Aquatic Chemistry IV - Organometal & Water Quality
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
30 Terms
What metal series are the biggest environmental concerns?
First transition series - scandium to zinc
Post transition series - indium to polonium
the heavy metals post transition (lead, cadmium, mercury) have large bioconcentration factor in marine organisms and are highly toxic with no known biological function
How do metals usually enter the environment?
as an insoluble through industrial waste
or as part of naturally occurring mineral deposits
Deposition from atmosphere will normally be as insoluble salts
How does pH impact the solubility of metals in water?
as pH decreases (more acidic), the solubility of metals in water increases - this is the acidity effect
an increase in pH (more alkaline) will eventually make transition metals precipitate and form sediments
How does acid rain and a low pH affect metals in the environment?
Acid rain affects the aqueous environment as toxic metal leaching will have a direct effect on the fish species and the acidity will affect the pH of the water they live in
Lead piping in soft acidic water areas is also a lot more problematic due to the increased solubility under acidic conditions than alkaline water areas.
What is co-precipitation?
When there is deposition of a high concentration of metals, there may be traces of other metal ion also deposited - this is co-precipitation
What are some common ligands in sediment formation?
Cl-
SO42-
OH-
How are metals taken in by organisms in water?
via the food chain
filter feeders like fish, sponge, mussels, oysters etc will take in metals from sediment
these metals will remain as ions in organisms but some (cadmium and mercury) can be converted into covalent organometals that can accumulate in fatty tissue and cause damage
What are suspended solids and some examples?
Small, solid particles that remain in suspension in water
May be in the form of bed sediments (clay, silt), organic/inorganic matter or algae and microscopic organisms
What are the two categories of supsneded solids?
Total Suspended Solids - TSS - solids that can be removed by filtration - heterogeneous mixtures
Total Dissolved Solids - TD - solids that are dissolved in water
How can water quality be analysed? (7 ways)
Turbidity - measure of the clarity (cloudiness due to particles in the water)
colour, odour, taste
pH - acidity or alkalinity
temperature
dissolved oxygen levels
total organic carbon (TOC)
Water Hardness
What must be done prior to testing a water sample?
collect sample and transport and store until use
Preservatives may be used to reduce sample degradation- e.g. nitric acid is an oxidizing agent that will keep metals dissolved in the solution and minimize their precipitation
What is turbidity?
the measure of relative clarity of the fluid
the haziness/cloudiness of the sample due to the presence of individual particles
these particles may not be visible to the naked eye and only be seen when there is a large number present
The particles are usually fine suspended solids like bed sediment (clay, silt) or fine organic and inorganic matter
Characteristics of water flow and agitation of precipitates solids are major factors of water turbidity
How is turbidity determined?
It’s determined by the amount of light that is scattered off the surface of the particles in the fluid - the light will be obstructed by the particles causing the cloudiness to appear
What factors affect turbidity?
water flow - direction, speed, changes in elevation
Point source pollution - pipelines from industrial applications, water treatment, urban and residential activity
Land use - run-off from agriculture and animal agriculture, construction etc
Resuspension - agitation of settles solids, becoming suspended
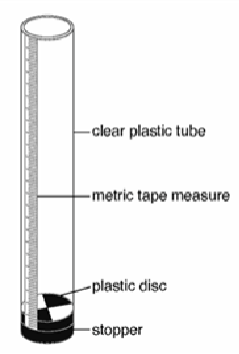
What is the method of measuring turbidity with a Turbidity Tube?
Measured in NTU
water is poured into the tube until the plastic disc is no longer visible
the level of turbidity is read of the measure on the tube
This method can be used at the water source
What is Turbidity measured in?
