Chemistry - Topic 6 - Organic Chemistry I
1/125
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
126 Terms
what are structural isomers?
molecules with the same molecular formula but different structural formulae
what are stereoisomers?
molecules with the same molecular formula and structural formula but a different spatial arrangement of atoms
what is chain isomerism?
the carbon chain is different
what is position isomerism?
functional groups can be in different positions on the carbon chain resulting in isomer
what is functional group isomerism?
some compounds with the same molecular formula can contain different functional groups
what are the conditions for E/Z isomerism?
there must be a double carbon bond
both Cs from the double carbon bond must have two different groups attached
around which bond is there restricted rotation and why?
a π bond
this is because π orbitals have electron density above and below plane but not in the middle (between atoms) so rotation not possible
what are the three steps to the mechanism of free radical substitution?
initiation
propagation (has two parts)
termination
explain the initiation step of the mechanism of free radical substitution
when exposed to ultraviolet light, a molecule of a halogen breaks apart into two halogen atom free radicals
the ultraviolet light provides the energy to break the covalent bond between the two halogen atoms
what happens in free radical substitution reactions?
H atoms are replaced by halogen atoms
what is a free radical?
species with an unpaired electron
they are very reactive
represented in mechanisms by a single dot
is formed by a homolytic fission of a covalent bond and results in the formation of radicals
what is homolytic fission?
homolytic fission is breaking a covalent bond in such a way that each atom takes an electron from the bond to form two radicals
explain the propagation step of the mechanism of free radical substitution
(molecule + radical → molecule + radical)
for every H replaced, there is one pair of propagation reactions
simply: 1 - the radical grabs a H; 2 - the alkyl radical grabs a Br
step 1 - the alkane/halogenoalkane reacts with the halogen free radical - this removes an H atom from the alkane/halogenoalkane - this produces HF/HCl/etc and a C based free radical
step 2 - the C based radical from step 1 reacts with the diatomic molecule e.g. F2 to put an atom of e.g. F onto the C based radical - this also produces another halogen free radical to continue the chain reaction in another step 1
explain the termination step of the mechanism of free radical substitution
( 2 radicals → molecule)
if two free radicals collide, they will form a molecule and stop the chain reaction
any two free radicals formed in the reaction could collide in this way
what is a homologous series?
a family of similar molecules with:
same chemical properties
trend in physical properties
same functional group
same general formula
what is the functional group of a molecule?
the atoms and bonds that determine the chemical properties of a compound
determines which homologous series a molecule belongs to
what are alkanes and cycloalkanes?
saturated hydrocarbons
what are alkenes and cycloalkenes?
unsaturated hydrocarbons
how are alkane fuels obtained?
fractional distillation
cracking
reforming
what is cracking?
converts heavier fractions, such as diesel oil and fuel oil, into more useful hydrocarbon fuels by breaking up large molecules into smaller ones, and converts straight-chain hydrocarbons to branched and cyclic hydrocarbons, in order to supply more of the smaller molecules
this is because shorter chained alkanes are in more demand than longer chained alkanes
also produces alkenes for plastic/polymer productions
cracking is the thermal decomposition of alkanes
converts carbon chains with 12 or more carbon atoms into a mixture of branched alkanes, cycloalkanes, alkenes and branched alkenes
what is fractional distillation?
first stage in refining crude oil
separates crude oil into different fractions
fuel fractions must be refined to remove sulphur compounds, which would cause air pollution when they burn
for smooth running, petrol must burn smoothly in the engines of vehicles and not in fits and starts. to ensure smooth combustion, companies produce fuel with a high octane number by increasing the proportions of branched alkanes and arenes, or blending-in oxygen compounds
the three main methods to increase the octane number of fuels are?
cracking
reforming
adding ethanol and ethers
how to produce a higher proportion of alkenes through cracking?
conduct cracking at higher temperatures in the presence of steam
how to produce a higher yields of branched and cyclic alkanes? state the conditions and explain why such a temperature is needed.
conducting cracking in the presence of a catalyst (catalytic cracking)
either a zeolite catalyst or silica + alumina (SiO2 + Al2O3)
650°C - high temperatures required to break the strong C-C covalent bonds
what is reforming?
