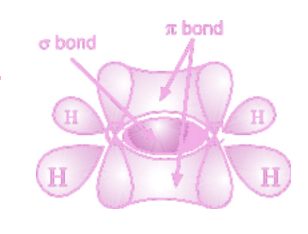
18
New cards
How can the bonding in benzene and other aromatic systems be described?
The bonding in benzene and other aromatic systems can be described in terms of sp2 hybridisation. The six carbon atoms in benzene are arranged in a cyclic structure with σ bonds between the carbon atoms. The unhybridised p orbitals on each carbon atom overlap side-on to form a π molecular system, perpendicular to the plane of the σ bonds. This π molecular system extends across all six carbon atoms. The electrons in this system are delocalised.
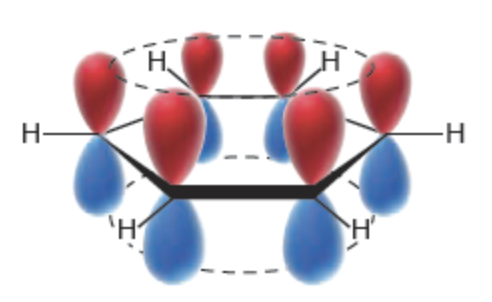
19
New cards
Describe sp hybridisation in alkynes.
The bonding in alkynes can be described in terms of sp hybridisation. The 2s orbital and one 2p orbital of carbon hybridise to form two degenerate hybrid orbitals. These adopt a linear arrangement.
The hybrid sp orbitals overlap end-on to form σ bonds. The remaining two 2p orbitals on each carbon atom lie perpendicular to each other and to the axis of the σ bond. The unhybridised p orbitals overlap side-on to form two π bonds.
The hybrid sp orbitals overlap end-on to form σ bonds. The remaining two 2p orbitals on each carbon atom lie perpendicular to each other and to the axis of the σ bond. The unhybridised p orbitals overlap side-on to form two π bonds.
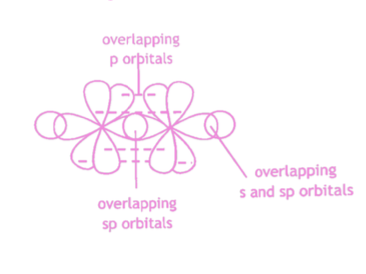
20
New cards
What is the highest bonding molecular orbital containing electrons called?
The highest occupied molecular orbital (HOMO).
21
New cards
What is the lowest antibonding molecular orbital called?
The lowest unoccupied molecular orbital (LUMO).
22
New cards
What can cause electrons to be promoted from HOMO to LUMO?
Absorption of electromagnetic energy.
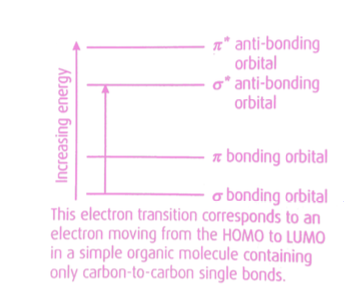
23
New cards
Why do most organic molecules appear colourless?
Because the energy difference between HOMO and LUMO is relatively large. This results in absorption of light from the ultraviolet region of the spectrum (so no visible light is absorbed).
24
New cards
What is a chromophore?
A group of atoms within a molecule that is responsible for absorption of light in the visible region of the spectrum. Light can be absorbed when electrons in a chromophore are promoted from the HOMO to the LUMO.
25
New cards
Where do chromophores exist?
In molecules containing a conjugated system.
26
New cards
What is a conjugated system?
A system of adjacent unhybridised p orbitals that overlap side-on to form a molecular orbital across a number of carbon atoms. Electrons within this conjugated system are delocalised.
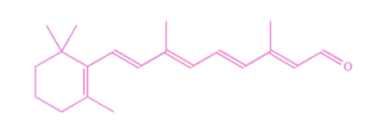
27
New cards
What are some molecules that have conjugated systems?
Molecules with alternating single and double bonds, and aromatic molecules.
28
New cards
What happens when there are more atoms in the conjugated system?
The smaller the energy gap between HOMO and LUMO since a lower frequency of light (longer wavelength, lower energy) is absorbed by the compound. When the wavelength of light absorbed is in the visible region, the compound will exhibit the complementary colour.