4. Atomic structure
1/142
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
143 Terms
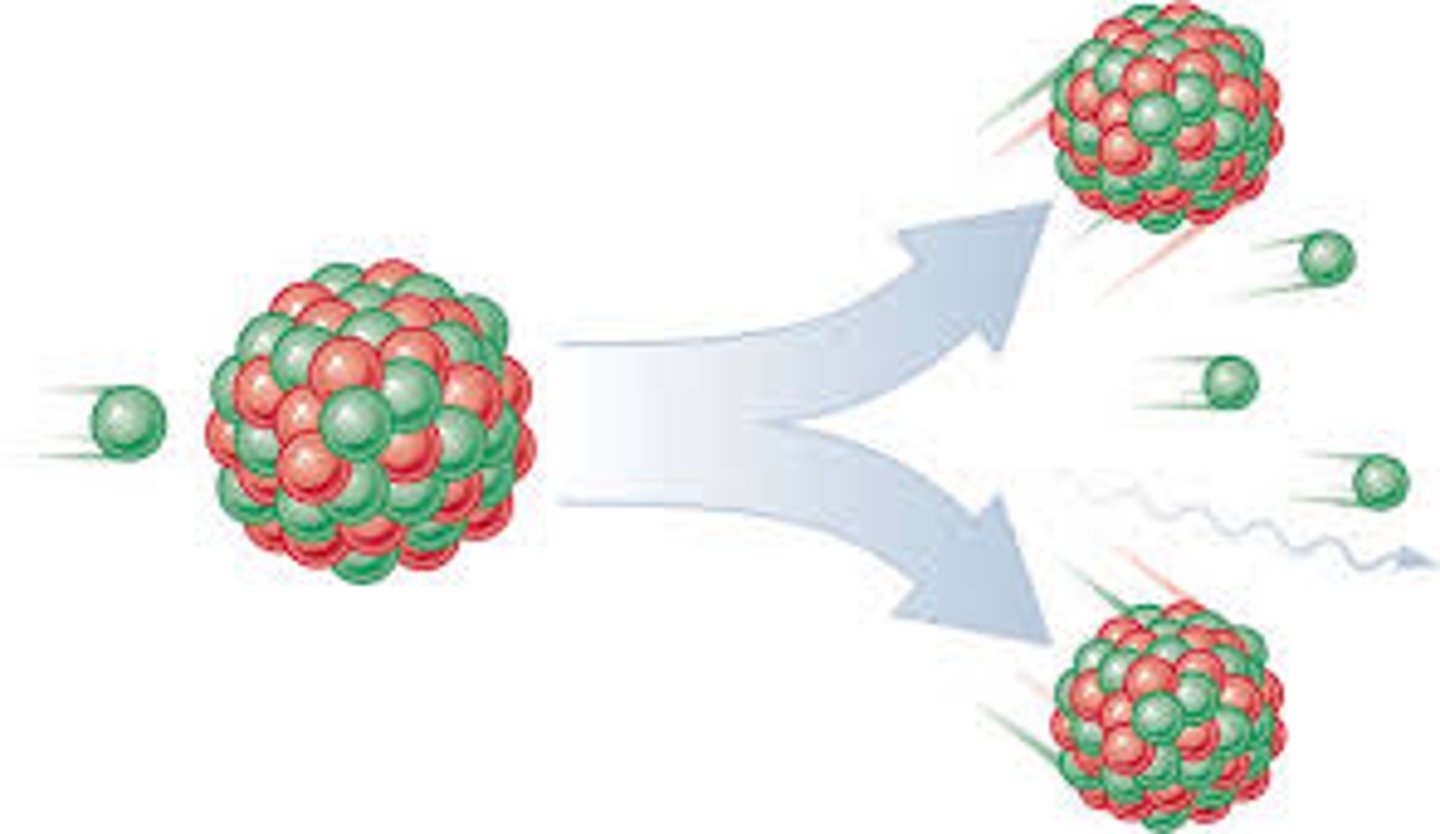
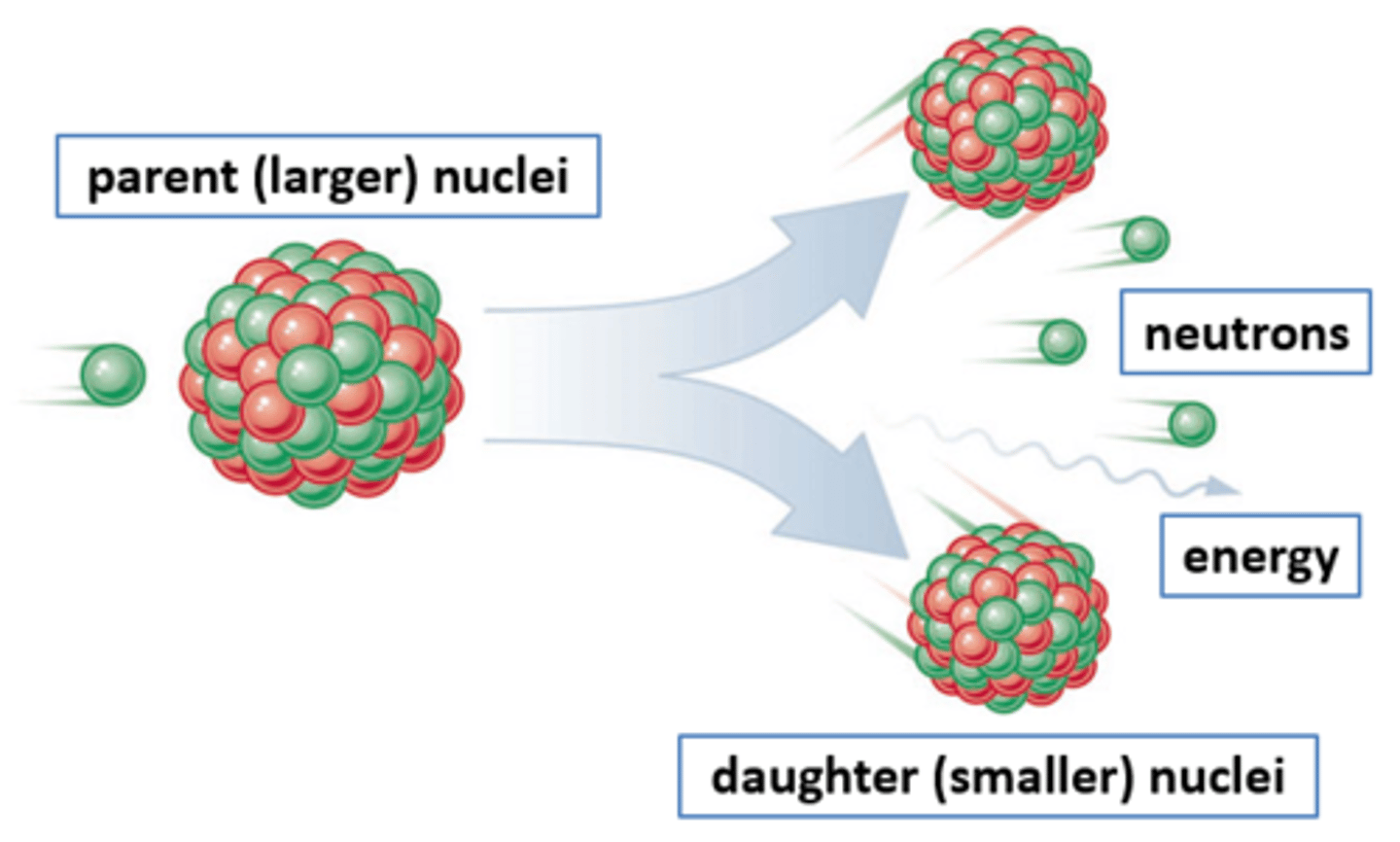
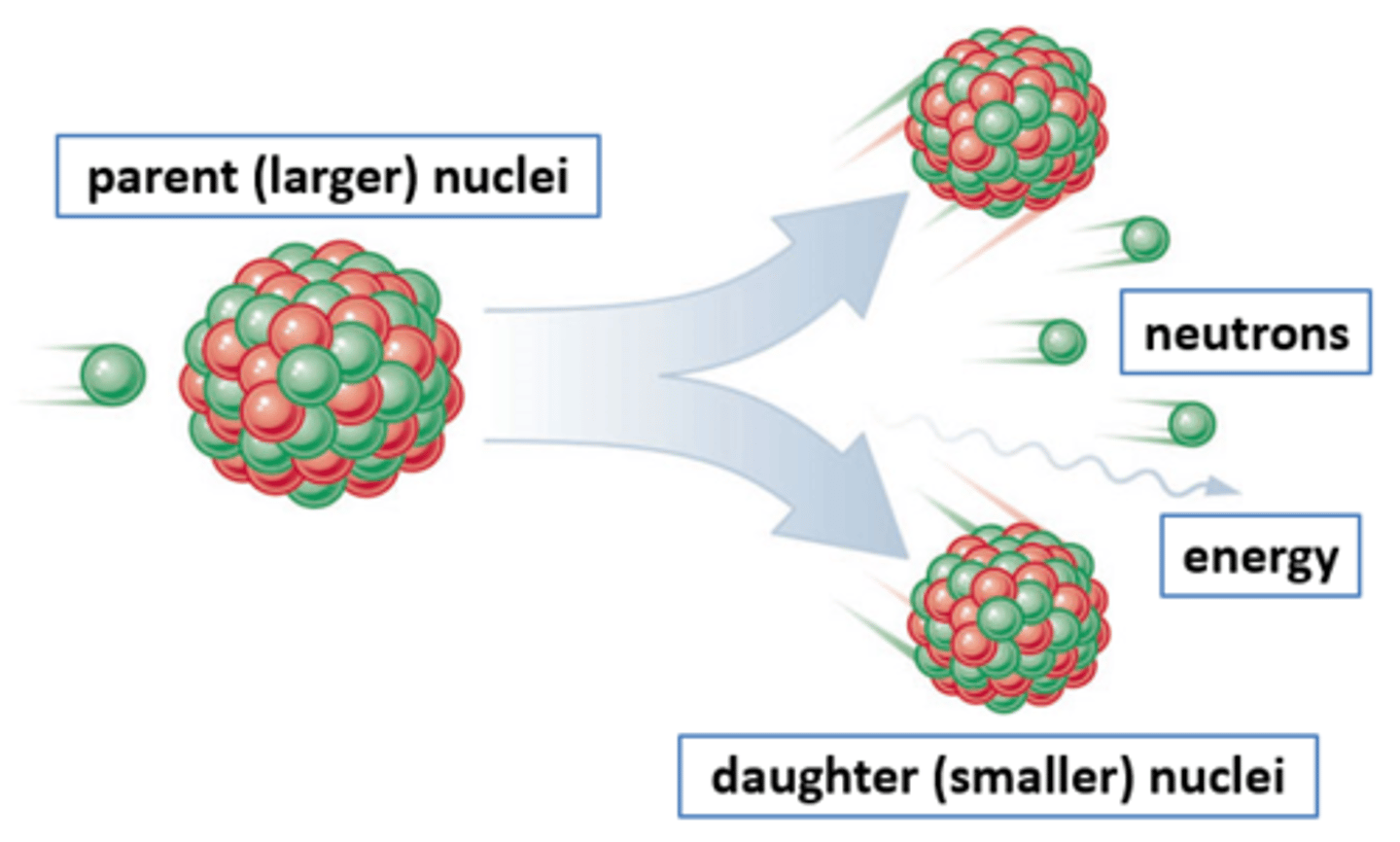
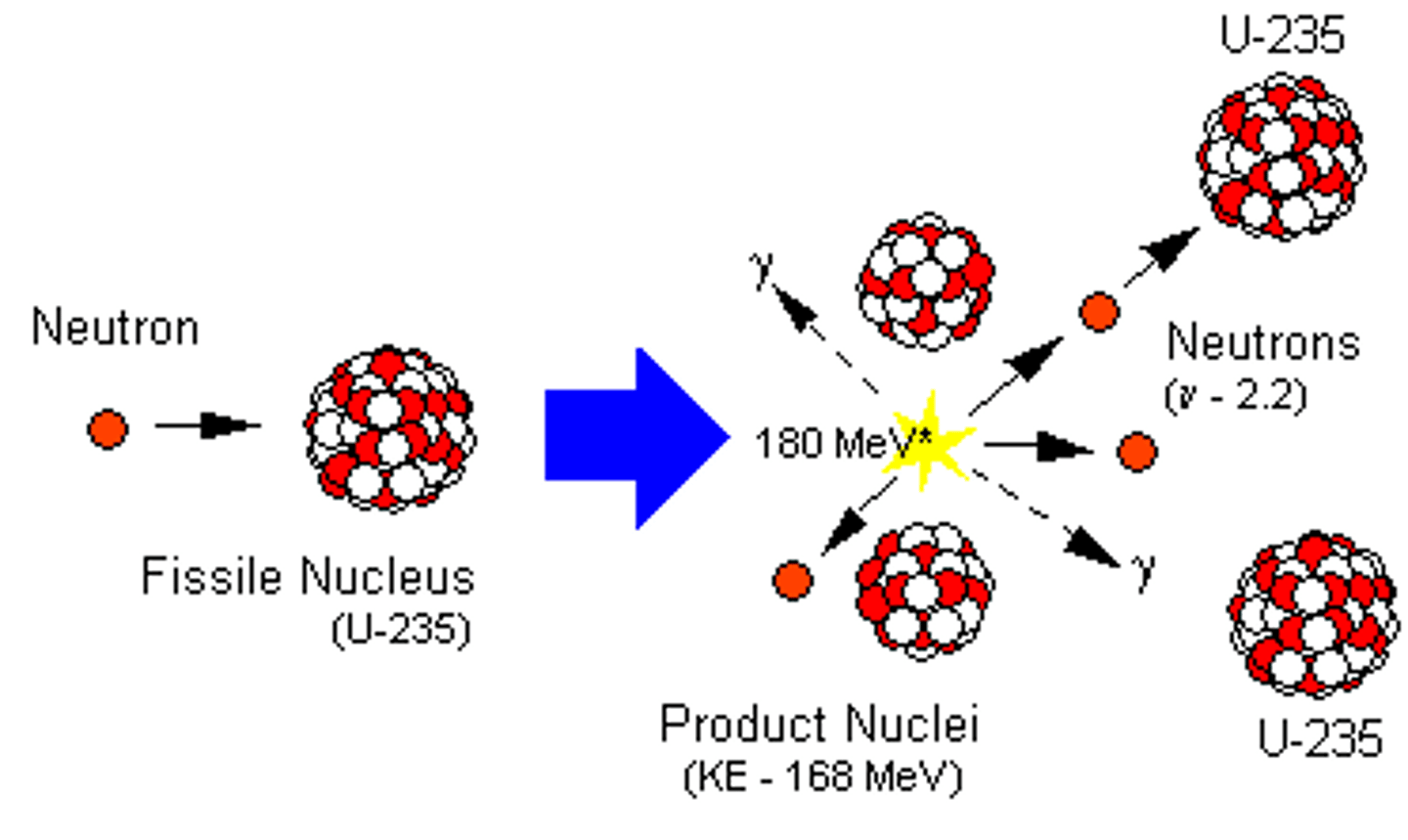
Nuclear fission
The splitting of a large and unstable nucleus
Fissile material
A material whose nuclei can split by nuclear fission e.g. uranium and plutonium

