Aldehydes, Ketones, and Hydride Reductions
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
16 Terms
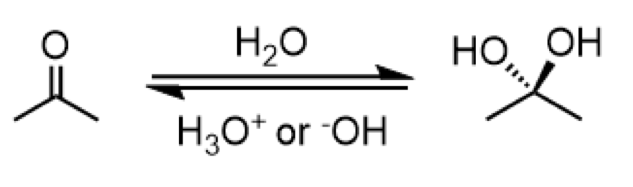
addition of water (hydrate formation)
reagents: H2O (forward), H3O+ or OH- (reverse)
formation of a double hydroxyl group attachment to a carbon
anti-addition
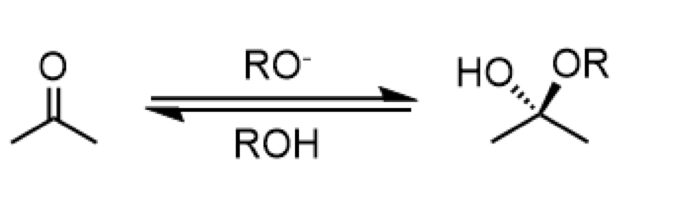
base-catalyzed addition of alcohol (hemiacetal formation)
reagents: RO- (forward), ROH (reverse)
anti-addition
formation of a hemiacetal
acid-catalyzed addition of alcohol (acetal formation); reversible with H3O+
reagents: ROH (forward), H3O+ (reverse)
anti-addition
formation of an acetal
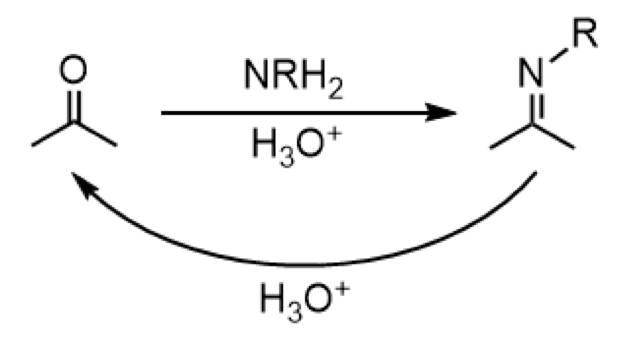
acid-catalyzed addition of primary amine (imine formation); reversible with H3O+
reagents: NRH2 (forward), H3O+ (reverse)
formation of imine
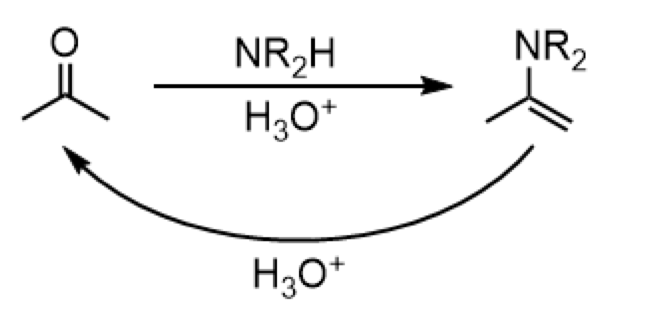
acid-catalyzed addition of secondary amine (enamine formation); reversible with H3O+
reagents: NR2H (forward), H3O+ (reverse)
formation of enamine
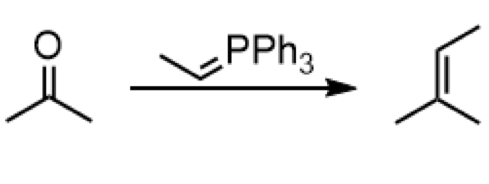
wittig reaction
reagents: PPh3=- (phsphorus ylide)
formation of alkene
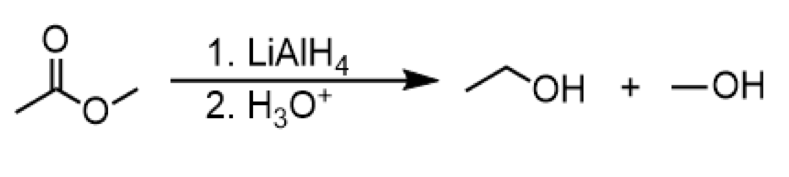
reduction of ester to primary alcohol
reagents: 1. LiAlH4 2. H3O+
