Key Concepts in Chemical Bonding and Molecular Structure
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
50 Terms
Valence Electrons
Electrons in the outermost shell of an atom.
Lewis Structure
Diagram showing bonds and lone pairs in molecules.
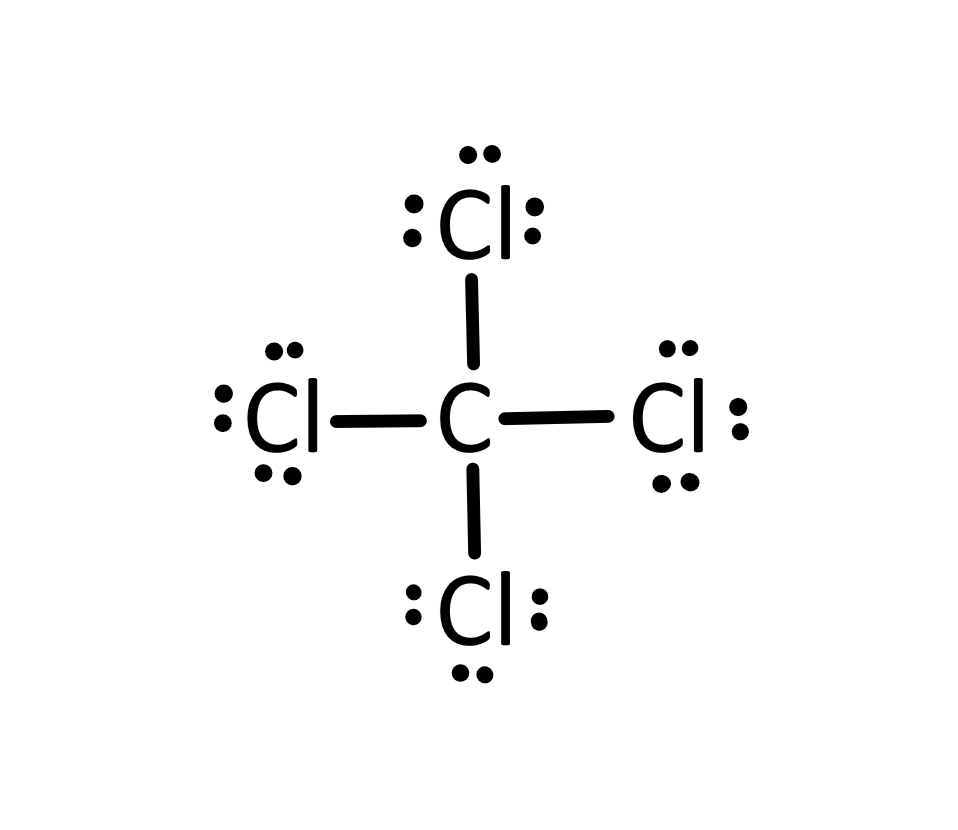
Structural Formula
Representation of molecule showing arrangement of atoms.
Lone Pair of Electrons
Two electrons not involved in bonding.
Covalent Bond
Bond formed by sharing electron pairs.
Electronegativity
Tendency of an atom to attract electrons.
Polar Molecule
charge distribution is also asymmetric so there are partial charges on the molecule
Polar bond
Uneven distribution of electrons, one side of the molecule being positive will other is negetive - asymetrical
Non-Polar Bond
molecule that has no electrial charge of partical charge - symmetrical
Molecular Symmetry
parts can be interchanged without changing the overall structure or orientation of the molecule
Structural Isomers
Compounds with the same formula but different structures.
Ionic Bond
Bond formed by the transfer of electrons.
Metallic Bond
Bond formed by delocalized electrons in metals.
Delocalized Electrons
Electrons that are spread over multiple atoms.
Alloy
Mixture of two or more metals.
Interstitial Alloy
Alloy with smaller atoms fitting between larger ones.
Substitutional Alloy
Alloy where some metal atoms are replaced.
Molecular Dipole
Separation of charge within a molecule.
Column 1 2 7
linear •-• •-•-•
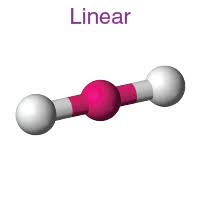
Column 3
triangular planer
Column 4
Terahedral
Column 5
Peramiddle
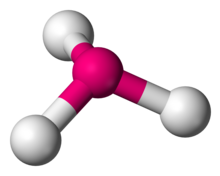
Column 6
bent
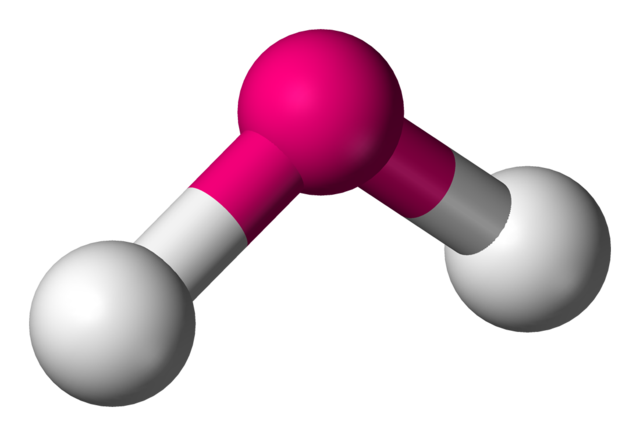
groups 2 3 4
symetrical - if surroundings are the same
groups 5 6
- asymetrical
meth-
1 carbon
eth-
2 carbons
prop-
3 carbons
but-
4 carbons
pent-
5 carbons
hex-
6 carbons
hept-
7 carbons
oct-
8 carbons
non-
9 carbon
dec-
10 carbons
-ane
single bond
-ene
double bond
-yne
triple bond
Oxygen (O2)
nonpolar
Nitrogen (N2)
nonpolar
Carbon Diozide (CO2)
nonpolar
most hydrocarbons like oil and gasoline are
nonpolar
water (H2O)
polar
Ammonia (NH3)
polar
Sulfer Dioxide (SO2)
polar
noble gases like helium neon and argon are
nonpolar