Spontaneous fission
The spontaneous splitting of a large and unstable nucleus (very rare)
Uranium-235
An unstable, fissionable isotope of uranium that is used in most nuclear reactors
Neutron (slow-moving)
An unstable nucleus must first absorb a slow-moving neutron to make it split
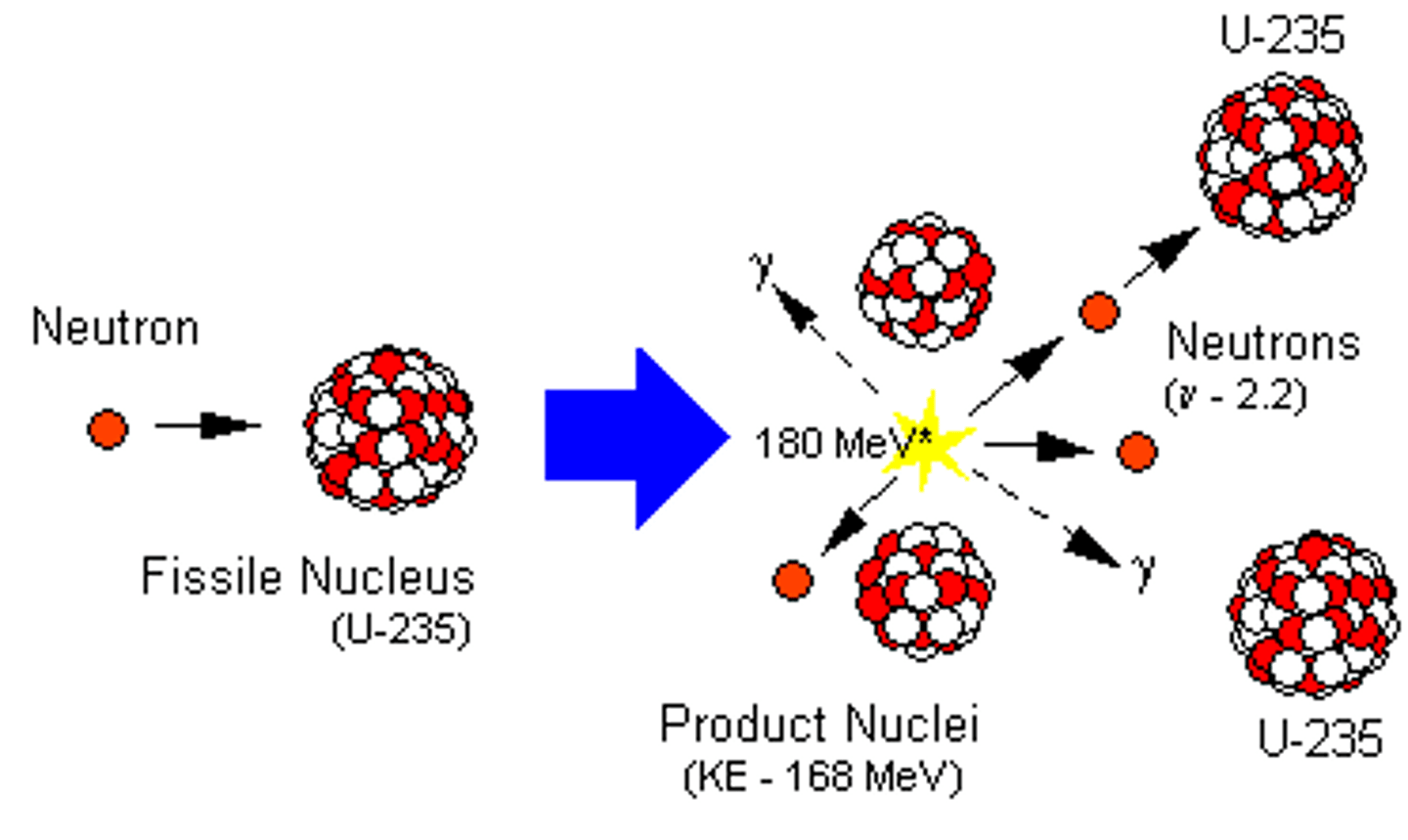
Uranium-236
An extremely unstable isotope of uranium that is formed when uranium-235 absorbs a neutron
Fission products
Two smaller nuclei (roughly equal in size), two or three neutrons and energy in the form of gamma rays
Energy of fission products
All of the fission products have kinetic energy so will move away from each other
Parent nuclei
The unstable nuclei that splits by fission (usually uranium-235)
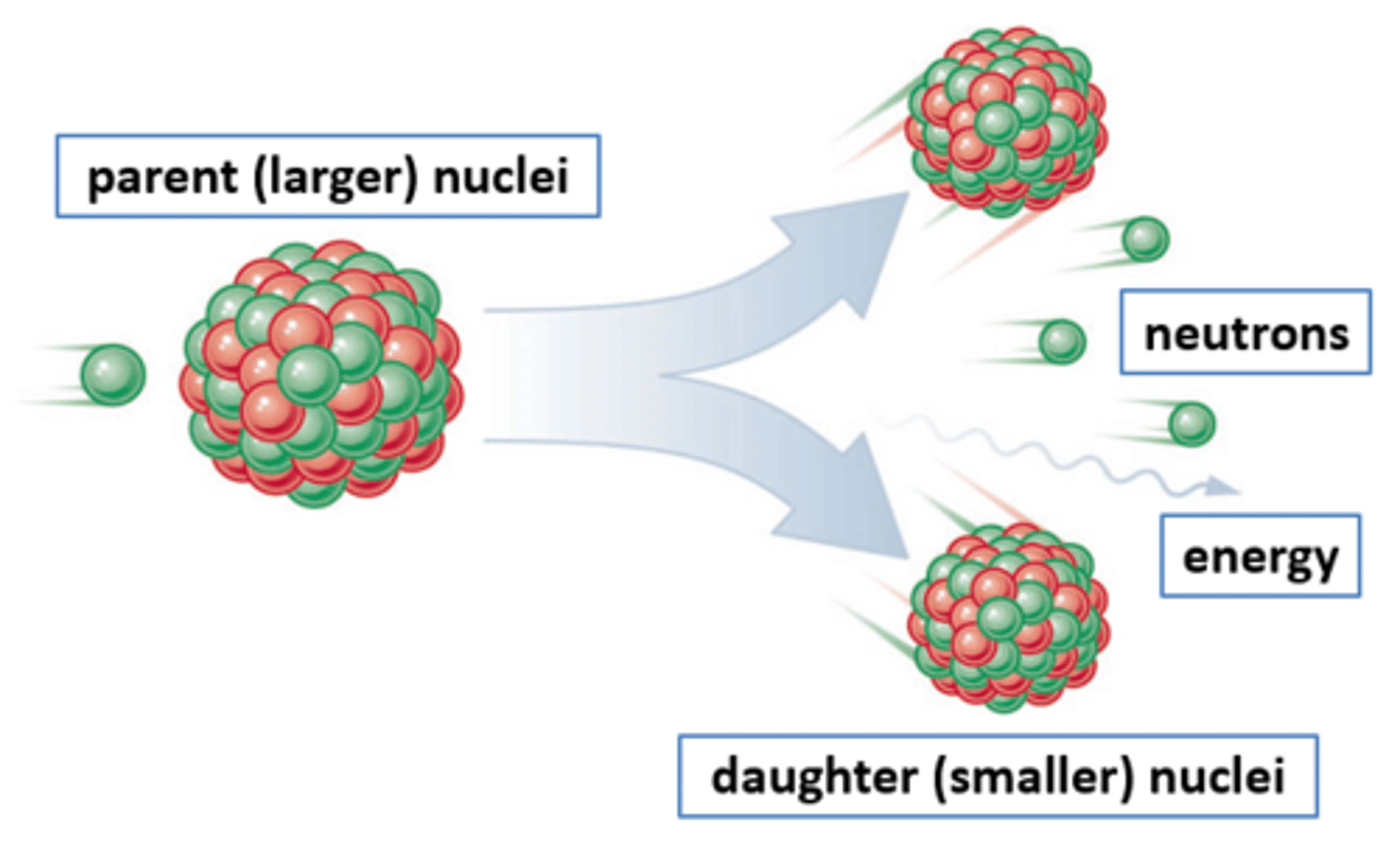
Daughter nuclei
The new nuclei that are produced when an unstable nucleus splits by fission
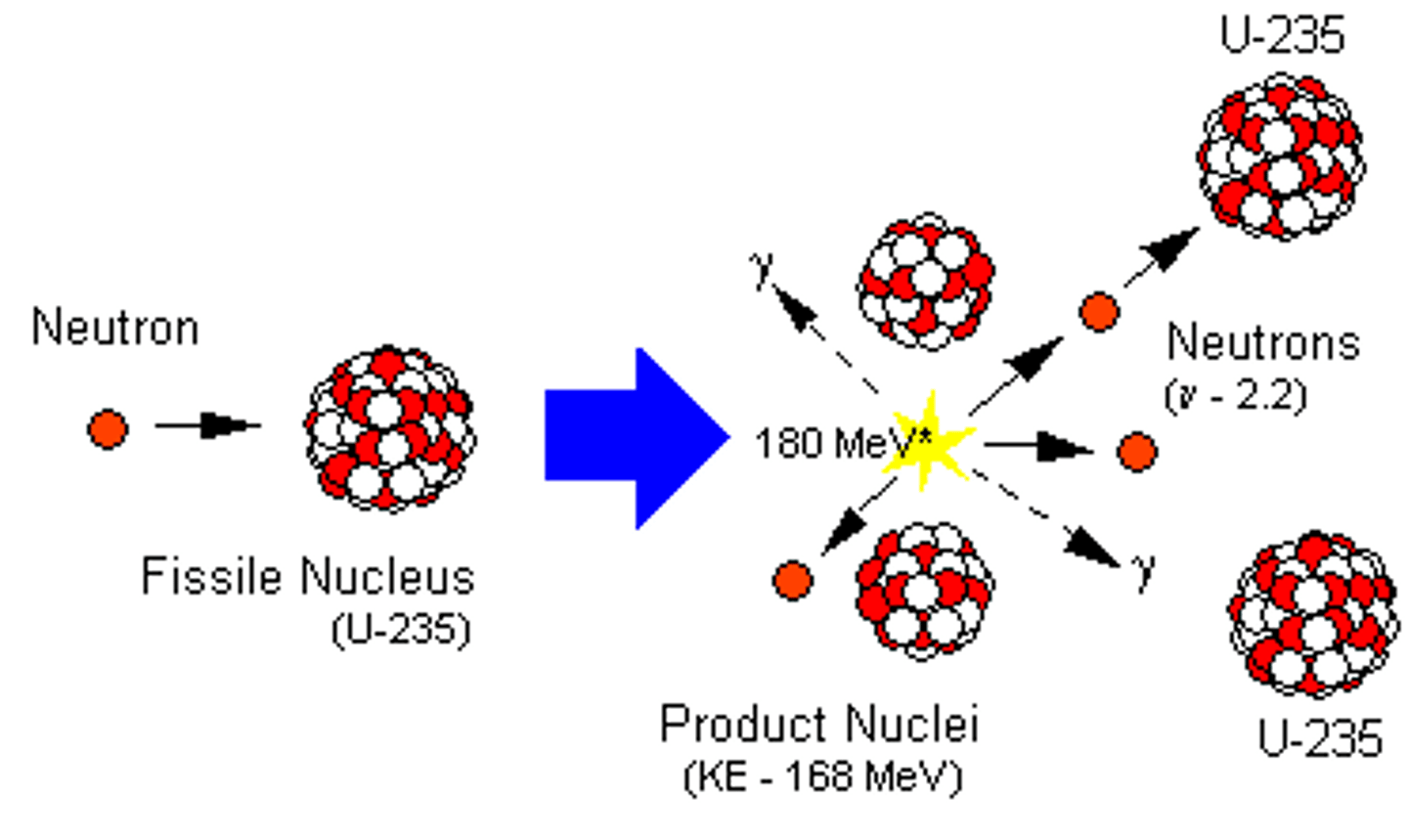
Chain reaction
When the neutrons released from a nucleus go on to cause the splitting of further nuclei
Controlled chain reaction
A chain reaction in which the rate of reaction is limited to prevent it from getting out of control
Uncontrolled chain reaction
A nuclear reaction which is not limited and eventually lead to an explosion
Nuclear reactors
Found in nuclear power stations and powered by a controlled chain reaction

Nuclear weapon
Uses an uncontrolled chain reaction to cause an explosion

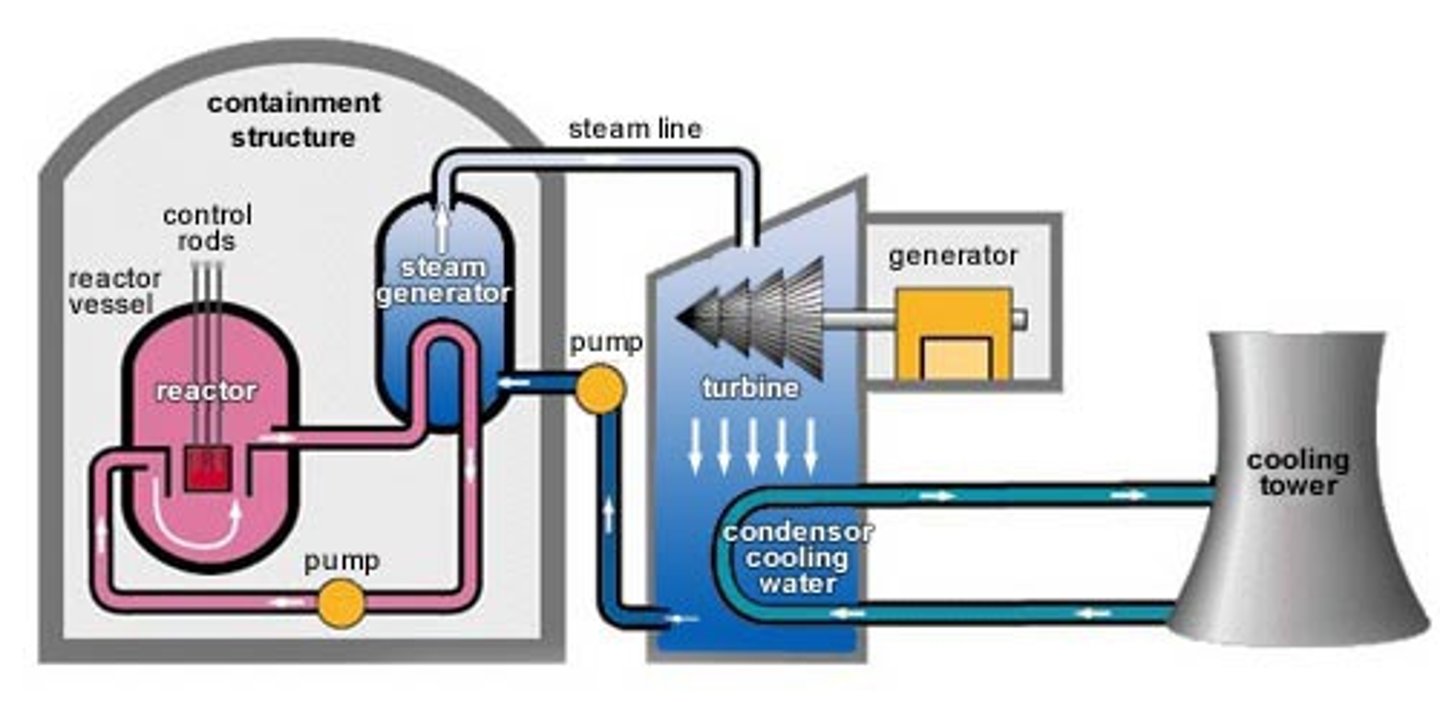
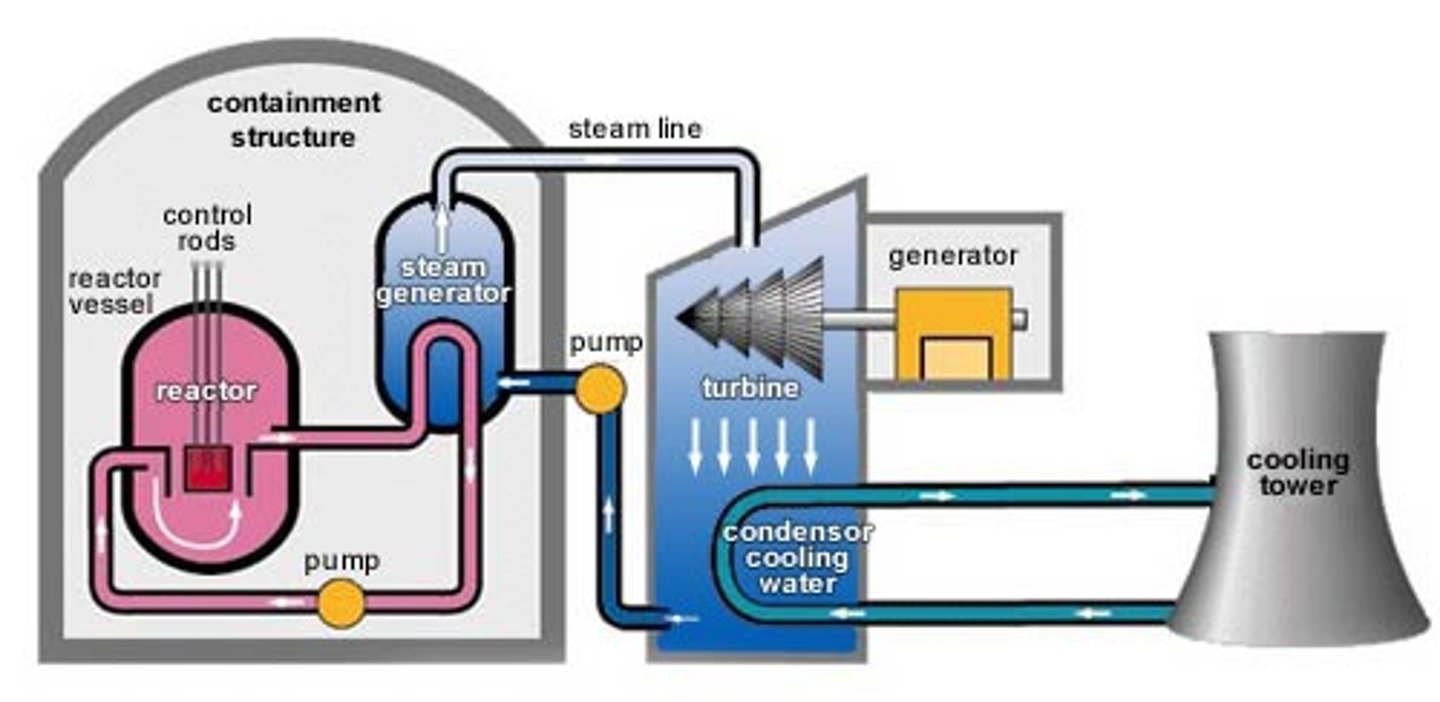
Nuclear reactor
A piece of equipment in a nuclear power station in which nuclear fission or fusion takes place

