Isotopes
Major Element Behavior
Major elements controls the mineral phases that form
The tendency for a major element to go into a phase depends on the overall chemistry of the system and what phases are competing for that element (P, T, phase diagrams)
The concentration of a major element in a phase is usually buffered by the system, so that it varies little in a phase as the system composition changes
Trace Element Behavior

Trace element concentrations in the Henry;s law region of concentration, so their activity varies in the direct relation to their concentration in the system
1/23
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
24 Terms
Major Element Behavior
Major elements controls the mineral phases that form
The tendency for a major element to go into a phase depends on the overall chemistry of the system and what phases are competing for that element (P, T, phase diagrams)
The concentration of a major element in a phase is usually buffered by the system, so that it varies little in a phase as the system composition changes
Trace Element Behavior
Trace element concentrations in the Henry;s law region of concentration, so their activity varies in the direct relation to their concentration in the system

Trace elements during mantle melting
Compositional differences can reflect:
Heterogenous source rocks
Chemically and/ or mineralogically
Different degrees of partial melting
Varying conditions under which the melting takes place (depth= P, H2O)
Stable and Radioactive Isotopes
Stable isotopes are those that remain indefinitely
Radioactive isotopes decay to other nuclides
The rate of decay is constant, and not affected by P,T,X…
Parent nuclide= radiative nuclide that decays
Daughter nuclide(s) are the radiogenic atomic products
they may be stable but also can be radioactive and decay again at different rate (U-series)
What creates the observed differences in isotopic ratios in rock and minerals?
mass fraction (stable isotopes)- paleoclimate
compatibility to different reservoirs- magmatic processes; source signature (Sr-Rb, Sm-Nd)
Time-dating (Sr-Rb, Sm-Nd, U-Th-Pb)
Mass Fractionation (as for stable isotopes)
Generally most effective for light element isotopes where the differences in mass between isotopes is greatest
E.g H, He, C, O, S
But also new techniques measure Mg,Fe,Si,V
If any mass fractionation does take place, the light isotope always fractionates, preferably into the phase with weaker bonding, and is generally favored in the vapor over the liquid and in the liquid over the solid
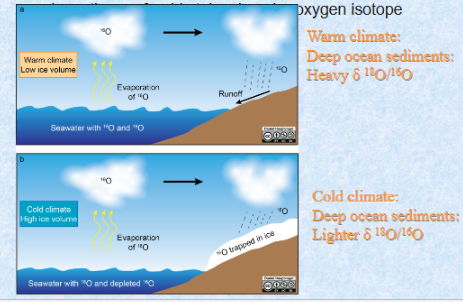
δ 18O/16O (or written as just δ 18O) application on Paleoclimate
Evaporation preferably takes lighter oxygen isotope (16O); condensation preferably takes heavier oxygen isotope
Daughters produced in varying proportions resulting from previous event of chemical
fractionation
40K → 40Ar by radioactive decay
Basalt → rhyolite by FX (a chemical fractionation process)
Rhyolite has more K than basalt
40K → more 40Ar over time in rhyolite than in basalt
40Ar/39Ar ratio will be different in each
Time
The longer 40K → 40Ar decay takes place, the greater the difference between the basalt and rhyolite will be
Radioactive Decay
→ age of a sample (t) if we know:
D the amount of the daughter nuclide produced
N0 the amount of the original parent nuclide
N the amount of remaining nuclide
y the decay constant for the system in question