increases the proportion of branched, cyclic and aromatic (benzene) hydrocarbons (from straight chain alkanes and cyclic alkanes)
branched and cyclichydrocarbons are required to increase the octane number of fuel to allow smoother burning/more efficient combustion
hydrogen is a valuable by-product of the process - it can be used elsewhere at the refinery
what are the conditions for reforming?
palladium catalyst
500°C
high pressure
platinum or rhodium catalyst supported on an inert material such as aluminium oxide
engines that burn petrol or diesel fuels can pollute the air for three main reasons:
incomplete combustion
fuel contains impurities
they run at such a high temperature that nitrogen and oxgen in the air can react
which pollutants are formed during the combustion of alkane fuels?
carbon monoxide (toxic)
oxides of nitrogen and sulphur (acid rain)
carbon particulates
unburned hydrocarbons (through incomplete combustion or evaporation of fuel - includes benzene which is carcinogenic)
suggest a reason why oil companies reform alkanes such as heptane?
has more efficient combustion/allows smoother burning/increases octane number/reduces knocking/is a more efficient fuel
what is an electrophile?
an electron deficient species that can accept a lone pair of electrons
how do catalytic converters solve some problems caused by pollutants?
they remove CO, NOx and unburned hydrocarbons from the exhaust gases, turning them into CO2, N2 and H2O
what property allows crude oil to be separated by fractional distillation?
crude oil is a mixture with boiling ranges
why should we use fuels such as biodiesel as opposed to petrol and diesel?
save fossil fuels
more sustainable
uses renewable resources
plants more carbon neutral
biodiesel has a smaller carbon footprint
what induces a dipole in Br2?
the pi bond in the double carbon bond
why do alkenes undergo addition reactions?
they contain a double carbon bond with a pi bond that is relatively weak and can break and bond to an electrophile
what is the positive inductive effect?
alkyl groups can stabilise a carbocation by pushing electrons towards the positive charge
what is polymerisation?
a reaction where many monomers join together
what is a monomer
a small molecule that can join together to form a polymer
what is a polymer?
a giant molecule formed from many monomers
what is the name given to the class of compounds used as radical initiators in addition polymerisation?
peroxides
(composed of O-O single bond) → likely to break with homolytic fission
waste polymers can be separated into which 3 specific types of polymers?
sorting (using IR camera) and recycling
incineration to release energy
use as a feedstock for cracking
advantages and disadvantages of processing method 1 of polymers?
sorting (using infrared camera) and recycling
reduces landfill waste, reduces consumption of raw/precious material
household plastics contain a mixture of polymers - hard and expensive to sort, also involves the use of energy (to make the new product)
advantages and disadvantages of processing method 2 of polymers?
incineration to release energy
reduces landfill waste + releases energy for generating electricity (generates energy for other processes)
releases greenhouse gases, releases toxic gases such as SO2, NOx, HCl that need to be removed
advantages and disadvantages of processing method 3 of polymers?
feedstock for cracking
reduces landfill waste, converts waste products into useful chemical feedstock
high energy input required (from crude oil), some polymers, e.g. PVC, release toxic gases like HCl when heated strongly
how can chemists limit the problems caused by polymer disposal?