Nuclear fission reactor
Used to power commercial nuclear power stations
Nuclear fuel
The uranium (or plutonium) isotopes that undergo nuclear fission

Fuel rods
Rods in the nuclear reactor that contain the fuel so that the neutrons released from fission will travel and cause fission in other rods
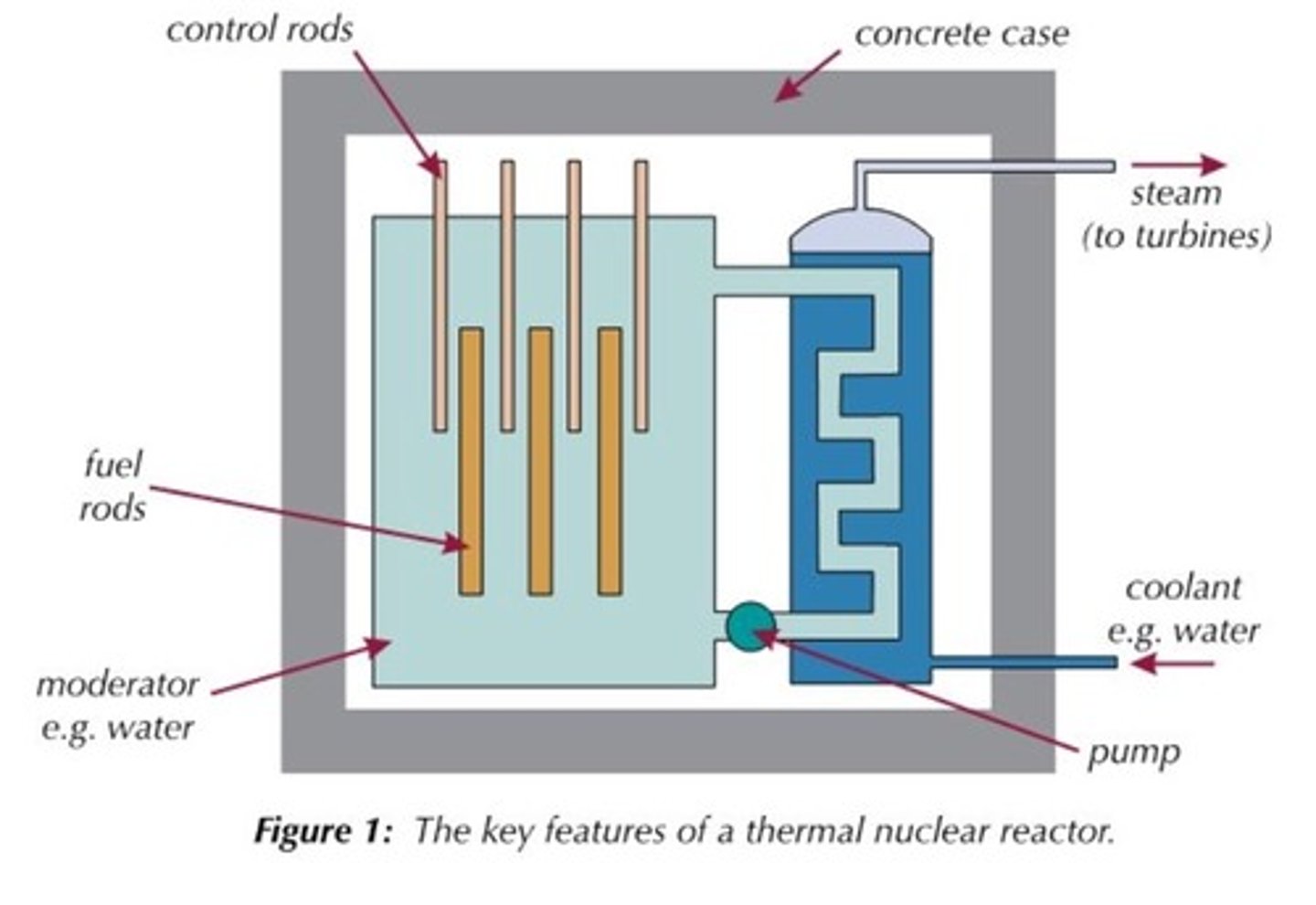
Moderator
Surrounds the fuel rods and slows the neutrons so that they are more readily absorbed by a nearby fuel rod
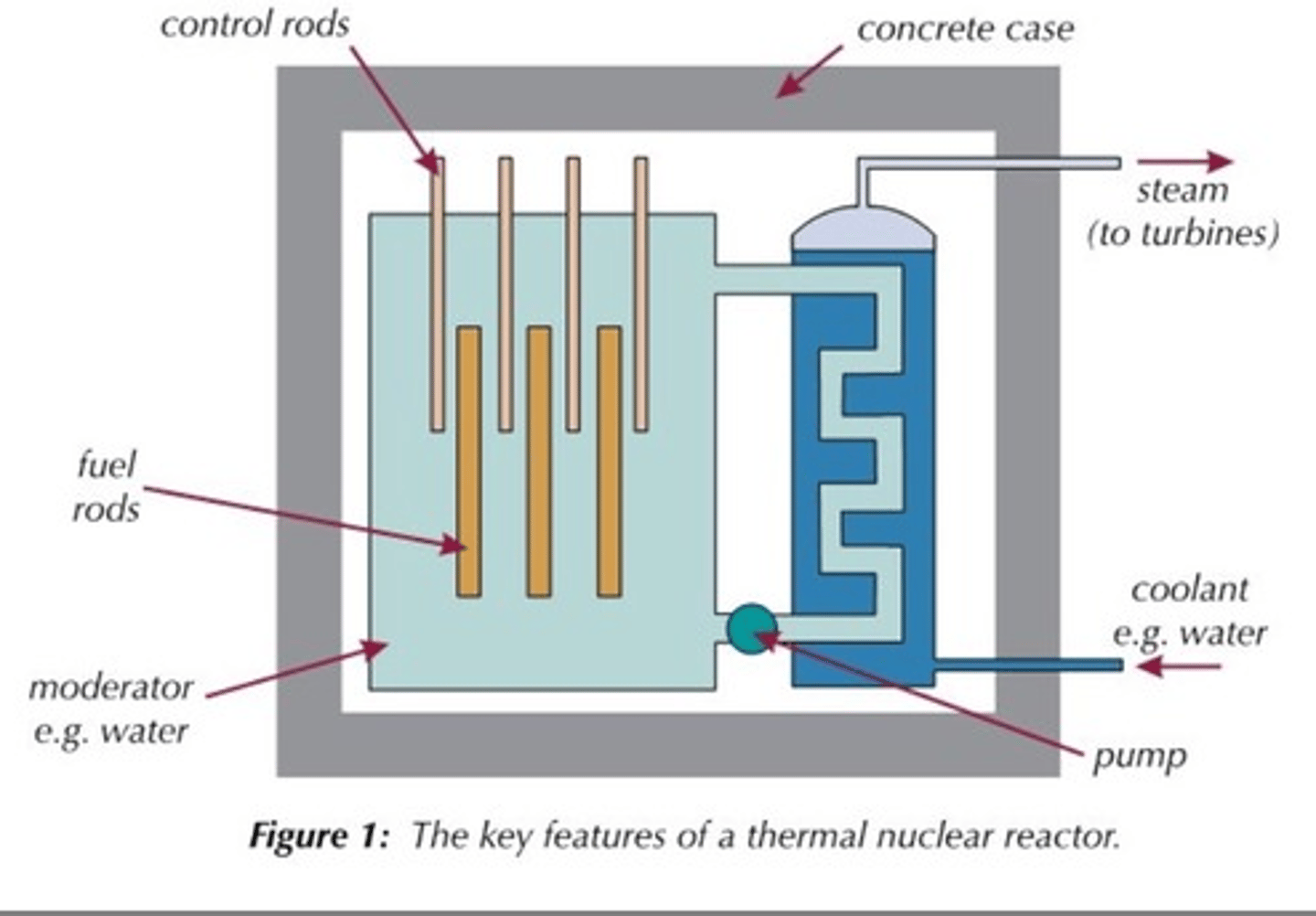
Material used for the moderator
Usually graphite
Chain reaction
When the neutrons released from a nucleus go on to cause the splitting of further nuclei
Control rods
Rods that stop neutrons from travelling between fuel rods by absorbing them in order to control the rate of the chain reaction
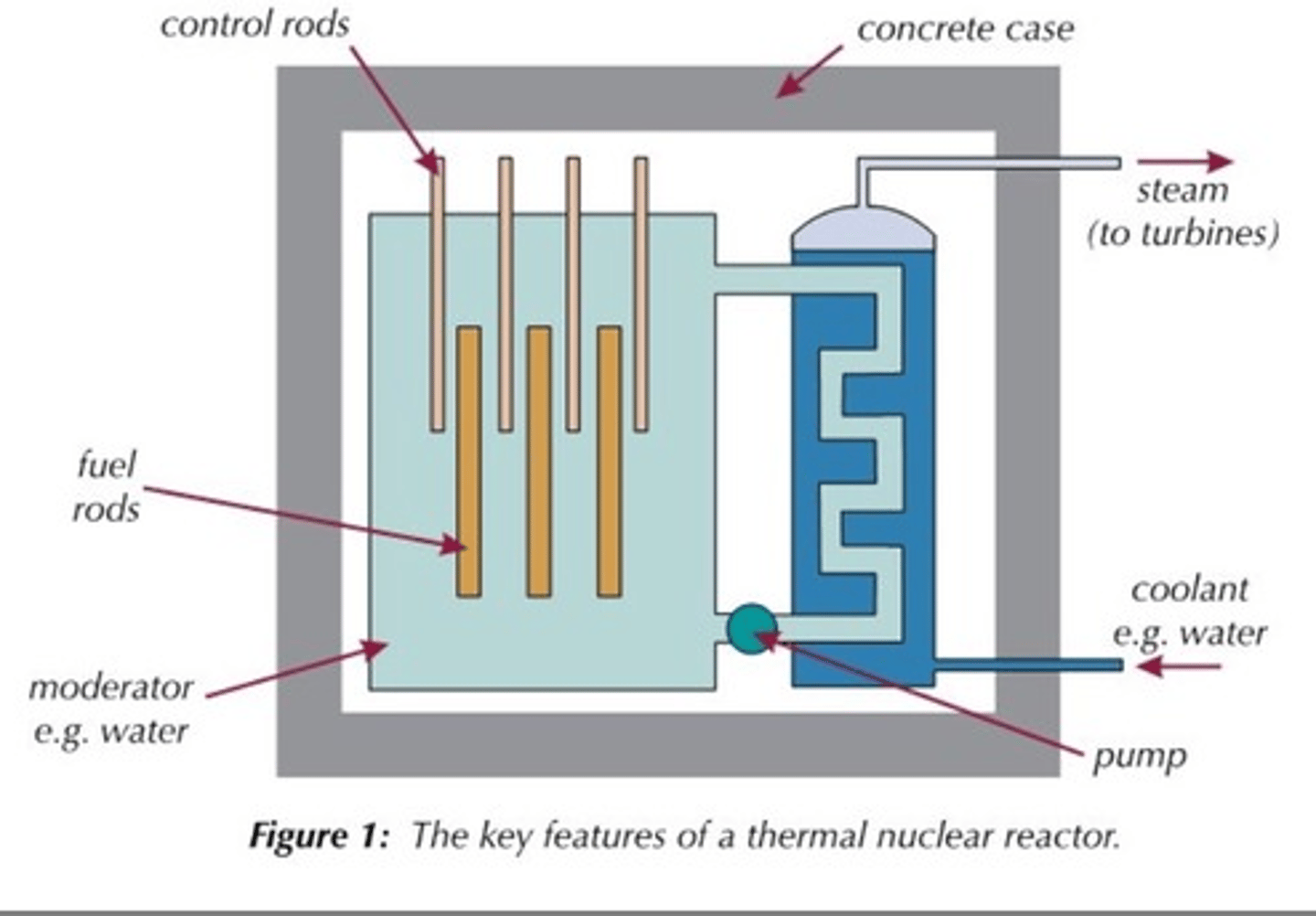
Materials used for control rods
Usually boron or cadmium
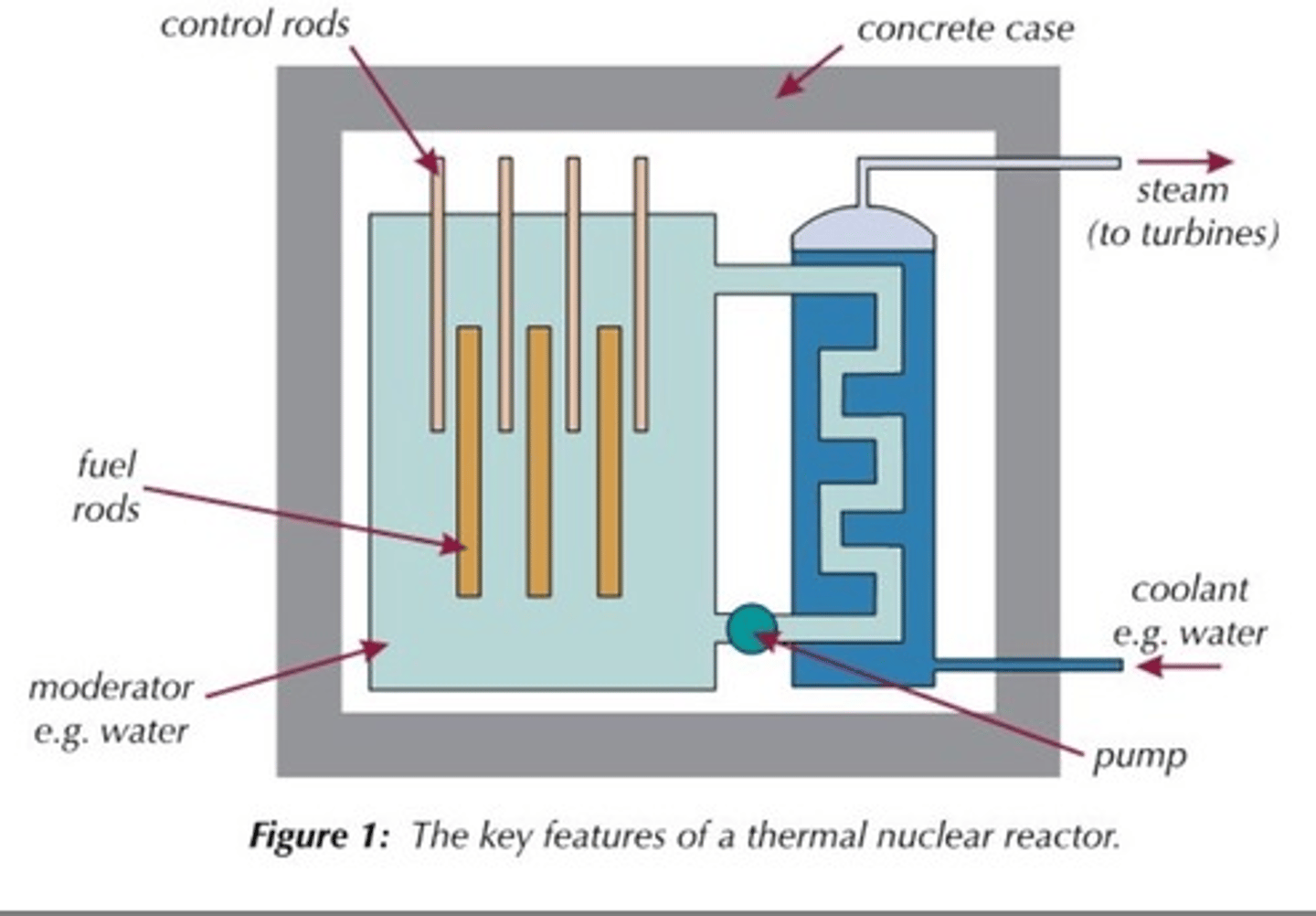