Isotopic Systematics
Heavy Isotopic ratio values are determined by the parent/daughter ratios in the original solid source
Magmatic processes (FC, Melting) DO NOT change ratios
The time over which this “closed system” evolves
Mixing of sources or melts will alter the ratios as will assimilation
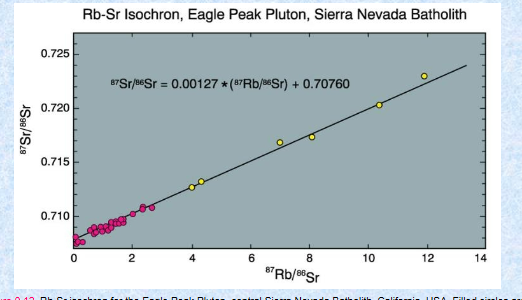
Sr-Rb System
Rb87→Sr87+ a beta particle (y = 1.42 x 10-11 a-1)
Half life is 48.8 Ga
Rb behaves like K → micas and alkali feldspar
commonly highly incompatible
Sr behaves like Ca → plagioclase and apatite (but not clinopyroxene)
Commonly moderately incompatible
Sr88:Sr87:Sr86:Sr84 ave. sample= 10:0.7:1:0.07
Sr86 is a stable isotope, and not created by breakdown of any other parent
Isochron Technique
Requires 3 or more cogenetic samples with a range of Rb/Sr
Could be:
• 3 cogenetic rocks derived from a single source by partial melting, FX, etc• 3 coexisting minerals with different K/Ca (~ Rb/Sr) ratios in a single rock
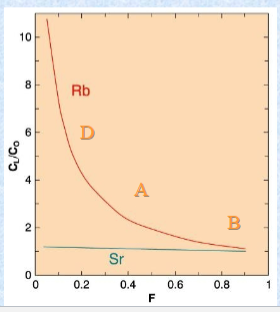
Isochron technique produces 2 valuable things:
1. The age of the rocks (from the slope = yt)
2. (87Sr/86Sr)o = the initial value of 87Sr/86Sr
Isotopic Systematics
Heavy Isotopic ratio values are determined by the parent/daughter ratios in the original solid source
AND
The time over which this “closed system” evolves
Mixing of sources or melts will alter the ratios as will assimilation
Magmatic processes (FC, Melting) DO NOT change ratios
Using magmas as “windows” to the mantle
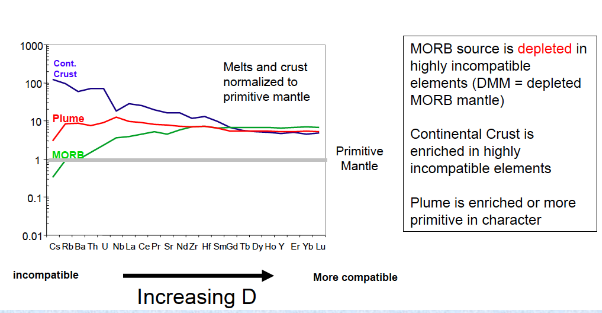
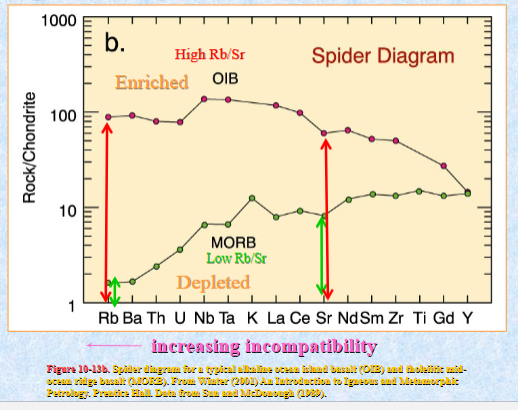
Spider diagram for oceanic basalts
The Sm-Nd System
l Both Sm and Nd are LREE
F Incompatible elements fractionate → melts
F Nd has lower Z → larger → liquids > does
Sm
Geochemical Reservoirs
Conceptual sources of melts/magmas that have distinct, trace element and isotopic compositions
They appear to be “end-member” types of compositions that = developed as the mantle evolved throughout Earth history
Incompatible trace elements became enriched or depleted from various melting and/or recycling events THUS the isotopic compositions evolved through time
Geochemical Reservoirs Examples

The U-Pb-Th System
Very complex system.
3 radioactive isotopes of U: 234U, 235U, 238U
3 radiogenic isotopes of Pb: 206Pb, 207Pb, and 208Pb
- Only 204Pb is strictly non-radiogenic
U, Th, and Pb are incompatible elements, & concentrate in early melts
Isotopic composition of Pb in rocks =…
The U-Pb-Th System