Chapter 1 - Properties of Fluids
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
80 Terms
Fluid Mechanics
a physical science dealing with the action of fluids at rest or in motion, and with applications and devices in engineering using fluids
Fluid Statics
Fluid Dynamics
Two major areas of fluid mechanics:
Fluid Statics
deals with fluids at rest
Fluid Dynamics
concerned with fluids in motion
Hydrodynamics
applied to the flow of liquids or to low-velocity gas flows where the gas can be considered as being essentially incompressible
Hydraulics
deals with the application of fluid mechanics to engineering devices involving liquids, usually water or oil
Hydraulics
deals with such problems as the flow of fluids through pipes or in open channels, the design of storage dams, pumps, and water turbines, and with other devices for the control or use of liquids, such as nozzles, valves, jets, and flowmeters
Ideal Fluids
Real Fluids
Two categories of fluids:
Ideal Fluids
assumed to have no viscosity (and hence, no resistance to shear)
Ideal Fluids
incompressible
Ideal Fluids
have uniform velocity when flowing
Ideal Fluids
no friction between moving layers of fluid
Ideal Fluids
no eddy currents or turbulence
Real Fluids
exhibit infinite viscosities
Real Fluids
non-uniform velocity distribution when flowing
Real Fluids
compressible
Real Fluids
experience friction and turbulence in flow
Newtonian Fluids
Non-Newtonian Fluids
Real fluids are further divided into:
Pseudoplastic Fluids
Delatant Fluids
Bingham Fluids
What is under non-Newtonian fluids?
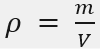
Mass Density
mass per unit volume
English: slugs/ft3
Metric: gram/cm3
SI: kg/m3
Units of Mass Density (ρ):
Density in pound-mass divided by g, which represents the acceleration due to gravity (ρlbm/g)
Density expressed in slugs (ρslugs) is equal to what?
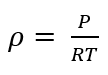
ρ - absolute pressure of gas in Pa
R - gas constant Joule/kg-°K
T - absolute temperature in °Kelvin
Formula of density for an ideal gas:
287 J/kg-°K
1716 lb-ft/slug-°R
What is the gas constant of air?
°K = °C + 273
Temperature conversion from °C to °K:
°R = °F + 460
Temperature conversion from °F to °R:
1.29
1.20
790
602
720
1260
13600
1000
What are the approximate room temperature densities of common fluids?
Air (STP)
Air (21 °F, 1 atm)
Alcohol
Ammonia
Gasoline
Glycerine
Mercury
Water
Specific Volume (Vs)
the volume occupied by a unit mass of fluid
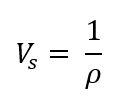
What is the formula for specific volume?
Specific Weight/Unit Weight (γ)
the weight of a unit volume of a fluid
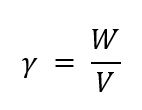
γ = ρg
What are the formulas for specific weight/unit weight (γ)?
English: lb/ft3
Metric: dyne/cm3
SI: N/m3 or kN/m3
Units of Specific Weight/Unit Weight (γ):
Specific Gravity (sg)
a dimensionless ratio of a fluid’s density to some standard reference density
Water at 4 °C
For liquids and solids, what is the reference density?
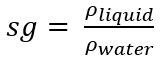
What is the formula of specific gravity (sg)?
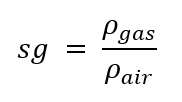
Density of air
In gases, what is the standard reference to calculate the specific gravity?
γ = 62.4 lb/ft3 = 9.81 kN/m3
ρ = 1.94 slugs/ft3 = 1000 kg/m3
sg = 1.0
For water 4 °C, what are the values for:
γ
ρ
sg?
Viscosity (µ)
determines the amount of its resistance to shearing forces
no viscosity
A perfect fluid would have _____.
Constant of Proportionality
What is the k?
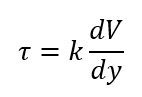
Dynamic of Absolute Viscosity
What is the constant of proportionality k called?
μ
What is the dynamic of absolute viscosity denoted by?
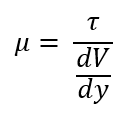
τ = shear stress in lb/ft2 or Pa
μ = absolute viscosity in lb-sec/ft2 (poises) or Pa-sec
y = distance between the plates in ft or m
U = velocity in ft/s or m/s
What is the formula for viscosity (μ)?
Kinematic Viscosity (v)
the ratio of dynamic viscosity of the fluid, μ, to its mass density, ρ
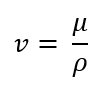
μ = absolute viscosity in Pa-sec
ρ = density in kg/m3
What is the formula for kinematic viscosity (v)?
English = lb-sec/ft2 (slug/ft-sec)
Metric = dyne-s/cm2 (poise)
SI = Pa-s (N-s/m2)
What are the common units of viscosity for (absolute, μ):
English
Metric
SI?
English = ft2/sec
Metric = cm2/s (stoke)
SI = m2/s
What are the common units of viscosity for (kinematic, v):
English
Metric
SI?
0.1
0.0001
1 poise = _____ Pa-sec
1 stoke = _____ m2/s
surface tension
The membrane of “skin” that seems to form on the free surface of a fluid is due to the intermolecular cohesive forces, and is known as _____.