Concrete shielding
Used to contain the products of the fission reaction as they are radioactive and can be hazardous
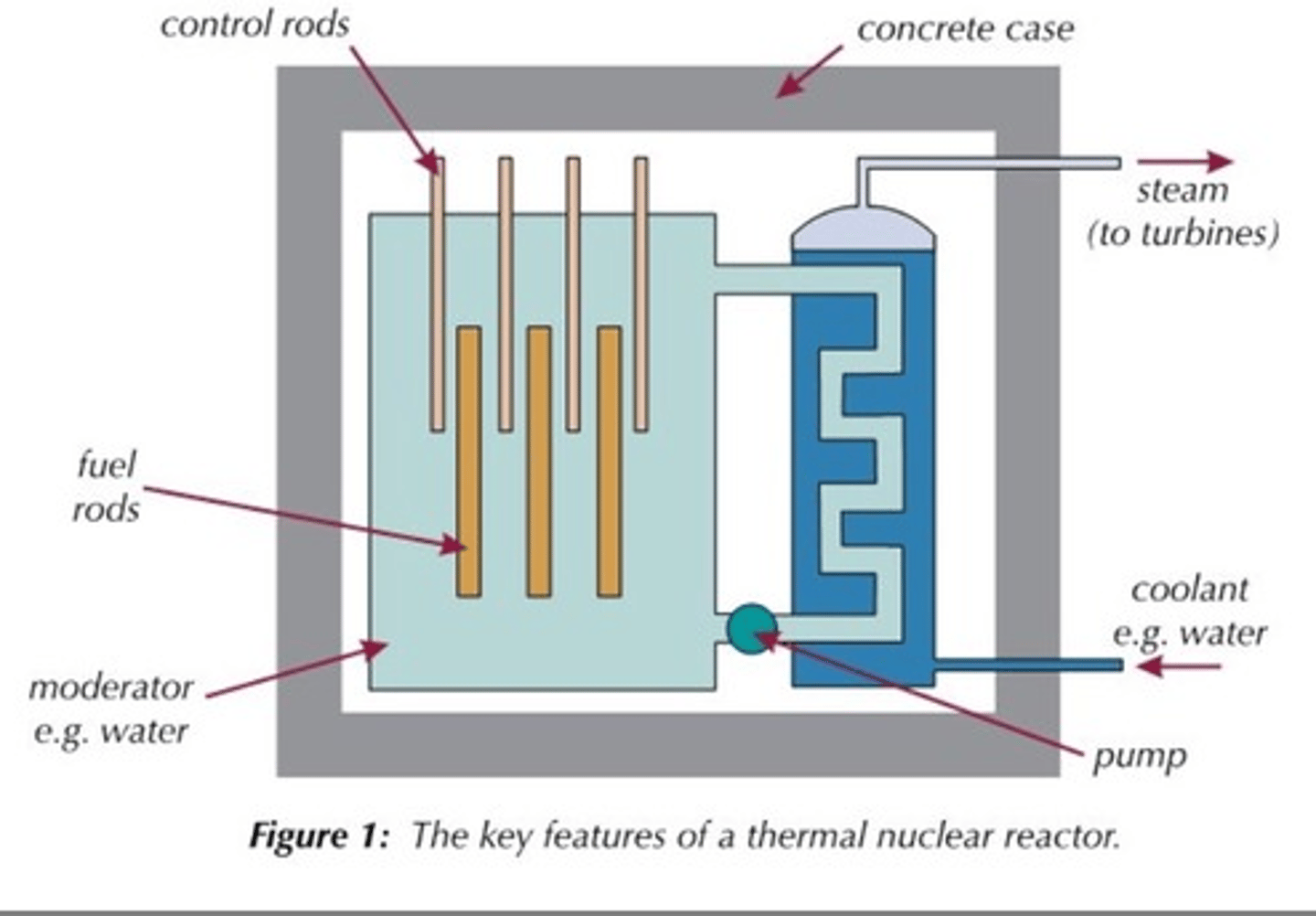
Coolant
heated up by the energy released from the fission reactions and then used to boil water to turn it into steam
Steam
Used to drive the turbines in a power station
Turbine
a device with blades that can be turned by water, wind, steam and is used to turn the generator in a power station
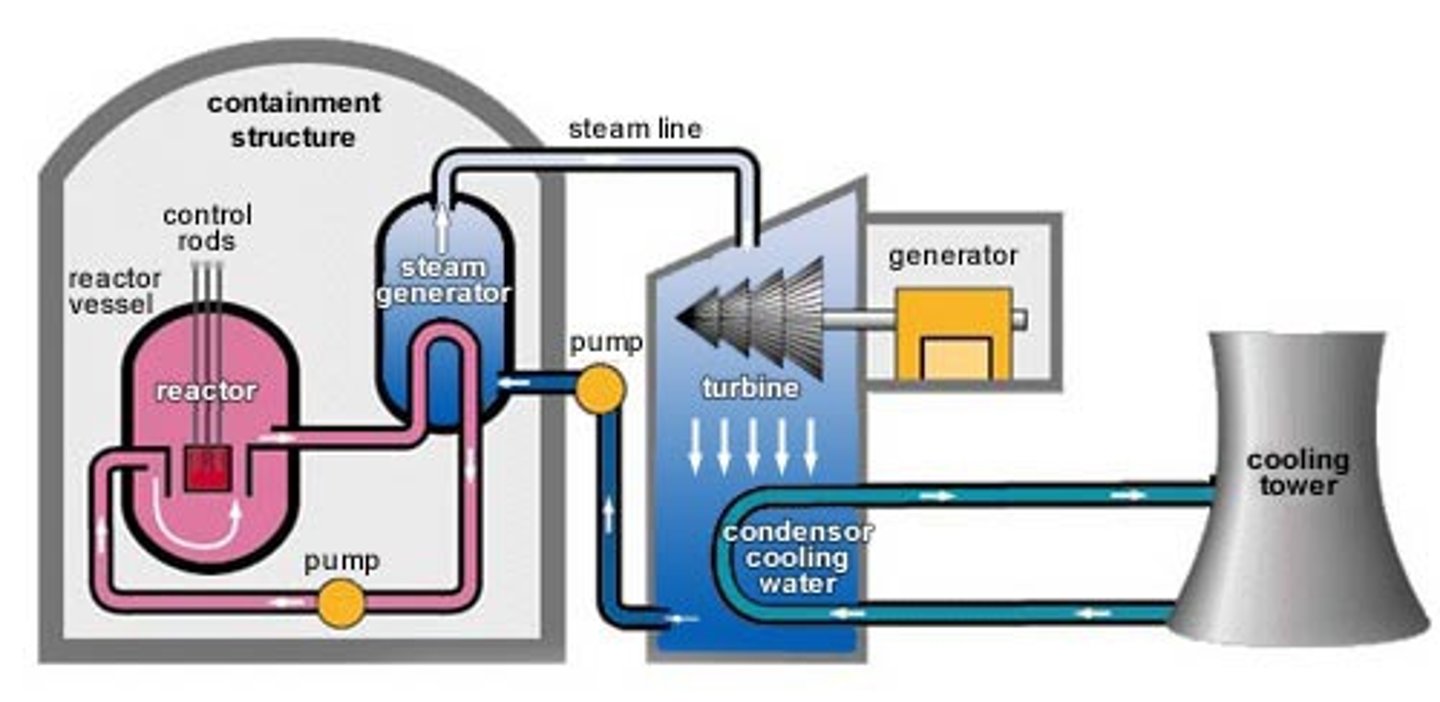
Generator
An electromagnetic device that produces electricity in a power station when it turned by the turbines
Nuclear fusion
The joining of two light nuclei to form a heavier nucleus
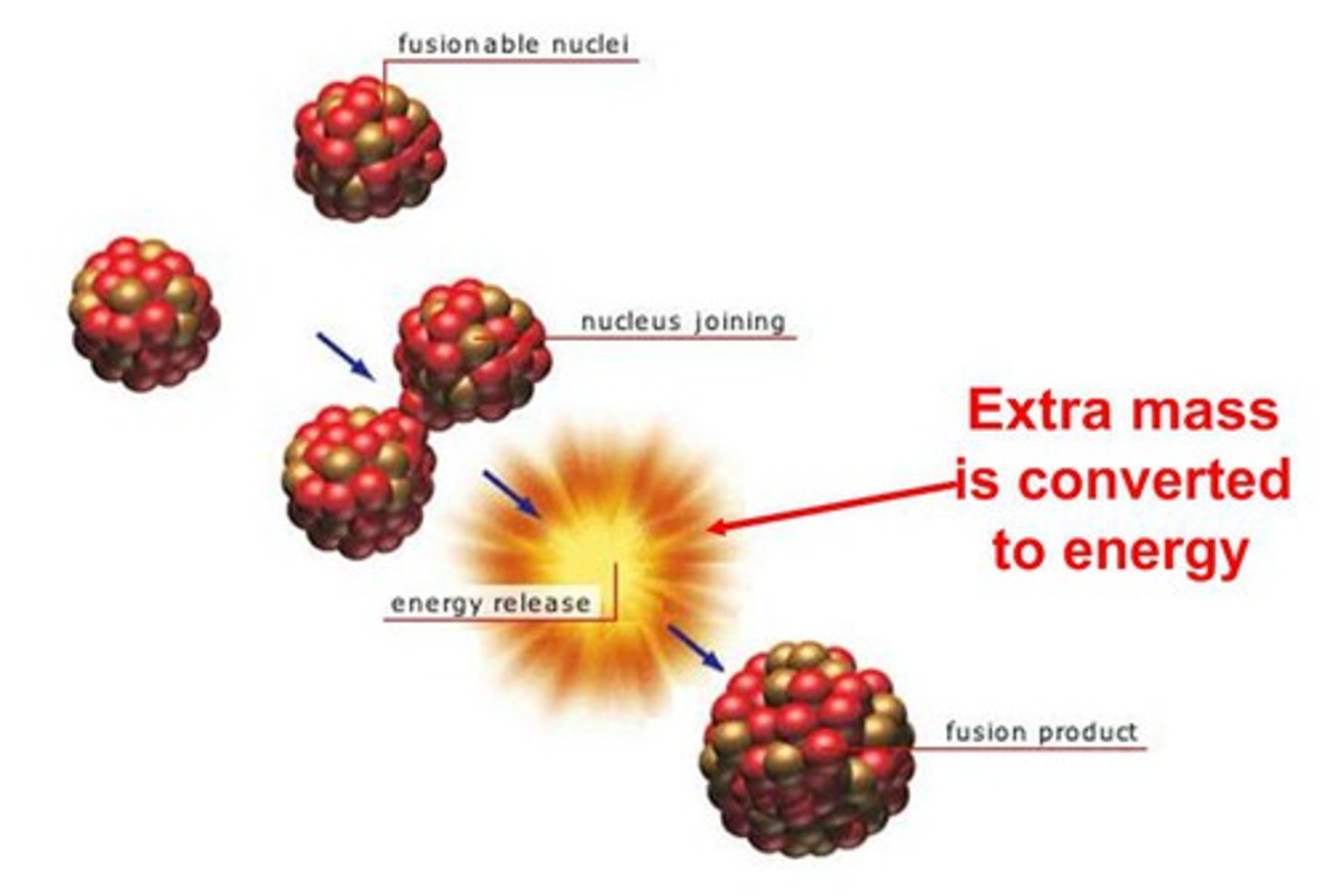
Fusion products
A heavier nucleus and energy in the form of gamma rays
Spontaneous fusion
Occurs in stars when two hydrogen nuclei fuse together to form a nucleus of helium