develop biodegradable polymers (including photodegradable)
advantages - break down or decompose naturally in a quicker time (within 9 months), will not require landfill as it can degrade in sunlight
disadvantages - cannot be reused, cannot be used in repeated outdoor applications e.g. window frames
removing toxic waste gases caused by incineration of plastics - calcium oxide or calcium hydroxide react with acidic gases
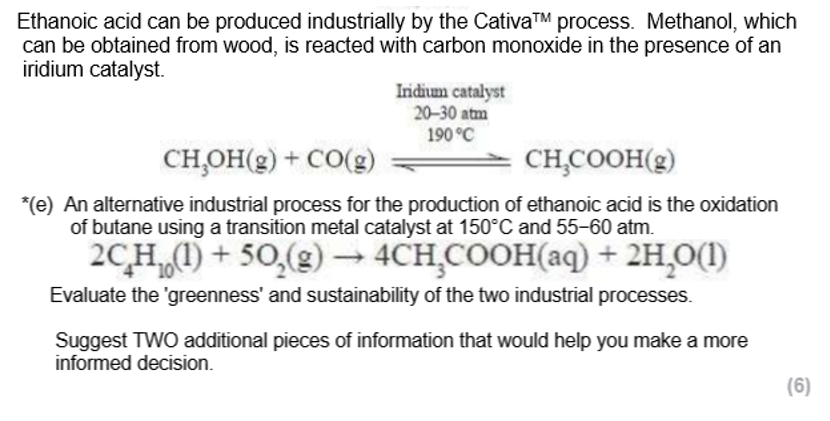
oxidation of alkene, observations, catalysts, conditions and type of product
KMnO4 and H2SO4 - acidified potassium manganate (VII)
room temperature
purple to colourless
produces a diol
electrophilic addition reaction (goes via a carbocation) - addition of steam to an alkene with acid catalyst, observations, catalysts and type of product
phosphoric acid catalyst (H3PO4)
reacts with H2O gas
produces alcohol
known as acidic elimination
catalytic hydrogenation - observations, conditions, catalysts and type of product + special reaction
nickel catalyst at 150°C or platinum catalyst at room temperature
reacts with hydrogen
alkane product
also a reduction reaction as adding hydrogen
this reaction is used to manufacture margarine from unsaturated vegetable oils
electrophilic addition reaction (goes via a carbocation) - alkene with bromine liquid, observations, catalysts and type of product
the pi bond induces a dipole in the Br2
forms a dibromoalkane
orange to colourless
can form a dihalogenoalkane using another halogen or a mixed interhalogen compound e.g. ICl
electrophilic addition reaction (goes via a carbocation) - alkene with bromine water, Br2(aq), observations, catalysts and type of product
makes some type of bromoalcohol + HBr
orange to colourless
UV light required
what is heterolytic bond fission?
when a covalent bond breaks, leaving one atom with both electrons from the covalent bond pair and hence the other atom does not retain any electrons from the bond pair
this leads to the formation of ions
what is a nucleophile?
a lone pair donor
tends to be delta negative or negative, must have a lone pair
what is a hydrolysis reaction?
water or hydroxide ions break a chemical compound into two compounds
which is a poor nucleophile, water or OH- ions
water
what does the reactivity of halogenoalkanes depend on?
the strength of the C-X bond
the halogenoalkane with the weakest C-X bond will have the fastest rate of reaction
why is it that the more branched a halogenoalkane gets, the faster the substitution reaction occurs?
in the tertiary halogenoalkane, the delta positive carbon is bonded to three electron-releasing alkyl groups. this reduces the magnitude of the positive charge on the carbon atom
this gives the intermediate carbocation greater stability
and allows easier escape of the leaving group (negative halide ion)
hence the C-X bond is weaker in branched halogenoalkanes and the substitution reaction faster
name the type of reaction, mechanism and product formed when a halogenoalkane reacts with dilute aqueous potassium hydroxide and is heated under reflux.
hydrolysis reaction
nucleophilic substitution reaction
alcohol
order the halogenoalkanes from most reactive to least reactive and explain why
iodoalkane
bromoalkane
chloroalkane
the reactivity of the halogenoalkane depends on the stength of the C-X bond
the halogenoalkane with the weakest C-X bond will have the fastest rate of reaction
explain the trend in boiling points as a molecule becomes increasingly branched
the boiling points decrease with increased branching
more branching gives less surface area of contact and therefore weaker London dispersion forces
less energy required to overcome London dispersion forces/intermolecular forces
explain why MTBE is added to petrol
to increase the octane number/to ensure smooth burning/to reduce knocking
explain why compound G has stereoisomers
there is restricted rotation around the C=C bond
and two different groups attached to each carbon atom of the C=C bond
state why the empirical formula of a poly(alkene) is the same as that of the monomer from which it is produced.
no atoms lost or gained
suggest why a small amount of UV light can result in the formation of a large amount of product.
because a chlorine radical is regenerated
suggest why calcium oxide reacts with sulphur dioxide
calcium oxide is basic/sulphur dioxide is an acidic gas
suggest why the calcium oxide is coated on a mesh
bigger surface area to react
why can the C=C double bond not be rotated?
the C=C double bond cannot be rotated due to the side-on overlap of p orbitals - this occurs if both the carbons have different groups attached to them.
suggest the name of a different haloalkane that could be used to improve the atom economy. explain your decision.