reduction of acyl chloride to primary alcohol
reagents: 1. LiAlH4 2. H3O+
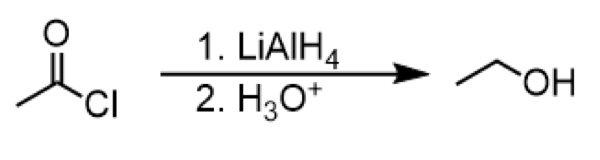
reduction of carboxylic acid to primary alcohol
reagents: 1. LiAlH4 2. H3O+
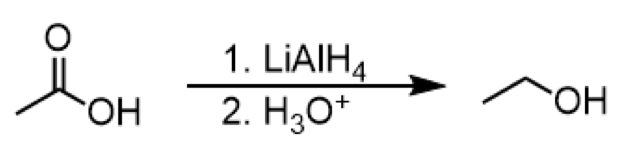
reduction of aldehyde to primary alcohol
reagents: 1. LiAlH4 2. H3O+ or 1. NaBH4 2. H3O+
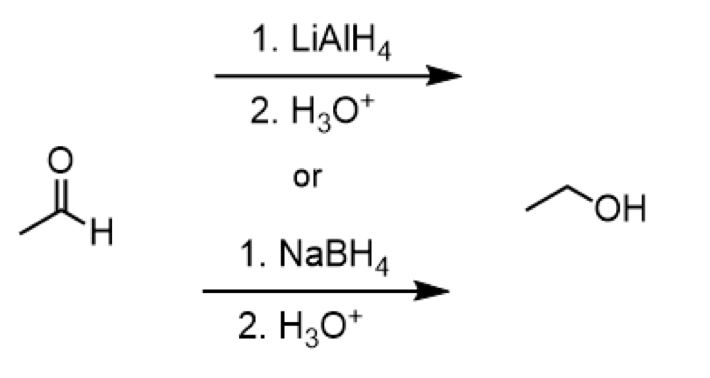
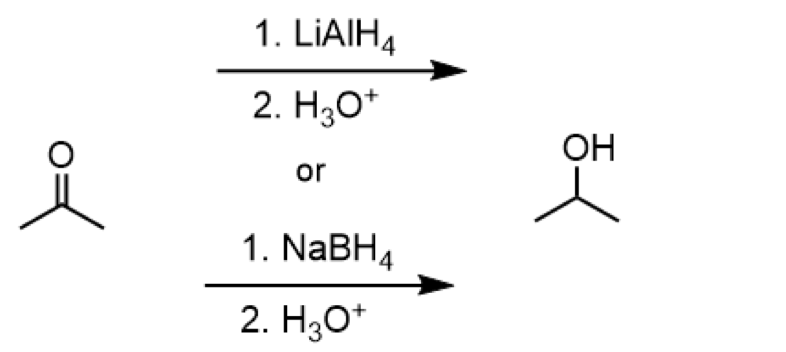
reduction of ketone to secondary alcohol
reagents: 1. LiAlH4 2. H3O+ or 1. NaBH4 2. H3O+
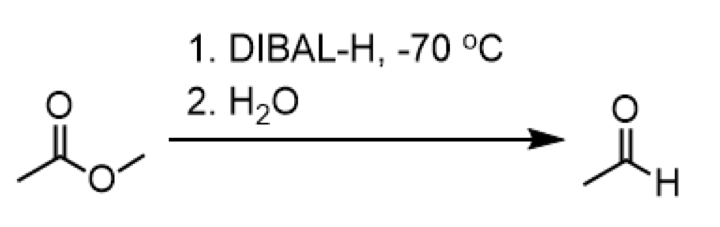
reduction of ester to an aldehyde
reagents: 1. DIBAL-H, -70oC 2. H2O
reduction of acyl chloride to aldehyde
reagents: LiAlH[O(CH3)3]3 or LTBA
![<ul><li><p>reagents: LiAlH[O(CH<sub>3</sub>)<sub>3</sub>]<sub>3</sub> or LTBA </p><p></p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/da6b3b0b-2b24-405b-9105-89d6ea748f40.png)
reduction of amide to amine
reagents: 1. LiAlH4 2. H2O
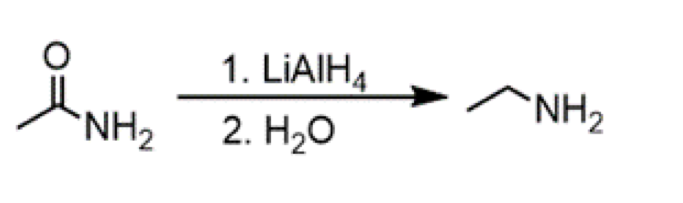
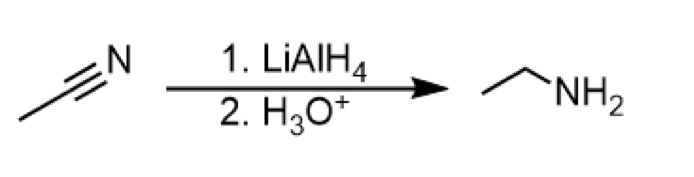
reduction of nitrile to amine
reagents: 1. LiAlH4 2. H3O+
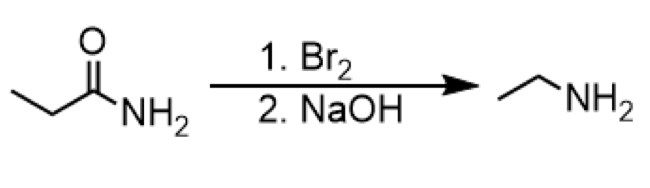
hofmann rearrangement
reagents: 1. Br2 2. NaOH
formation of primary amine from amide (with ethyl group)