Conditions required for fusion
Very high temperatures and pressure
Mass-energy conversion
When some of the mass of the fusing particles is converted into the energy of the emitted radiation
repulsion
it requires the fusing of nuclei, which are positive particles; two positive charges repel each other
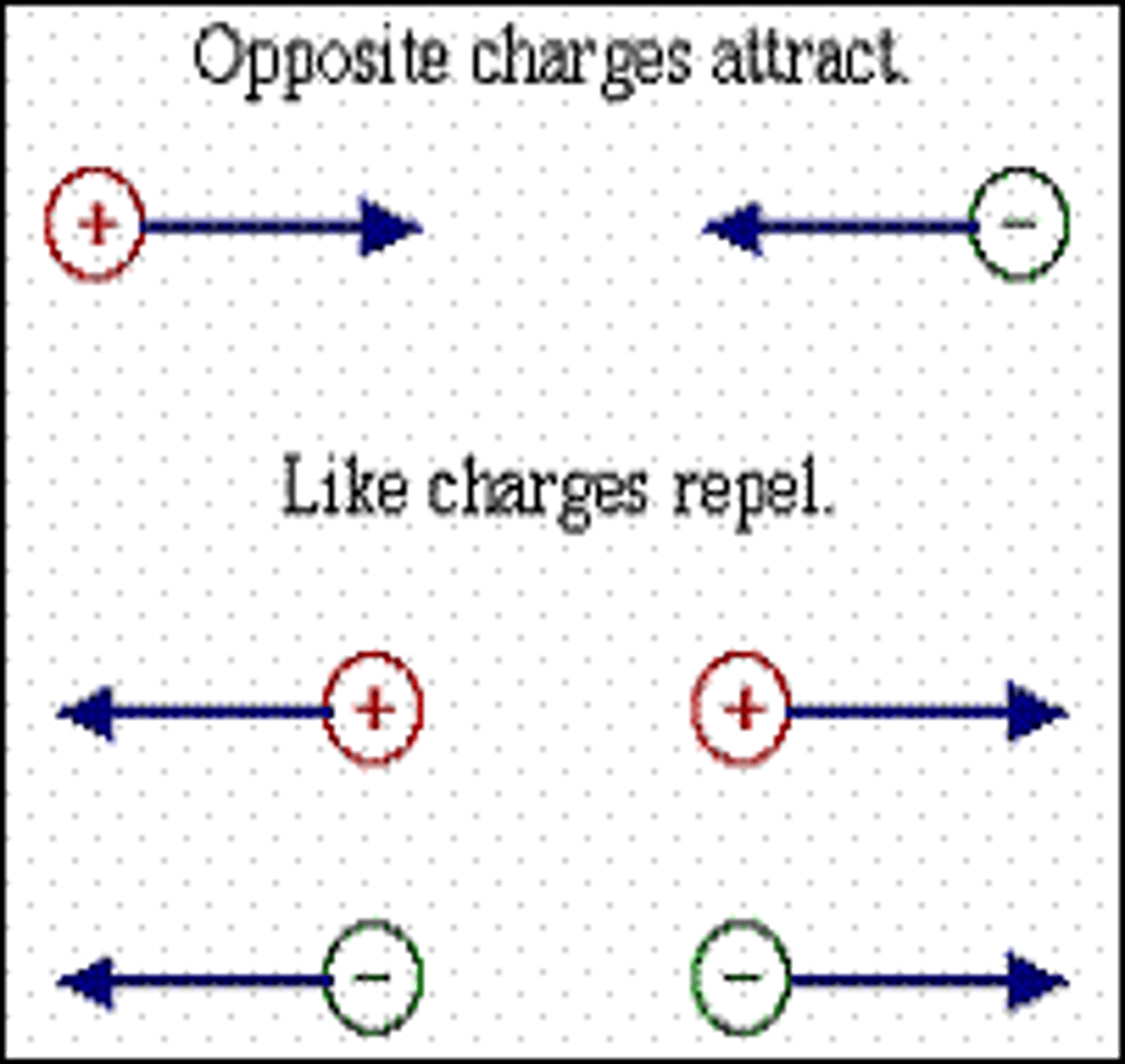
What happens when two nuclei approach each other
They repel each other because they have the same charge
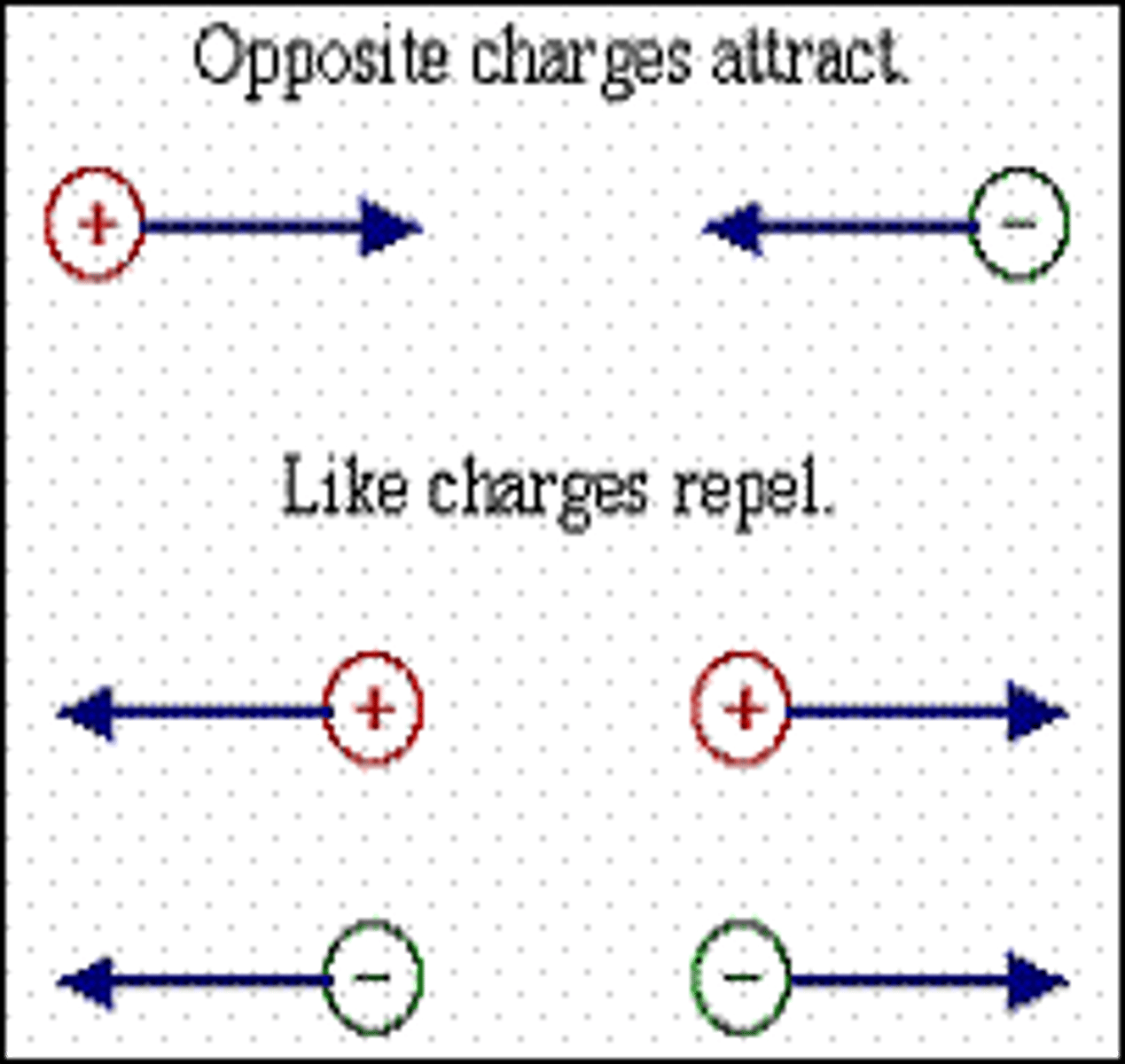
How to overcome nuclei repulsion
Fusion has to happen under intense pressure and very high temperatures in order to force the nuclei together
Advantages of fusion over fission
There are no radioactive waste products from fusion
Disadvantages of fusion over fission
The very high temperatures and pressures required for fusion makes it very difficult to build a viable power station
Stable nuclei
Nuclei that contain a certain amount of neutrons compared to protons
Unstable nuclei
Nuclei with too few or too many neutrons
Radioactive decay
A random process in which an unstable nucleus will emit one or more types of nuclear radiation to become more stable
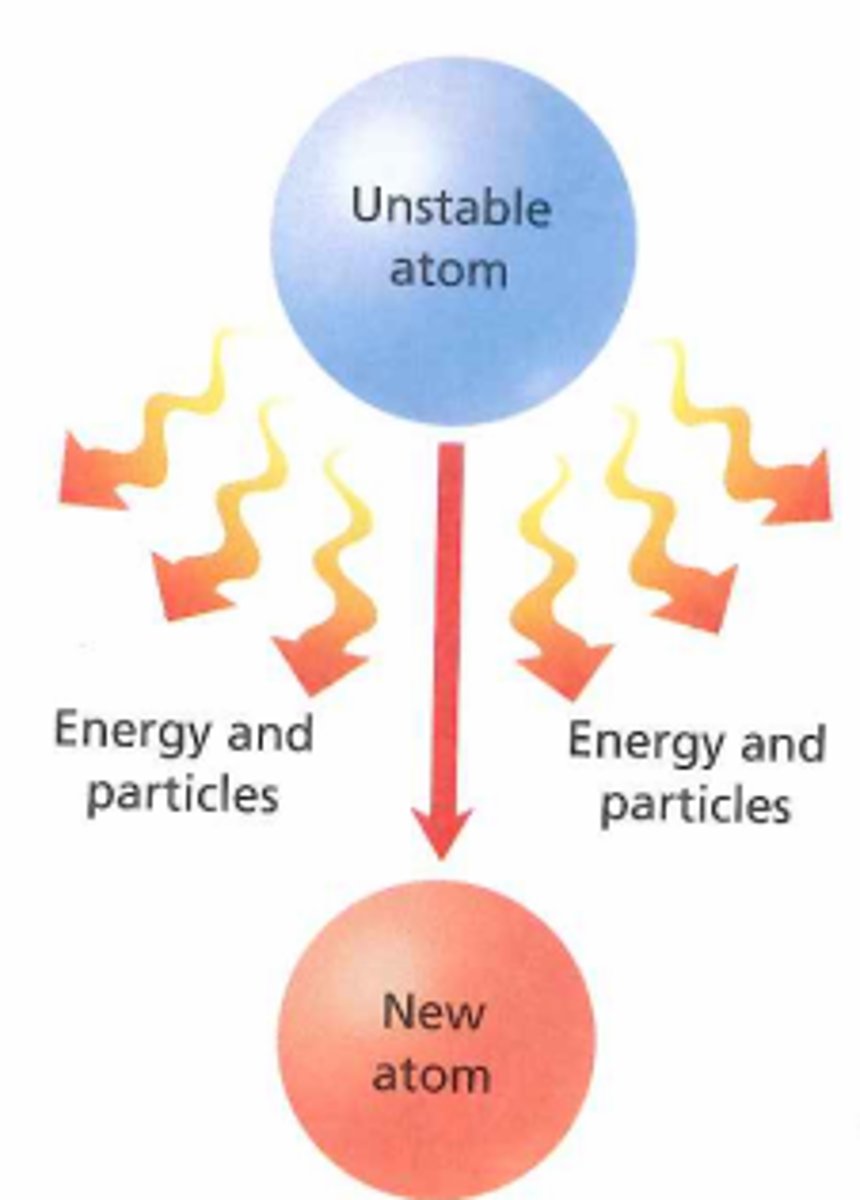
Nuclear radiation
Radiation emitted from an unstable nucleus during radioactive decay
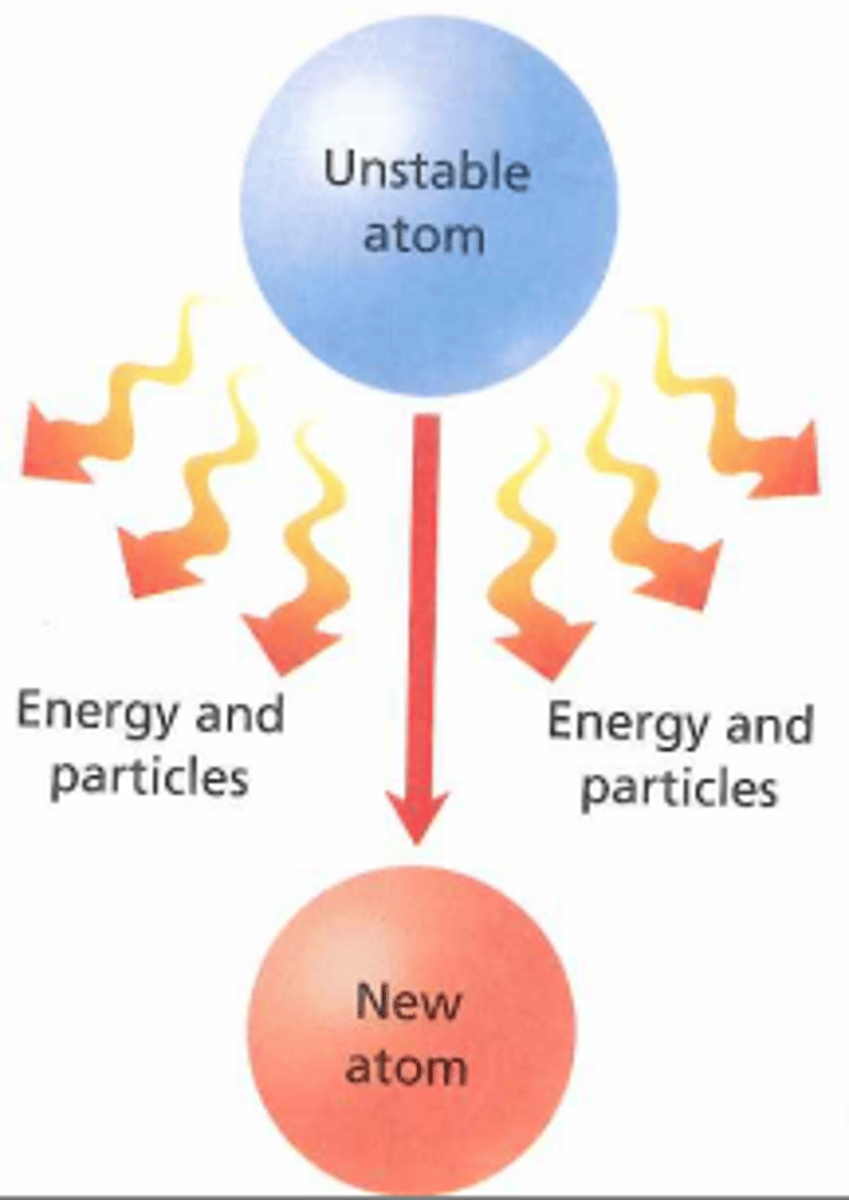
Types of nuclear radiation
Alpha particle, beta particle, gamma ray, neutron
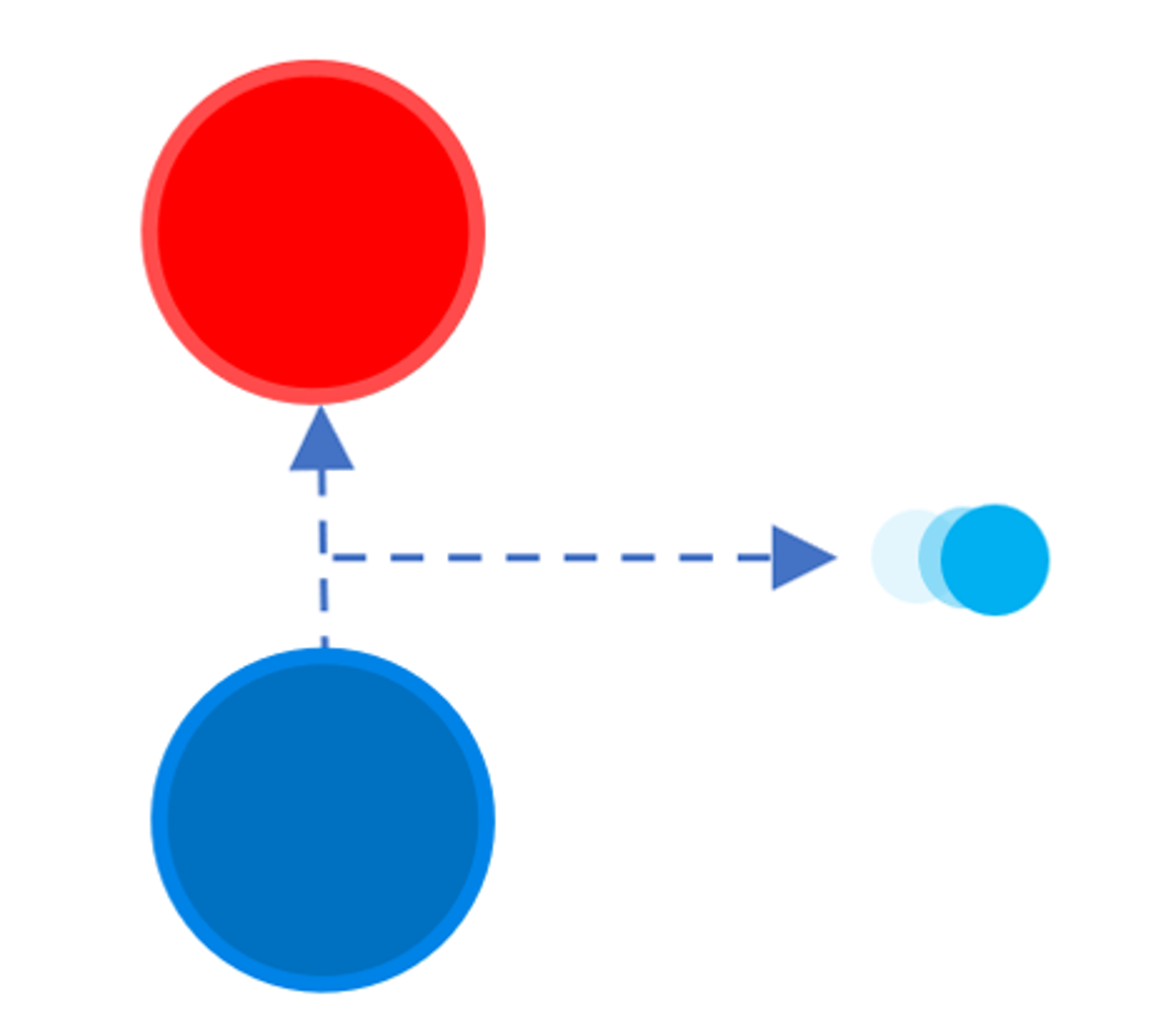
Alpha particle (α)
Consists of two neutrons and two protons and is identical to a helium nucleus