1-chloropropane or 1-fluoropropane as the total Mr of the products will be less (so the atom economy will be greater).
why is it that as the length of the carbon chain increases, the solubility of alcohols in water decreases?
as the alkyl group increases in size, there are stronger london dispersion forces between the alcohol molecules
these forces are weaker than the intermolecular forces between water and alcohol molecules therefore lower solubility
although the OH group can still form hydrogen bonds with water, this is outweighed by the long, non-polar alkyl chain
explain two points of procedure that would maximise the yield of the aldehyde (2)
heat and distil (to remove the aldehyde from the mixture before it reacts further)
use a stoichiometric amount of oxidising agent (rather than excess)
explain why alkanes are unreactive. (2)
C-H bond enthalpy is large or C-H bonds are non-polar
so alkanes are not attacked
give a test to identify HCl gas
rod dipped in NH3 → should see white clouds (solid - NH4Cl)
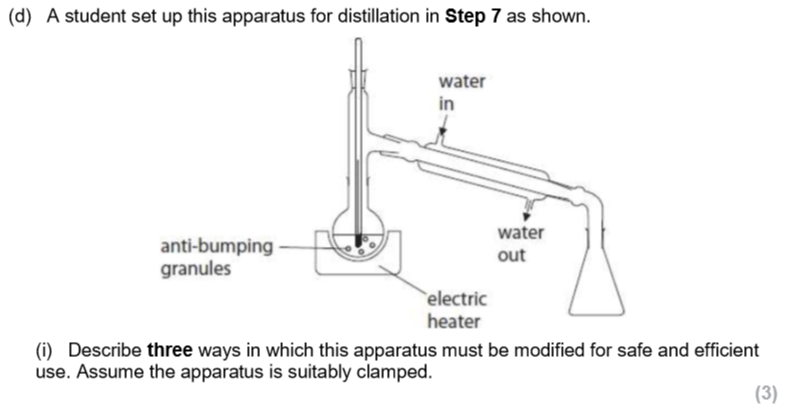
why should anti-bumping granules be added to the round-bottomed flask?
prevents superheating/localised heating
prevents violent boiling
promotes smooth and uniform boiling
explain fully why ethanal, rather than ethanoic acid, is produced and collected when the apparatus for distillation is used. (2)
ethanal has a low boiling point, lower than ethanoic acid, due to not having an O-H bond therefore cannot form hydrogen bonding
ethanal is taken away from the mixture so cannot be oxidised further.
explain why the production of carbon monoxide and soot is hazardous
CO is toxic because it binds to haemoglobin and limits the ability of blood to transport oxygen around the body
soot particles are toxic because particulates enter the body through airways and damage the lungs
explain why nitrogen monoxide is formed in car engines but is not formed at room temperature and pressure
in car engines there is a high temperature therefore a greater fraction of nitrogen and oxygen molecules collide with energy greater than the high activation energy leading to a higher frequency of successful collisions and a faster rate of reaction
nitrogen monoxide is also formed in aircraft engines which is released into the atmosphere. explain how this leads to the formation of acid rain using chemical equations to support your answer.
2NO + O₂ → 2NO₂
4NO₂ + O₂ + 2H₂O → 4HNO₃
why is cracking important in industry?
it produces more petrol
short chain alkanes are more useful products
demand is greater for shorter chain alkanes
it produces ethane/short chain alkenes for making poly(ethene)
smaller alkanes give less pollution/burn more efficiently
recycles waste products
as a source of hydrogen
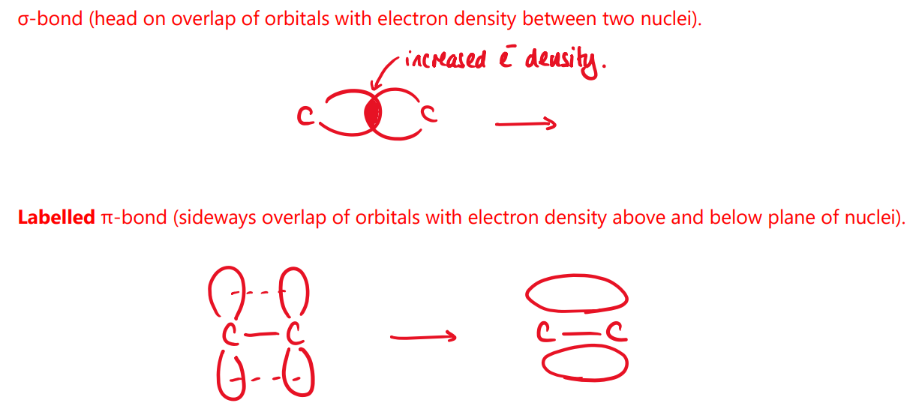
the alkene funntional group consists of a C=C bond. this bond contains two different types of covalent bond. draw an annotated diagram to show how there two different types of covalent bond are formed in this functional group.
