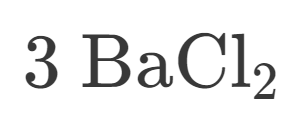Introduction to Chemical Formulas - Chemistry
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Metal
What is the yellow part?
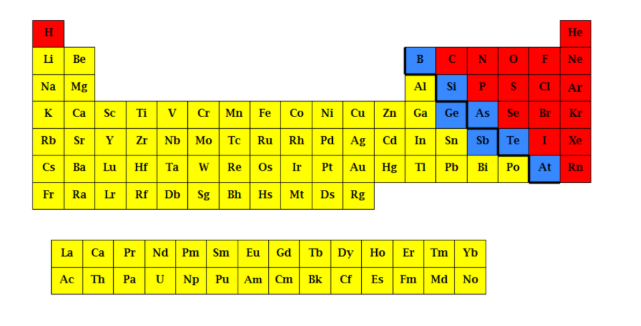
Metalloids
What is the blue part?
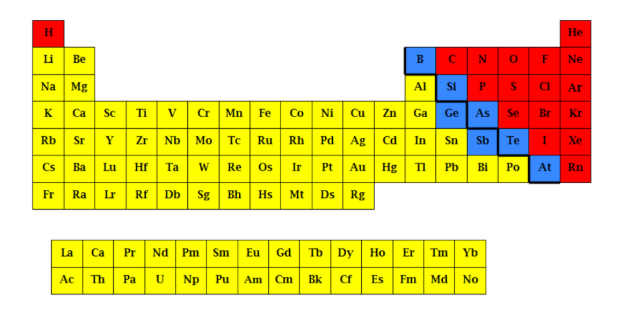
Non-metals
What is the red part?
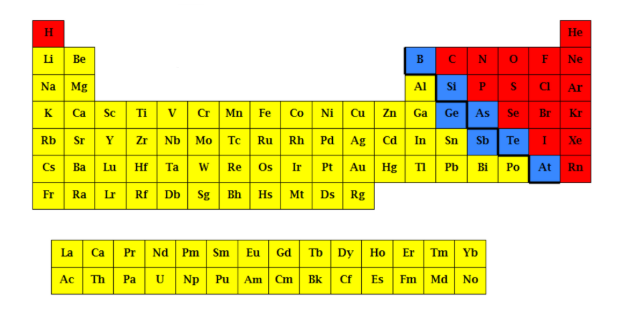
Nonmetal elements
Located to the right of the stair, including the two separate rows at the bottom of the periodic table
metal elements
located to the left of the stair
Metalloid elements
located along the stair-step line
Subscript
tells us the number of atoms in a compound, placed at the bottom right corner of them.
Parentheses
placed around a group of atoms to tell us the number of the atom in the group. Ex: Al(SO4)3
polyatomic ion
Atom groups originated from charged compound
1
How many Nitrogen are in this?
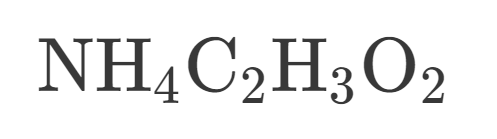
7
How many Hydrogen are in this?
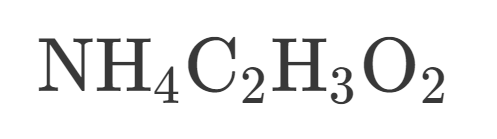
2
How many Carbon is in this?
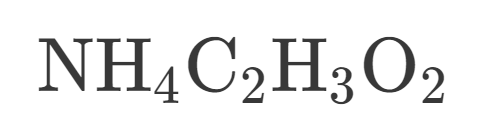
2
How many Oxygen is in this?
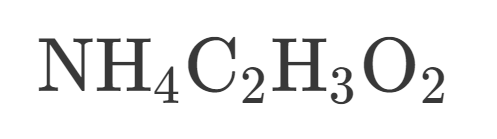
12
How many atoms are in this?
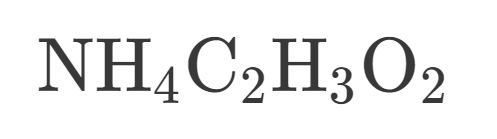
Covalent
Is this Covalent or Ionic?
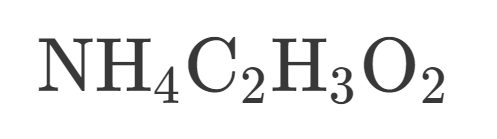
3
How many Barium is in this?
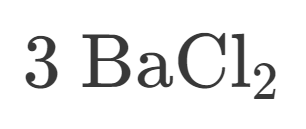
6
How many Chlorine is in this?
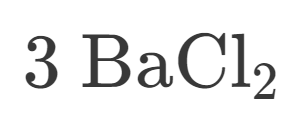
9
How many atoms are in this?
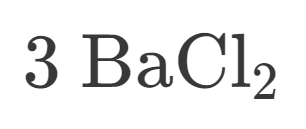
Ionic
Is this Ionic or Covalent?