Beta particle (β)
A high speed electron ejected from the nucleus
Gamma ray (γ)
Electromagnetic radiation from the nucleus
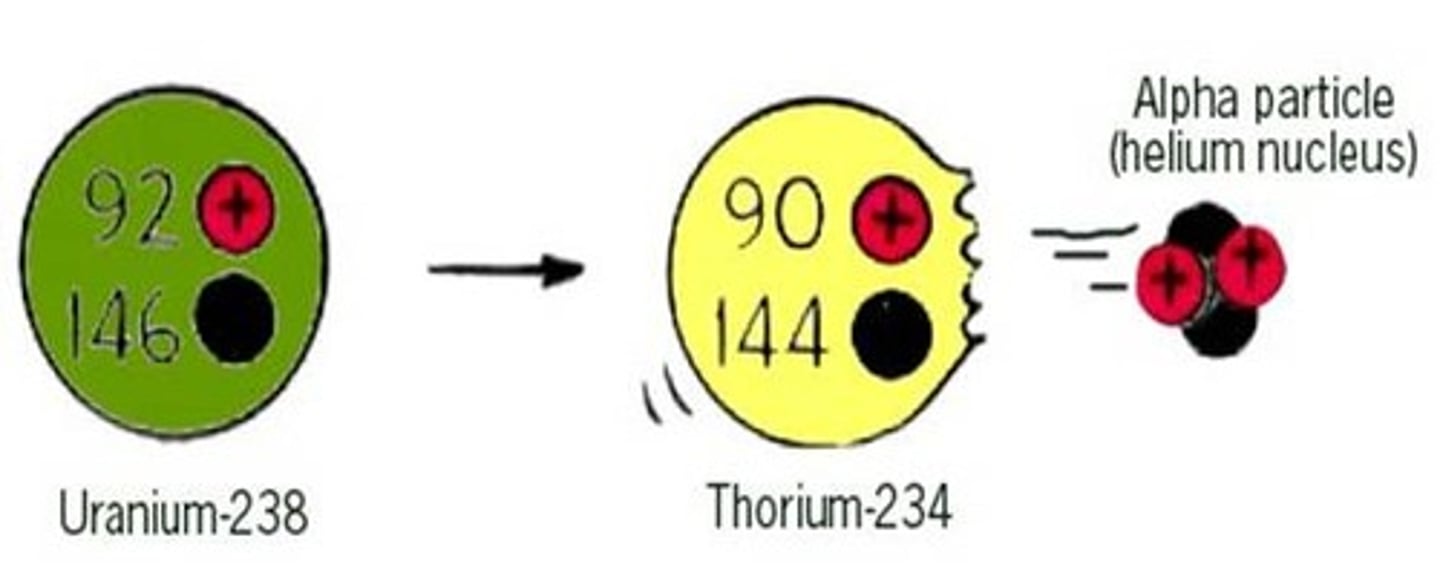
Change to the nucleus due to alpha decay
The nucleus loses two protons and two neutrons
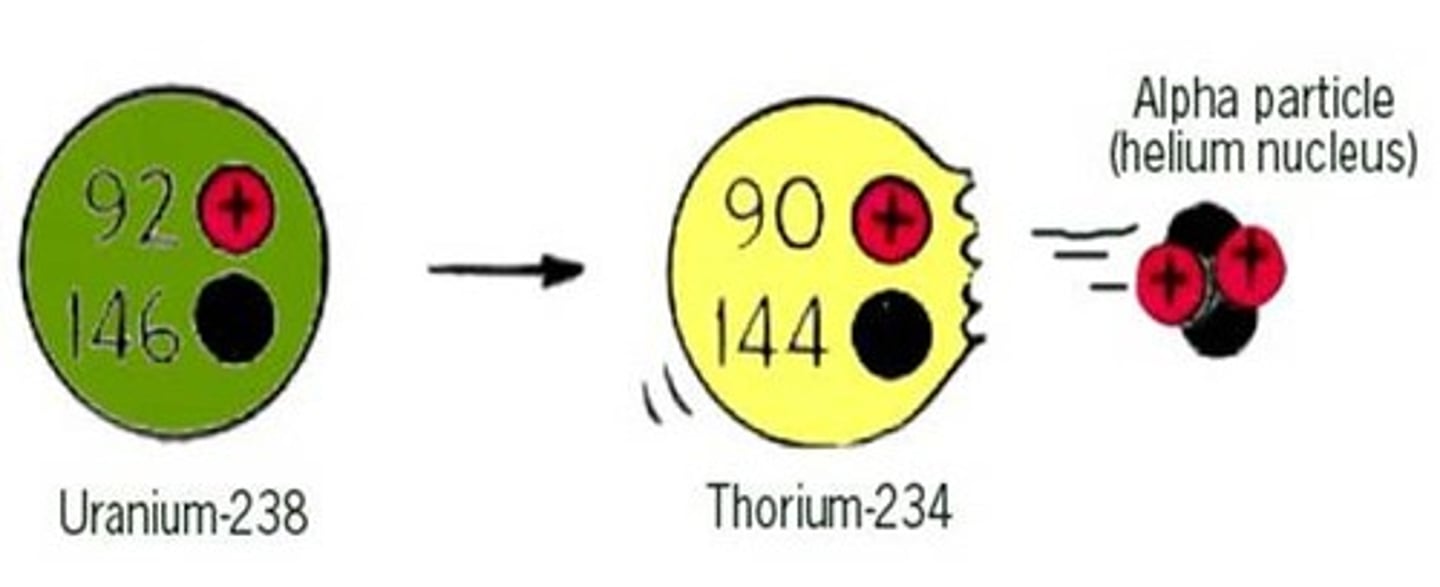
Effect of alpha decay on atomic and mass numbers
The mass number decreases by four and the atomic number decreases by two
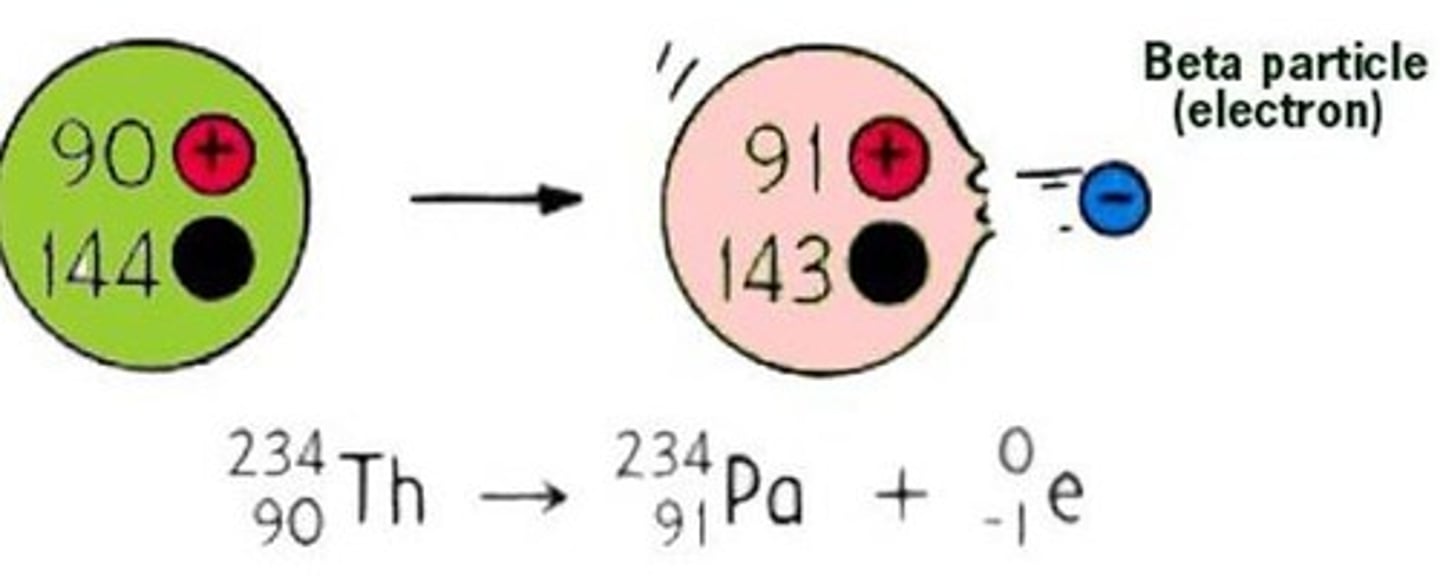
Change to the nucleus due to beta decay
A neutron turns into a proton
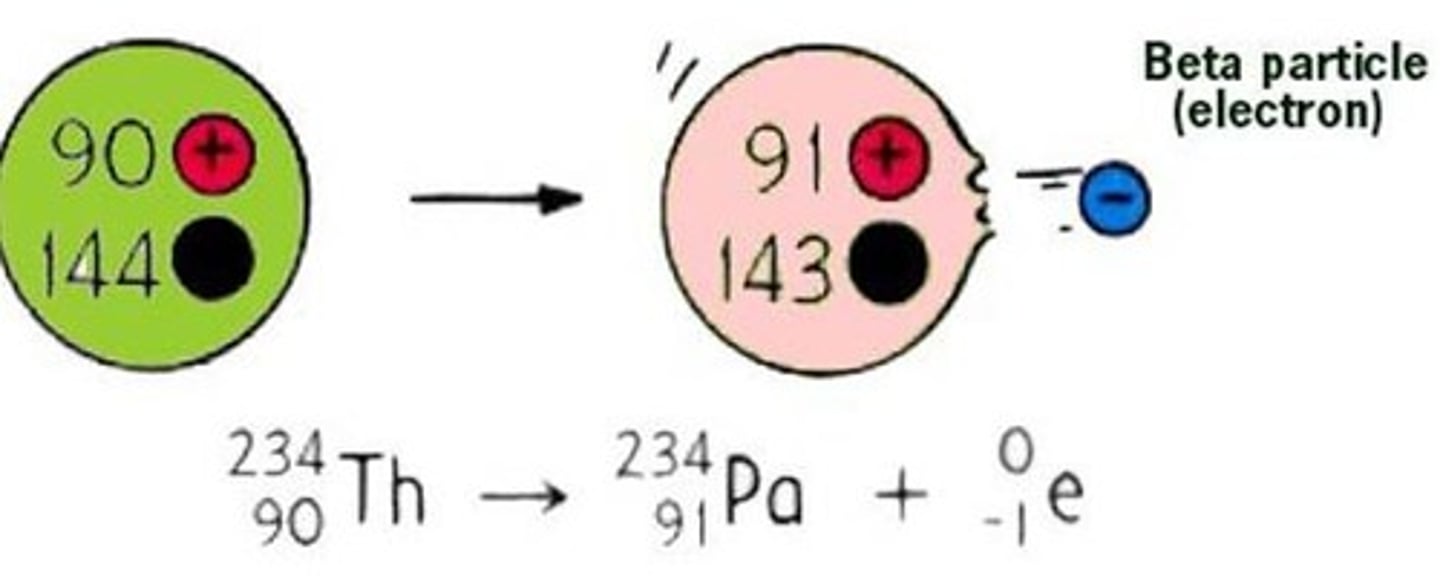
Effect of beta decay on atomic and mass numbers
The atomic number increases by one and the mass number remains the same
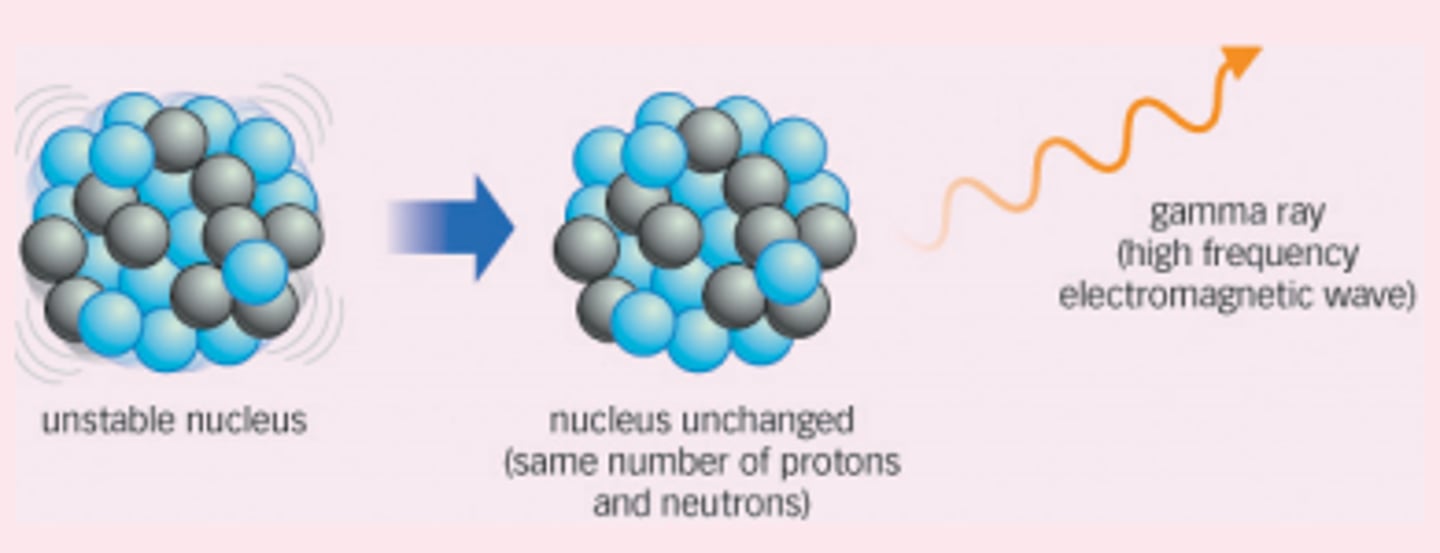
Change to the nucleus due to gamma decay
There is no change to the particles in the nucleus
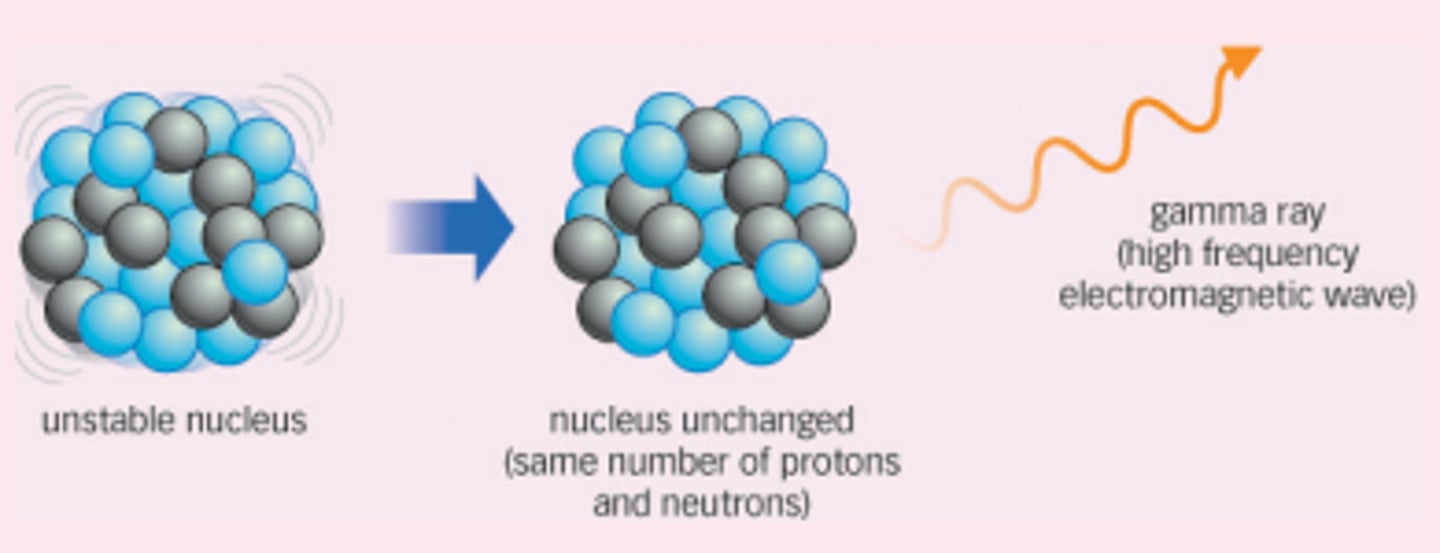
Effect of gamma decay on atomic and mass numbers
There is no change to the atomic or mass numbers
Geiger-Muller (GM) tube
A device that can detect ionising radiation and is used to measure the activity of a radioactive source
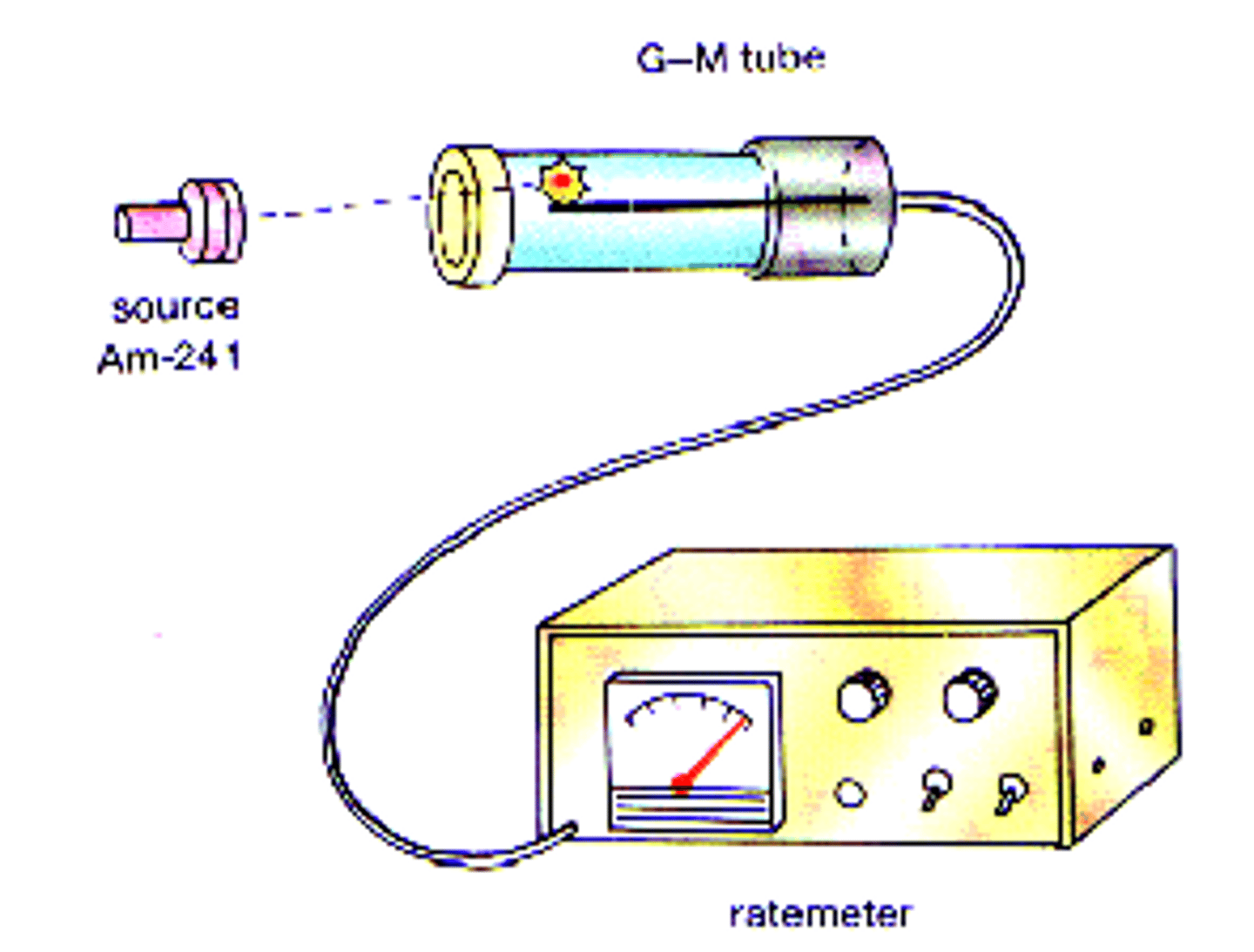
activity
the rate at which a source of unstable nuclei decays
becquerels (Bq)
the SI unit for activity
count-rate
the number of decays recorded each second by a detector
Relative mass and charge of a alpha particle
Mass = 4 and charge = +2
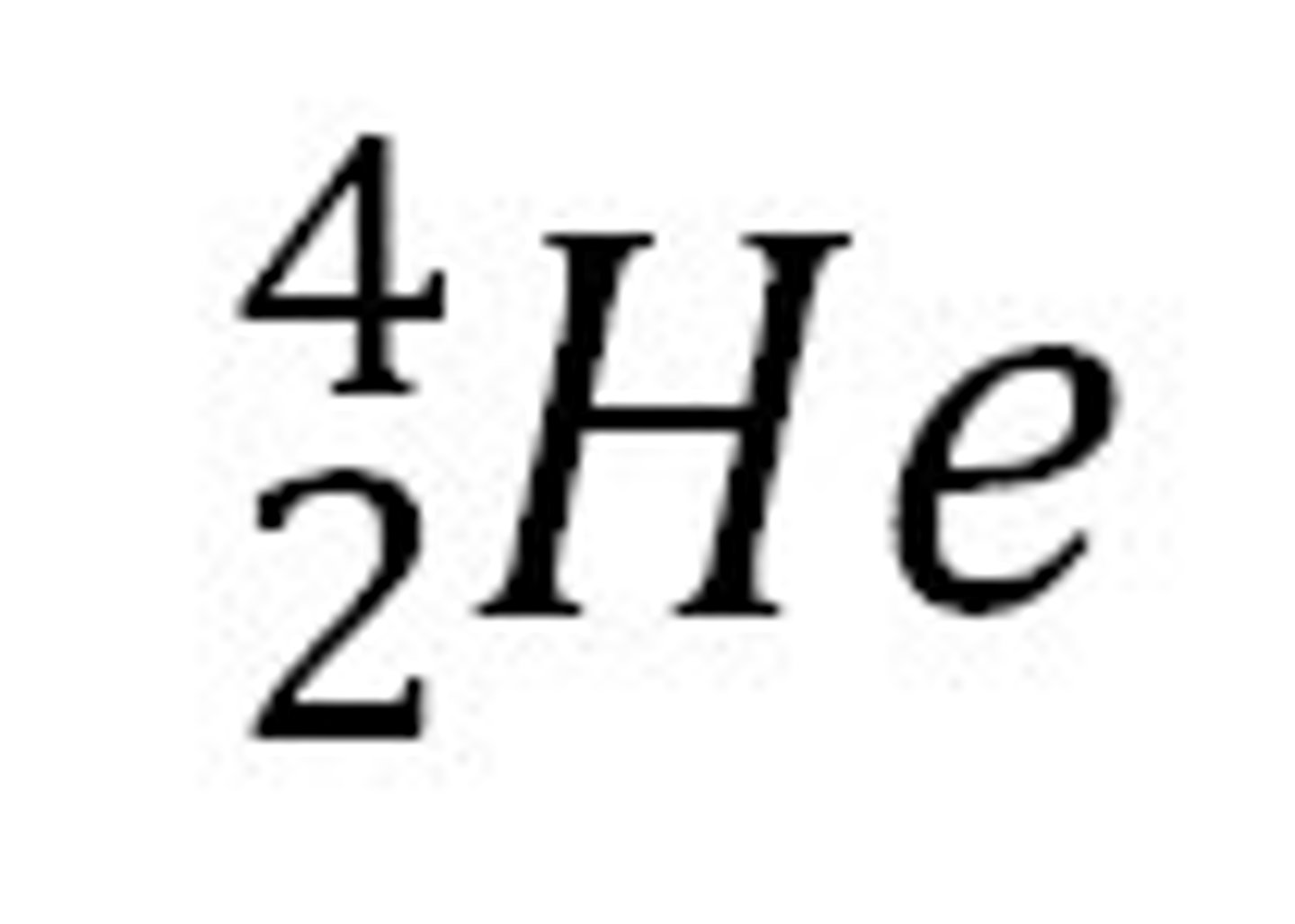
Relative mass and charge of a beta particle
Mass = approx. 1/2000 and charge = –1

