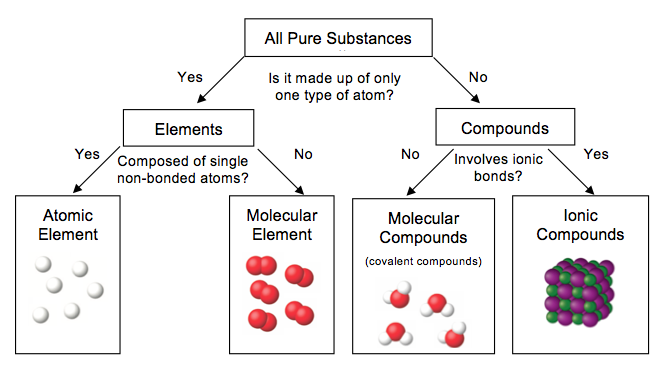
Classification of Matter
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
4 Terms
Compounds and Their Composition:
A compound is a substance made up of two or more different types of atoms bonded together.
The atoms in a compound combine in whole-number ratios. For example, a compound could have 1 red atom for every 1 blue atom.
This means that each particle of the compound always has the same number of atoms in the same ratio, giving the compound a fixed composition.
Because of this fixed ratio, compounds are considered pure substances. Every particle is the same, and there’s no mix of different types of substances.
A mixture:
contains 2 or more substances.
Mixtures:
A mixture is made by combining two or more substances without changing their chemical identities.
Mixtures can be homogeneous or heterogeneous:
Homogeneous mixtures have the same distribution of components throughout (like a well-mixed smoothie).
Heterogeneous mixtures have uneven distribution of components (like a salad, where you can see the individual ingredients).
Classification of matter