Nephelometric Turbidity Unit - NTU
NTU are established relative measures that are based on the intensity of light scattered by a sample to the intensity of light scattered by a standard reference suspension
the NTU values for surface water are 1 - 1000 and filtered water under steady state operation are 0.05 - 0.15 NTU
How is turbidty measured using a turbidimeter?
more accurate method compared to the tube
It measures the intensity of light passing through the sample and calculates the turbidity in NTU and compares the real and reference intensity

How is water colour tested?
water samples undergo visual inspection and compared to the relevant scale - the APHA/Pt-Co scale (platinum-cobalt) which measures the yellow colouring in water.
As water is colourless, discolouration can be caused by dissolved organic matter and suspended solids
Colour will usually bet tested in conjunction with turbidity using electronic instrumentation
How is odour measured?
Measured using the Threshold Odour Number (TON)
This is the dilution ratio at which odour is just detectable by human sense of smell
IF the odour is no longer present, it has been diluted too much
If the odour is still too strong, it has not been diluted enough
TON is a whole number that indicates how many times the sample needs to be diluted with odour-free water before the odour is only just detectable

What odour/taste indicates Hydrogen Sulphide is present? what is the treatment for this?
Rotten egg odour - high levels of hydrogen sulphide that have been produced by bacteria in an oxygen deficient environment
Sewage or musty smell - hydrogen sulphide present in lower levels
Treatments: chlorination, aeration, filtration
Where is hydrogen sulphide present and what causes it?
It is present in still, light limited bodies of water - wells, vats/containers
Degrading macroalgae (seaweed) and microalgae release H2S
High concentrations of H2S are deadly to humans and animals
What odour/taste indicates calcium or iron is present? what is the treatment for this?
Sweet taste - the sweetness comes from imbalances in alkaline and pH levels
Caused by high levels of calcium or iron in the water
Hard water is often described as having a sweet taste due to alkaline properties
Treatments: flushing water transfer systems
What odour/taste indicates Chloride ions are present? what is the treatment for this?
Salty taste - saltiness is attributed to higher levels of chlorides present - this may be due to industrial applications
E.g. sea water has high levels of sodium chloride, hence the salty taste
Treatment: distillation, osmosis
Why is rainwater naturally slightly acidic? (not acid rain)
The presence of dissolved CO2
The weak carbonic acid gives rainwater a pH of 5.6
How are minerals affected by low pH in water?
minerals like limestone and dolomite will dissolve more rapidly when the pH decreases
CaCO3 + CO2 (aq) + H2O = Ca2+ (aq) + 2HCO3- (aq)
What else causes acidity in water?
Increases in the production of SO2 and NO2 from fossil fuel burning
These gases disperse in the atmosphere via tall chimneys as flue gas
The gases react with O2 and atmospheric liquid water to form H2SO4 and HNO3
This forms acidic aqueous solutions
2SO2 (g) + O2 (g) → 2SO3 (g)
SO3 (g) + H2O → H2SO4 (aq)
NO (g) + ½ O2 (g) → NO2 (g)
3NO2 (g) + H2O → 2HNO3 (aq) + NO (g)
What is acid rain?
rainfall or precipitation with a high acidic content
It contaminates fresh water, ground water and soil and decreases the systems pH and also results in damage to plant life
How does water pH chanes from acid rain affect freshwater fish population -?
pH below 6.5 - Salmon population decrease
pH below 6.0 - Perch population decrease
pH below 5.5 - Eel population decrease
pH below 5.0 - little life possible
How does temperature affect the characteristics of water?
Temperature will impact the chemical and biological characteristics
It can affect metabolic rates and photosynthesis in aquatic life: metabolic rates will increase with temperature, photosynthesis will peak around 40°C and then the rate will decrease if temp goes beyond 40°C.
Increased temp will increase solubility
Dissolved oxygen and gas concentrations will decrease with a temp increase
The pH of the system will decrease with temperature causing acidity
What causes the water temperature to change?
Solar energy and atmospheric heat transfer
A change in the water turbidity
Conflunce - where water sources meet and heattrasnfer occurs from warmest to coolest
Thermal pollution - effluent streams entering natural water courses at significantly higher temperatures