sigma bond - head-on overlap of s orbitals with electron density between two nuclei
pi bond - sideways overlap of p orbitals with electron density above and below plane of nuclei
what does a positive Fehling’s solution test signify?
blue solution to brick-red ppt (Cu2O)
aldehyde - CHO present
what does steamy fumes and vigorous effervescence with PCl5 imply?
OH present (alcohol or carboxylic acid)
what does no reaction with hot acidified potassium dichromate (VI) solution signify?
tertiary alcohol or carboxylic acid
give a possible reason why HCl gas forms steamy fumes when it mixes with moist air
droplets of HCl form
explain why poly(but-1-ene) is considered to be unsustainable. describe ways that addition polymers such as poly(but-1-ene) can be disposed
but-1-ene is derived from crude oil, which is a finite, non-renewable resource
methods of disposal include:
incineration to provide energy
cracking polymers to produce useful chemical feedstock
sorting (with an IR camera) and recycling
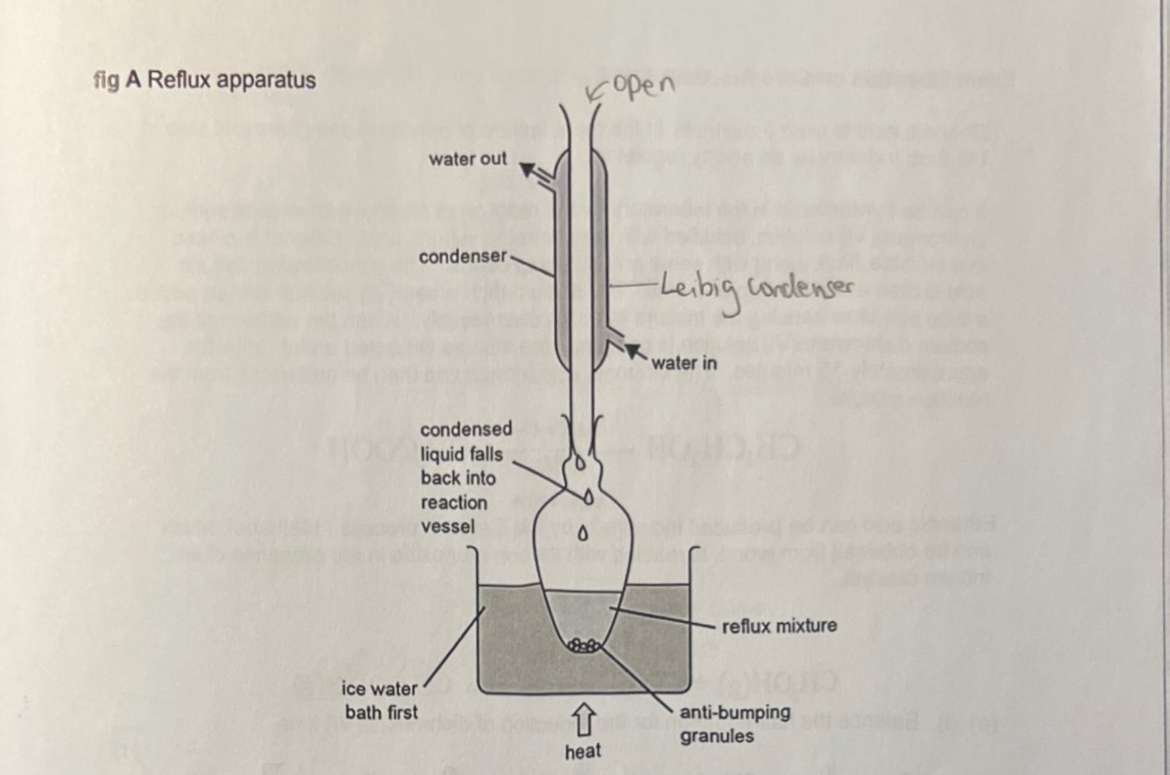
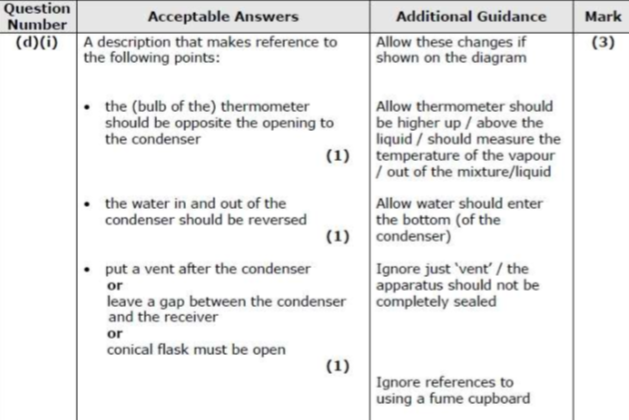
draw and annotate the apparatus used for reflux
continual boiling and condensing of reaction mixture to ensure the reaction takes place without the contents of the flask boiling dry
for prolonged, intense heating
cold water in the condenser
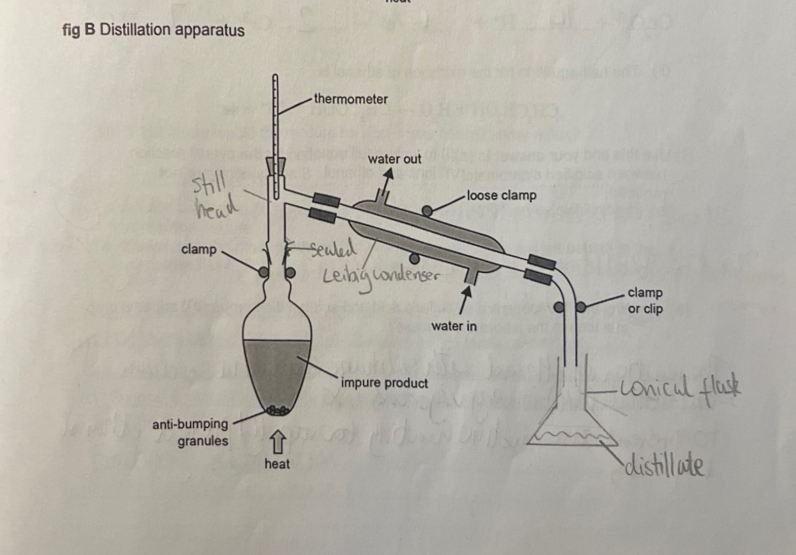
draw and annotate the apparatus used for distillation
contents of flask are heated and react then boil off and condense into a separate vessel
distillation is for shorter periods of gentler heating
cold water in the condenser
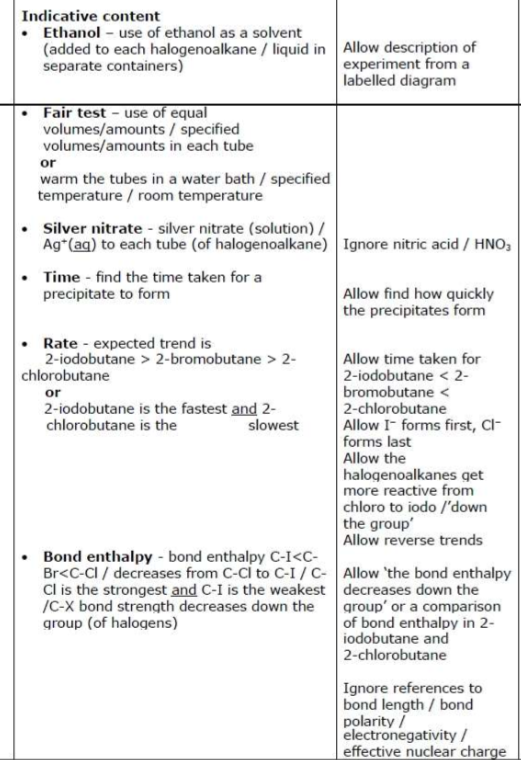
core practical 4: devise an experiment to compare the rates of the hydrolysis of 2-chlorobutane, 2-bromobutane and 2-iodobutane.