Relative mass and charge of a gamma ray
Mass = 0 and charge = 0
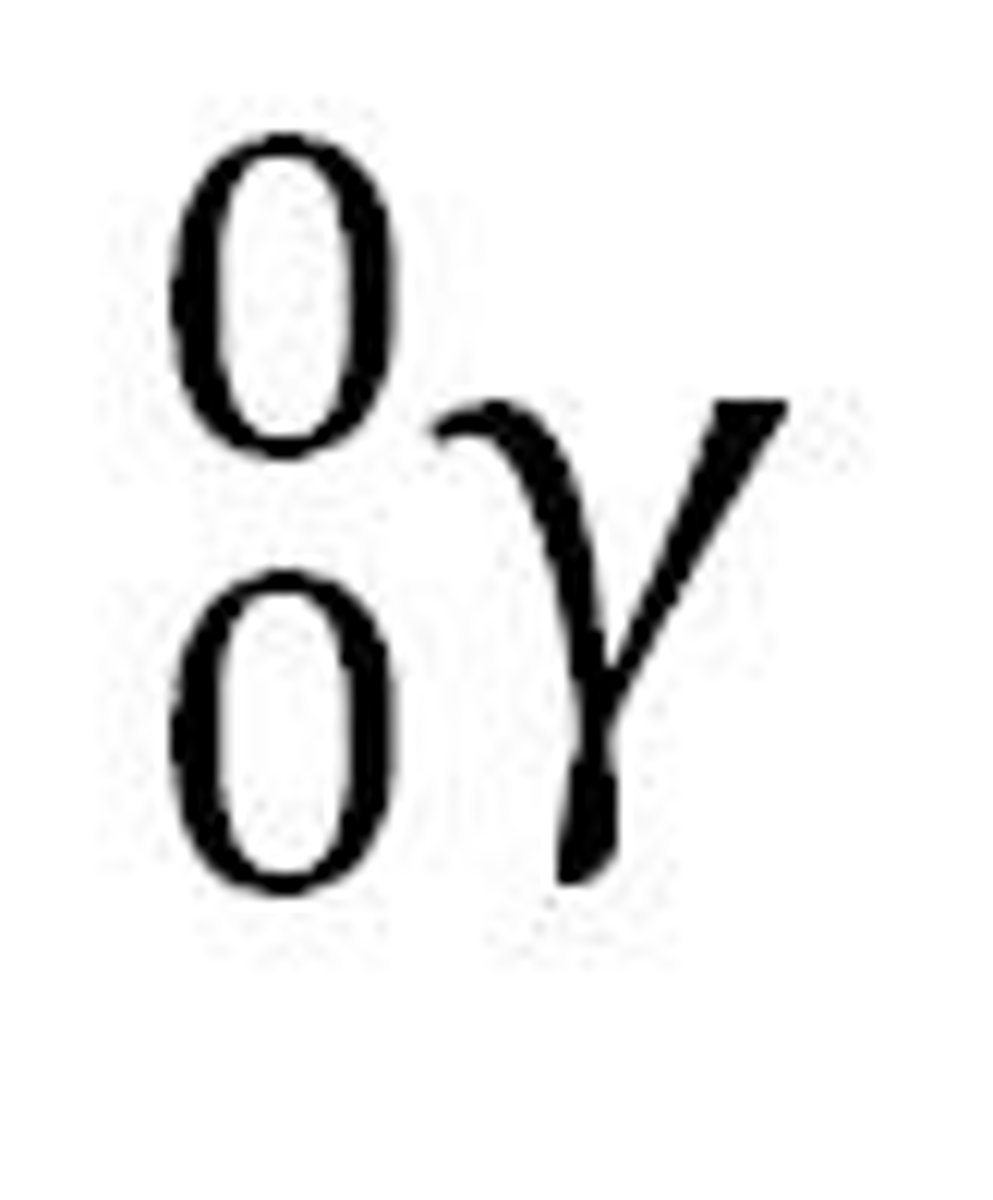
Ionising power
The ability to ionise materials
Effect of charge on ionising power
The greater the charge of the radiation, the more it will ionise materials
Ionising power of an alpha particle
Highly ionising
Ionising power of a beta particle
Moderately ionising
Ionising power of a gamma ray
Weakly ionising
Penetrating power
The ability to penetrate (pass through) matter
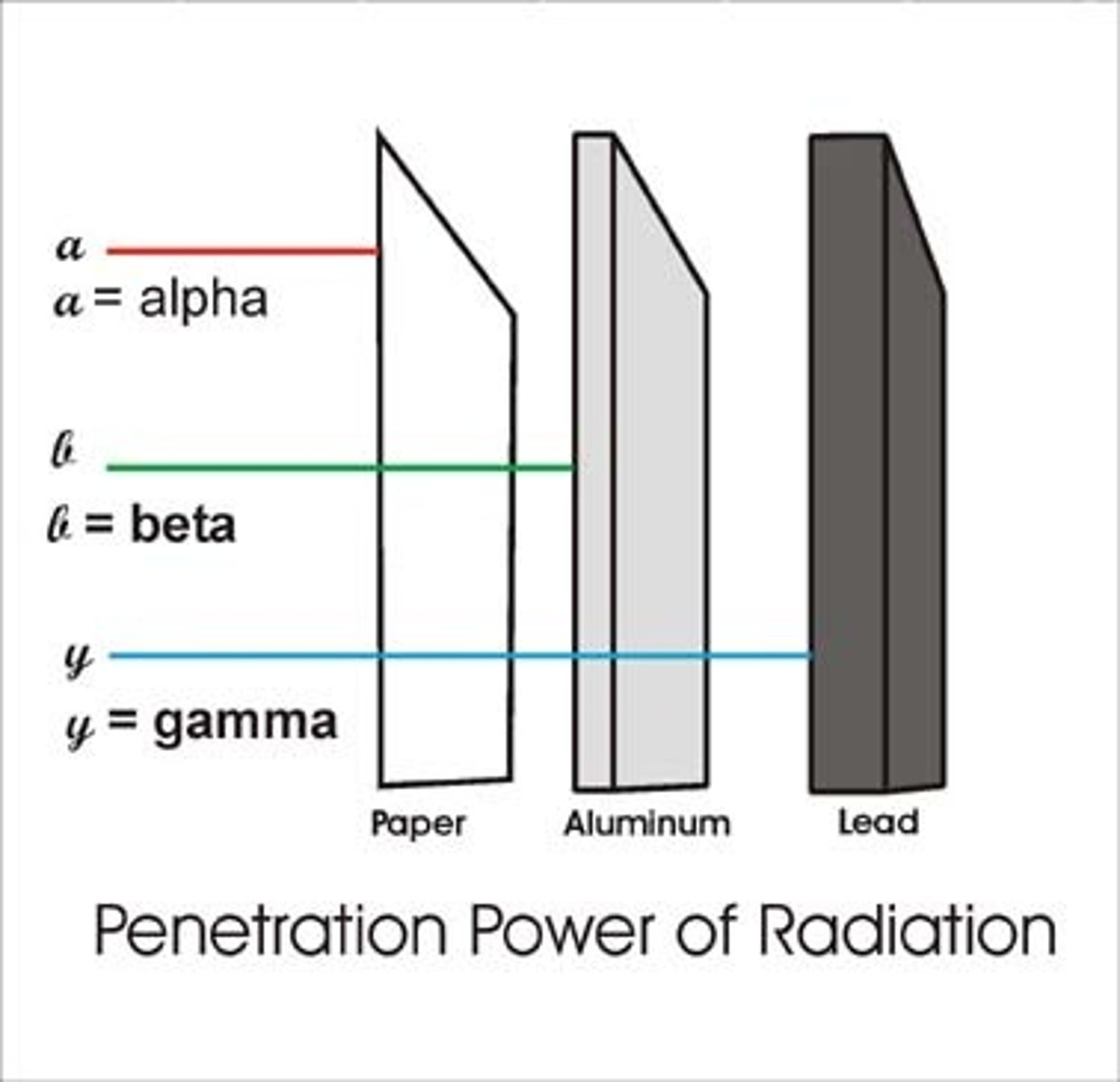
Effect of mass on penetrating power
The greater the mass of the radiation, the less it will penetrate materials
Penetrating power of an alpha particle
Weakly penetrating
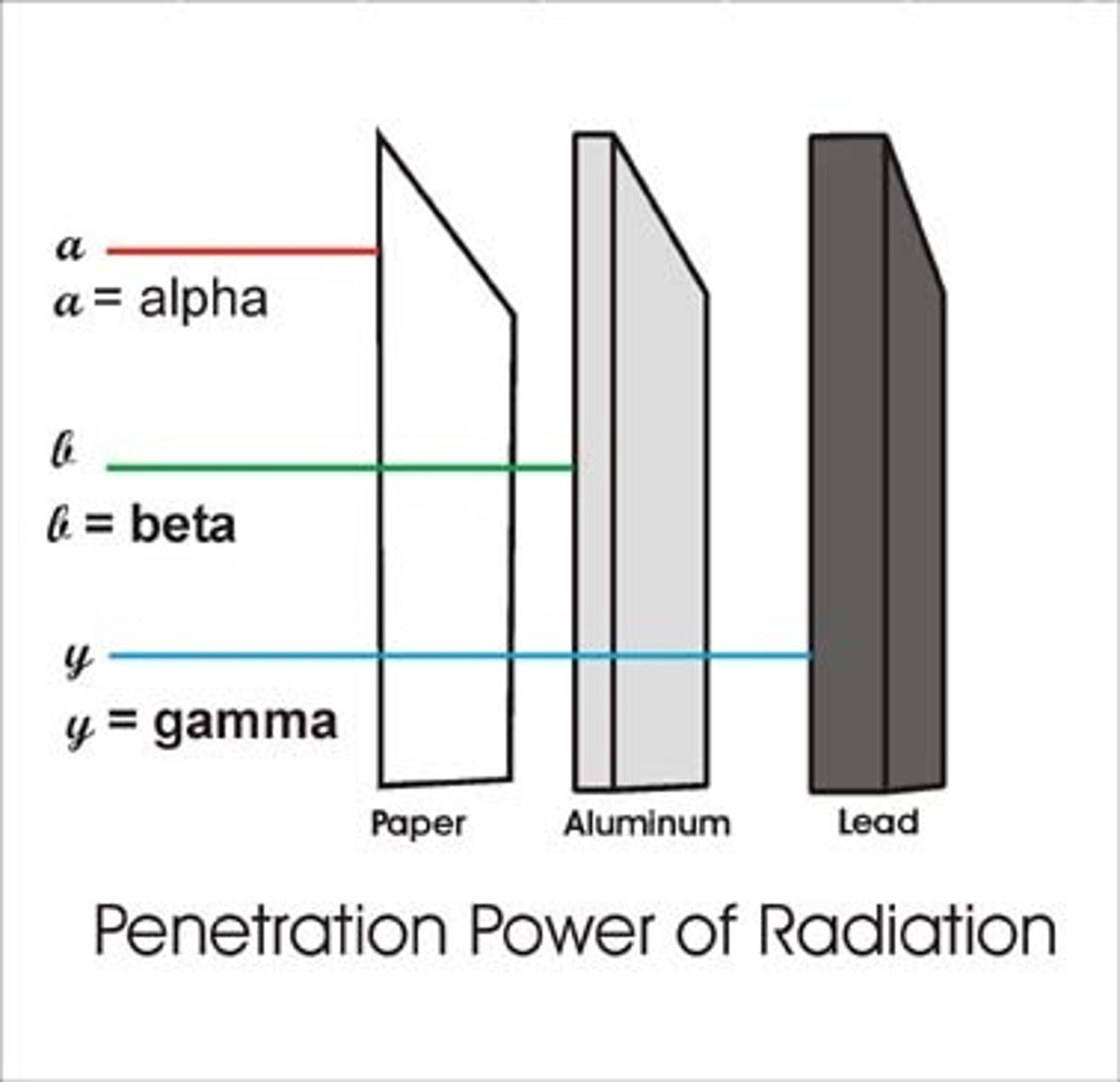
Penetrating power of a beta particle
Moderately penetrating
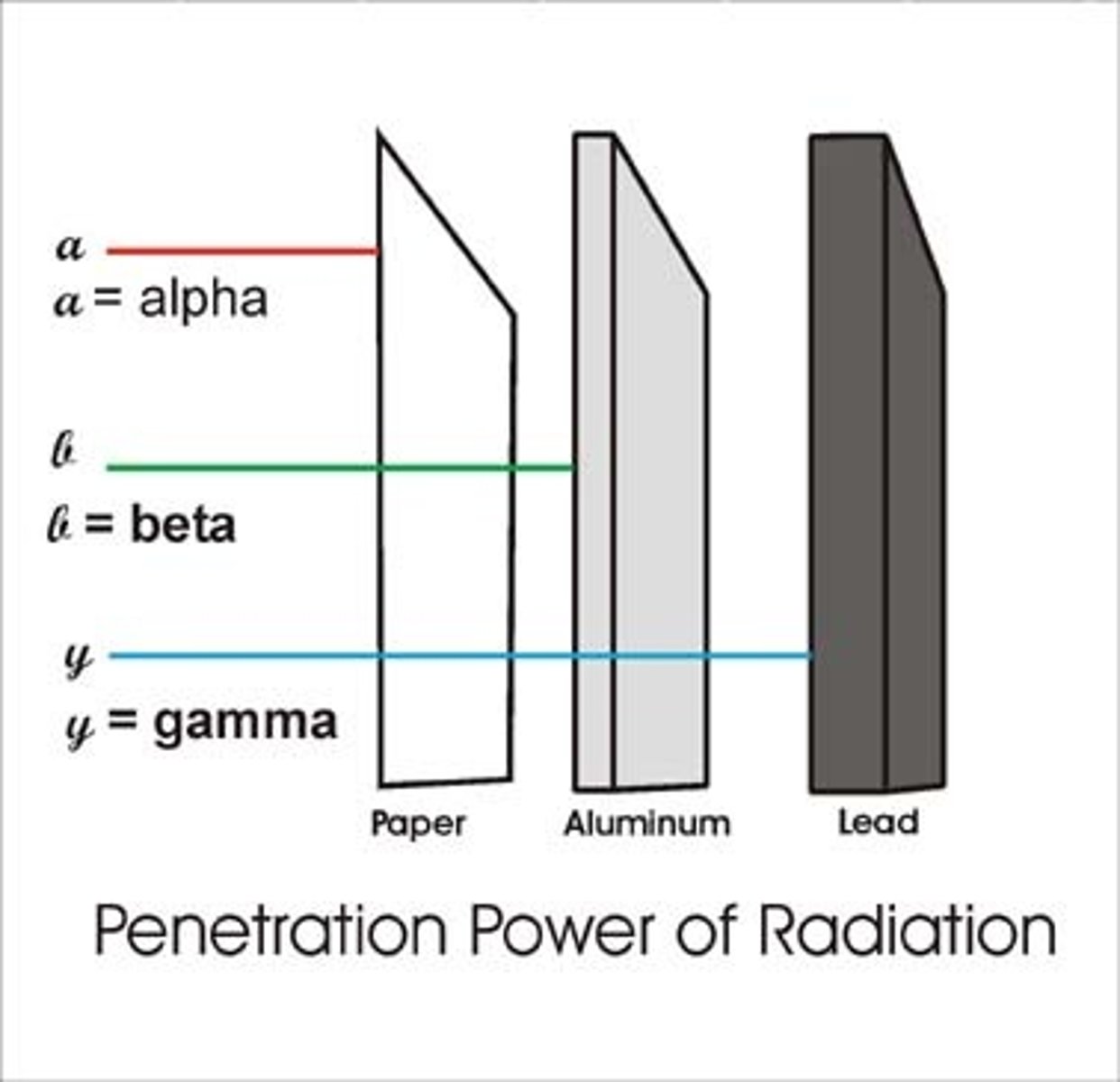
Penetrating power of a gamma ray
Strongly penetrating
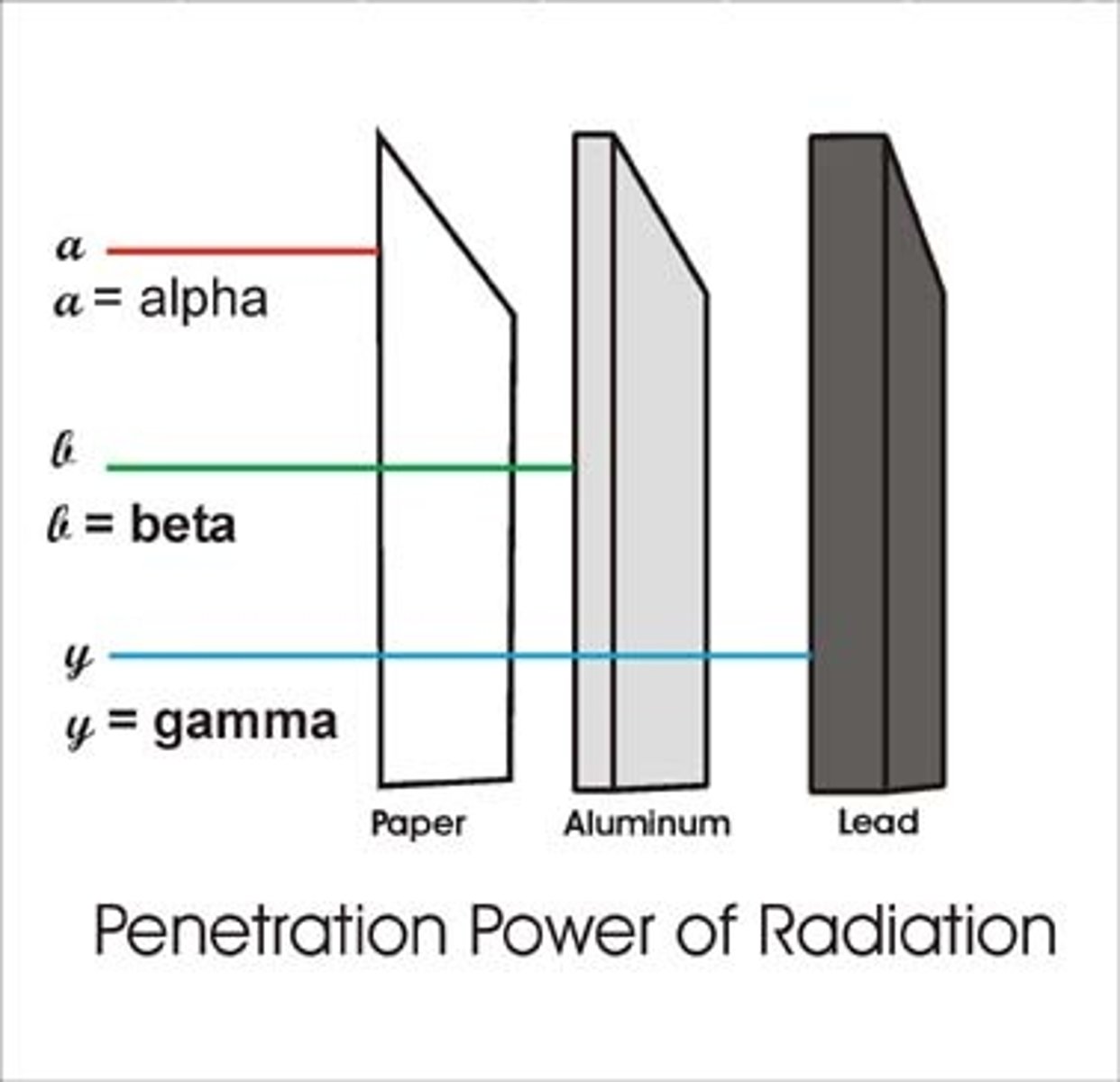
Materials that stop alpha particles
Skin or paper