Surface Tension (σ)
the reason that insects are able to sit on water and a needle is able to float on it
Surface Tension (σ)
causes bubbles and droplets to take on a spherical shape, since any other shape would have more surface area per unit volume
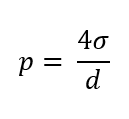
σ = surface tension in N/m
diameter of the droplet in m
p = gage pressure in Pa
What is the formula of surface tension (σ)?
Capillarity/Capillary Action
the name given to the behavior of the liquid in a thin-bore tube
surface tension, cohesion, adhesion
The rise or fall of a fluid in a capillary tube is caused by _____ and depends on the relative magnitudes of the _____ of the liquid and the _____ of the liquid to the walls of the containing vessel.
adhesion > cohesion, cohesion > adhesion
Liquids rise in tubes they wet (_____) and fall in tubes they do not wet (_____).
Capillary
important when using tubes smaller than about 3/8 inch (9.5 mm) in diameter
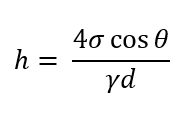
h = capillary rise or depression in m
γ = unit weight in N/m3
d = diameter of the tube in m
σ = surface tension in Pa
What is the formula of capillarity/capillary action?
0°
What is the angle for complete wetting, as with water on clean glass?
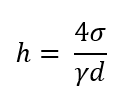
What will be the formula if the angle is 0° for capillarity?
Mercury-glass = 140°
Water-paraffin = 107°
Water-silver = 90°
Kerosene-glass = 26°
Glycerin-glass = 19°
Water-glass = 0°
Ethyl alcohol-glass = 0°
What are the contact angles for:
Mercury-glass
Water-paraffin
Water-silver
Kerosene-glass
Glycerin-glass
Water-glass
Ethyl alcohol-glass?
Compressibility/Coefficient of Compressibility (β)
the fractional change in the volume of a fluid per unit change in pressure in a constant-temperature process
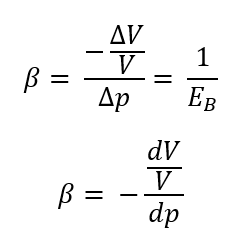
△V = change in volume
V = original volume
△p = change in pressure
dV/V = change in volume (usually in percent)
What are the formulas for compressibility (β)?
Bulk Modulus of Elasticity (EB)
expresses the compressibility of the fluid
Bulk Modulus of Elasticity (EB)
the ratio of the change in unit pressure to the corresponding volume change per unit of volume
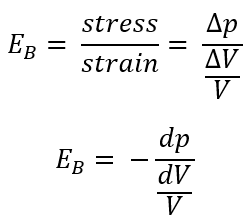
What are the formulas for bulk modulus of elasticity (EB)?
Pressure Disturbances
imposed on a fluid move in waves
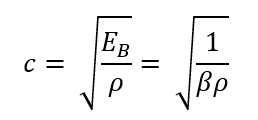
celerity, acoustical, sonic velocity
The velocity or _____ of pressure wave (also known as _____ or _____) is expressed as:
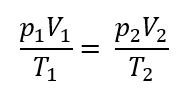
PROPERTY CHANGES IN IDEAL GAS
For any ideal gas experiencing any process, the equation of state is given by:
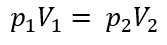
Boyle’s Law
PROPERTY CHANGES IN IDEAL GAS
When temperature is held constant, what is the original formula reduced to?
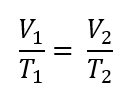
Charle’s Law
PROPERTY CHANGES IN IDEAL GAS
When temperature is held constant (isothermal condition), what is the original formula reduced to?
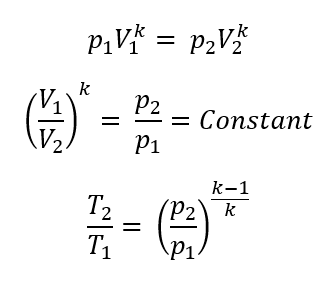
p1 = initial absolute pressure of gas
p2 = final absolute pressure of gas
V1 = initial volume of gas
V2 = final volume of gas
T1 = initial absolute temperature of gas in °K (°K = °C + 273)
T2 = final absolute temperature of gas in °K
k = ratio of the specific heat at constant pressure to the specific heat at constant volume (adiabatic exponent)
For Adiabatic or Isentropic Conditions (no heat exchanged), what are the formulas?
Molecular Activity
(in a liquid) will allow some of the molecules to escape the liquid surface
condense
Molecules of the vapor also _____ back into the liquid.
equilibrium processes
The vaporization and condensation at constant temperature are _____.
Vapor Pressure/Saturation Pressure
the equilibrium pressure exerted by free molecules
volatize liquids, barometers
Liquids near their boiling point or that vaporizes easily are said to _____. Liquids with low vapor pressure are used in accurate _____.
temperature
The tendency toward vaporization is dependent on the _____ of the liquid.
Boiling
occurs when the liquid temperature is increased to the point that the vapor pressure is equal to the local ambient (surrounding) pressure
local ambient pressure, vaporize
A liquid’s boiling temperature depends on the _____, as well as the liquid’s tendency to _____.
Mercury = 0.000173
Turpentine = 0.0534
Water = 2.34
Ethyl Alcohol = 5.86
Ether = 58.9
Butane = 218
Freon-12 = 584
Propane = 855
Ammonia = 888
What are the typical vapor pressures (in kPa, 20 °C) for:
Mercury
Turpentine
Water
Ethyl Alcohol
Ether
Butane
Freon-12
Propane
Ammonia