state the trend in the rates of reaction. justify your answer. (6)
suggest two ways you could improve the accuracy and one way to improve the reliability of your results
water bath at 50°C
improve accuracy by:
using colder water in water bath so it takes a longer time for the precipitate to form, so there is a smaller percentage uncertainty in the measurement of time
use a more accurate timer (more highly callibrated) as this will reduce percentage uncertainty in time measurement
improve reliability by…
repeating the measurement of time three times for each halogenoalkane and take a mean of the time values for each halogenoalkane
when is steam distillation used?
for more temperature sensitive molecules
state the purpose of adding ethanol to each of the test tubes when hydrolysis halogenoalkanes. (1)
ethanol is added to dissolve both the halogenoalkane and water
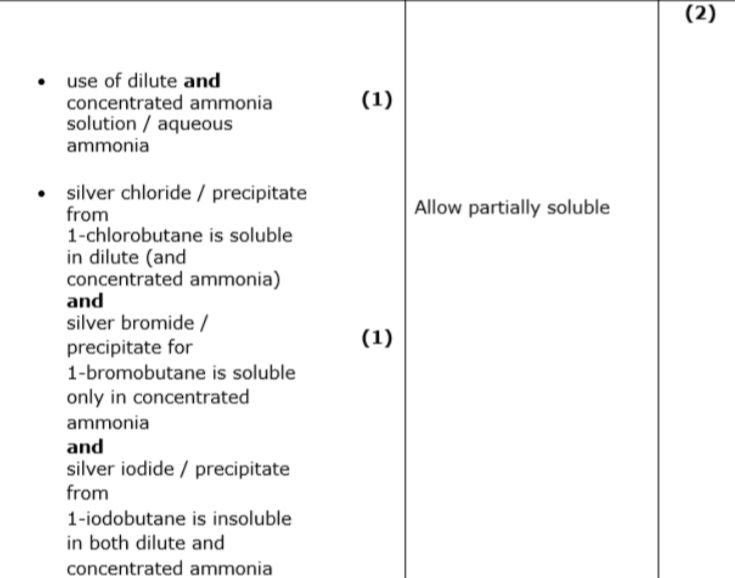
identify further reagents that can be added, including relevant observations, to confirm the identity of the halogen atom present in each halogenoalkane. (2)


core practical 5: the oxidation of ethanol
why are the concentrated sulphuric acid and sodium dichromate (VI) added a drop at a time in the laboratory process? (1)
to prevent the mixture heating too rapidly/to prevent ethanol evaporating

they have a (very) high boiling temperature/are ionic compounds

(oxidation of ethanol using concentrated sulphuric acid and sodium dichromate (VI)


cool the mixture because the reaction with concentrated sulphuric acid is very exothermic/releases a lot of heat
anti-bumping granules are added to prevent violent boiling OR promote smooth boiling OR to provide nucleation centres for bubble formation OR to prevent superheating
heating under reflux is used to prevent the loss of any volatile substances

brown vapour is Br2
bromide ions are oxidised by concentrated sulphuric acid
bromoethane was prepared from the reaction of ethanol with sulphuric acid and potassium bromide.
suggest why the flask containing the ethanol was frequently placed in a stream of cold running water as the concentrated sulphuric acid was gradually added. (1)
ethanol would evaporate/is volatile/has a low boiling temperature
bromoethane was prepared from the reaction of ethanol with sulphuric acid and potassium bromide.
the potassium bromide was initially lumpy and not a fine powder. state the apparatus that would be suitable for breaking up the lumps of potassium bromide into a powder. (1)
a pestle and mortar
bromoethane was prepared from the reaction of ethanol with sulphuric acid and potassium bromide.
the round-bottomed flask contains ethanol, concentrated sulphuric acid and potassium bromide.
explain why an orange colour was seen in the round-bottomed flask when it was first gently heated. (2)
orange colour is Br2
that is formed by the oxidation of bromide ions by sulphuric acid