Materials that stop beta particles
A few millimetres (mm) of aluminium

Materials that stop gamma rays
Several centimetres (cm) of lead or about 1 metre (m) of concrete

Range (in air)
How far nuclear radiation can travel in air before it is absorbed
Range of an alpha particle
Less than 5 centimetres (cm)
Range of a beta particle
About 1 metre (m)
Range of a gamma ray
Over 1 kilometre (km)
Irradiation
When an object is exposed to nearby nuclear radiation; the irradiated objects do not become radioactive themselves

Sterilisation
The process used to ensure that a sample contains no living things
Using irradiation to sterilise food
By exposing fruit to gamma rays which destroy any bacteria on the fruit but do not affect the fruit itself

Using irradiation for medical purposes
To sterilise surgical instruments and kill cancerous tumours deep inside the body using beams of gamma rays (a gamma knife)

Radioactive contamination
When unwanted radioactive atoms are mixed with other materials causing the materials to become radioactive as well

Using contamination for medical tracers
A radioactive isotope (beta or gamma emitter) is injected into the body, then later passes out of the body where it is detected and used to form an image
Using contamination to check for leaks in water pipes
A radioactive isotope (beta or gamma emitter) is added to the water supply and if there is a crack, contaminated water will leak into the ground causing a build-up of radiation that can be detected
Factors to consider when choosing a radioactive source
The type of radiation it emits, the half-life and the toxicity (whether it is poisonous or not)
Why a source with a long half life is unsuitable for use inside the body
The damaging effects of the radiation would last for too long and the patient would receive too high a dose
When alpha radiation is most dangerous
Inside the body
Why alpha radiation is most dangerous inside the body
Alpha radiation is strongly ionising and cannot penetrate through skin so it is highly likely to ionise cells within the body
Dangers of irradiation and contamination
Irradiation or contamination from radioactive decay damages living cells and causes mutations which can lead to radiation sickness or cancer
How to reduce irradiation
Irradiation can blocked with suitable shielding and will stop as soon as the source is removed
Why it is difficult to reduce contamination
Once an object is contaminated, the radiation cannot be blocked from it and it is usually very difficult to remove all of the contamination from material
Precautions to take when using radioactive sources
Keep sources in a lead-lined box when not in use, wear protective clothing, avoid contact with bare skin, limit exposure time, use tongs to handle sources, monitor exposure etc

Background radiation
Radiation that is around us all of the time
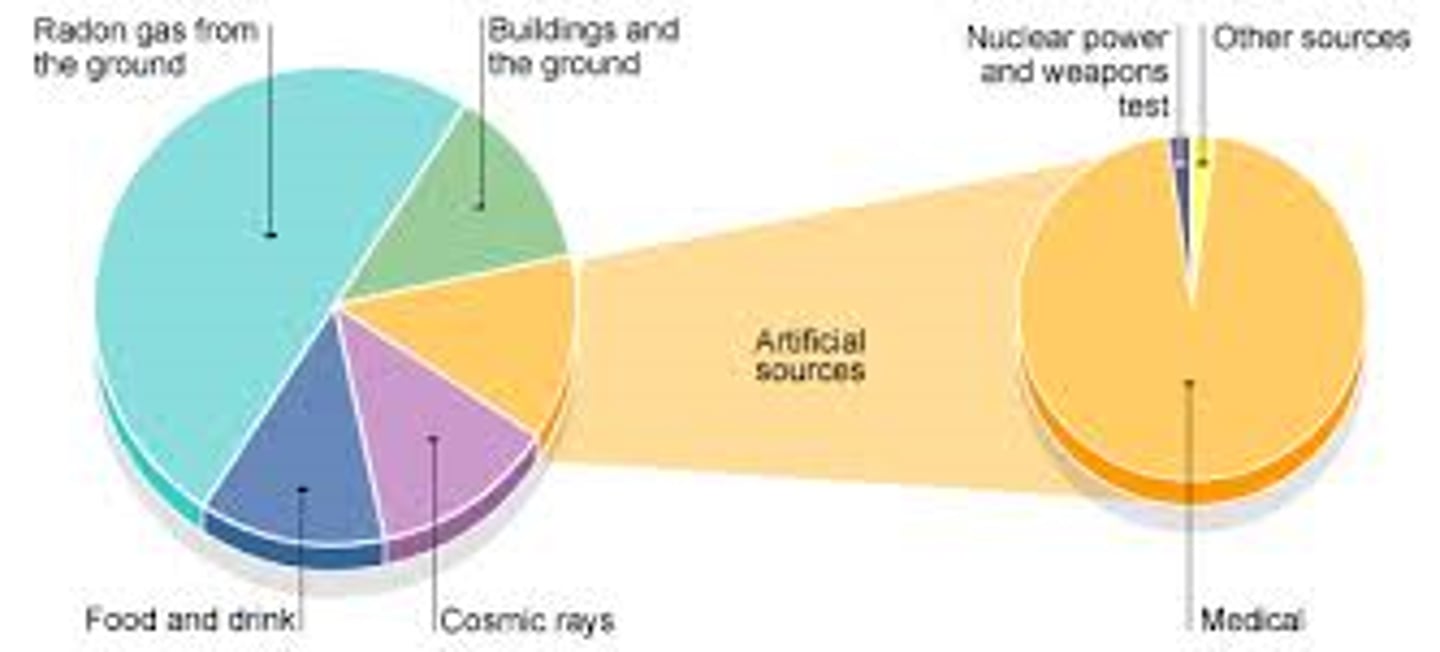
Sources of background radiation
Natural sources such as rocks and cosmic rays from space and man-made sources such as the fallout from nuclear weapons testing and nuclear accidents
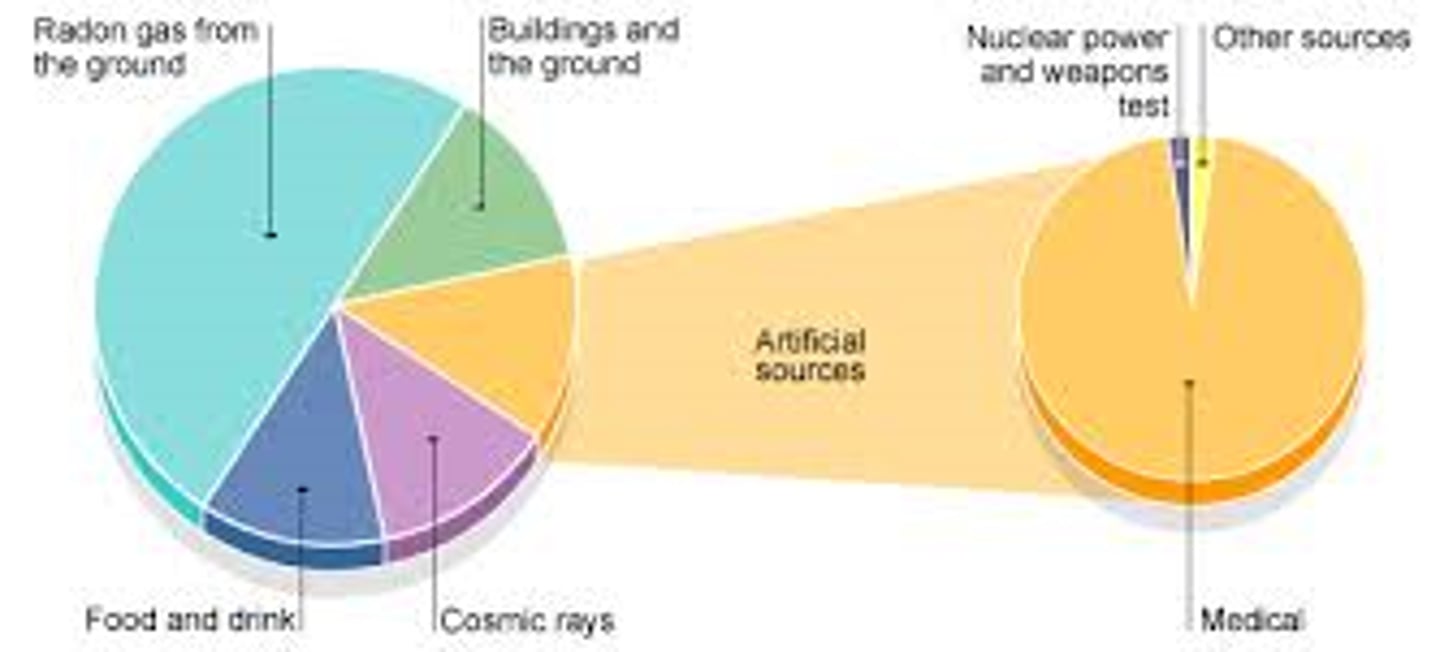
Factors affecting exposure to background radiation
Location and occupation among other things
Activity
The number of decays per second from an unstable nucleus
Becquerel (Bq)
The SI unit for activity
Why activity is unsuitable to measure radiation exposure
The activity of two sources could be the same, but one could emit alpha whereas the other emits beta (so they would each have a different effect on a person)
When alpha radiation is more dangerous to a person than beta
When the radioactive source is inside the body
Why alpha radiation is more dangerous to a person than beta when inside the body
An alpha particle is more ionising and it cannot penetrate the skin so will not be able to escape from the body