Orgo 2 Ch14 - IR Spectroscopy, Mass Spectrometry
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
16 Terms
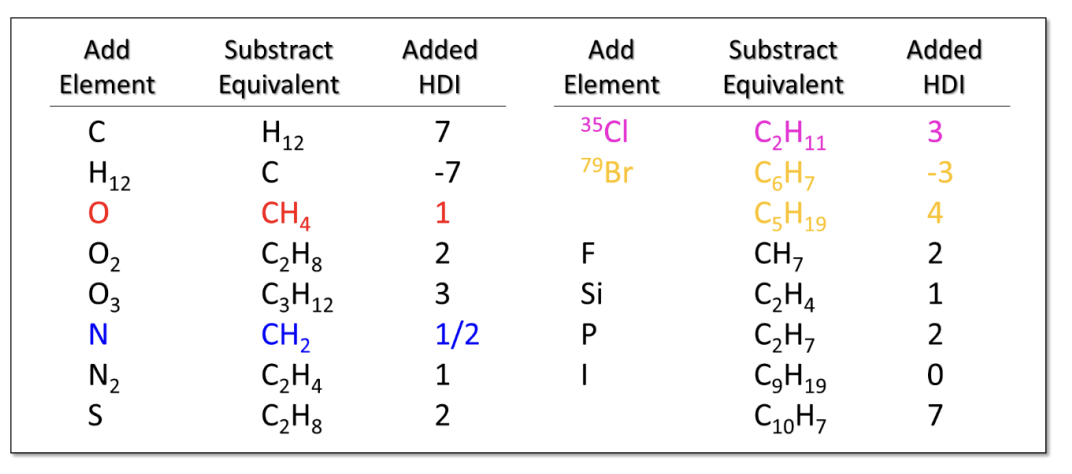
IR Region #1: 3,000 cm-1 C-H Bonds
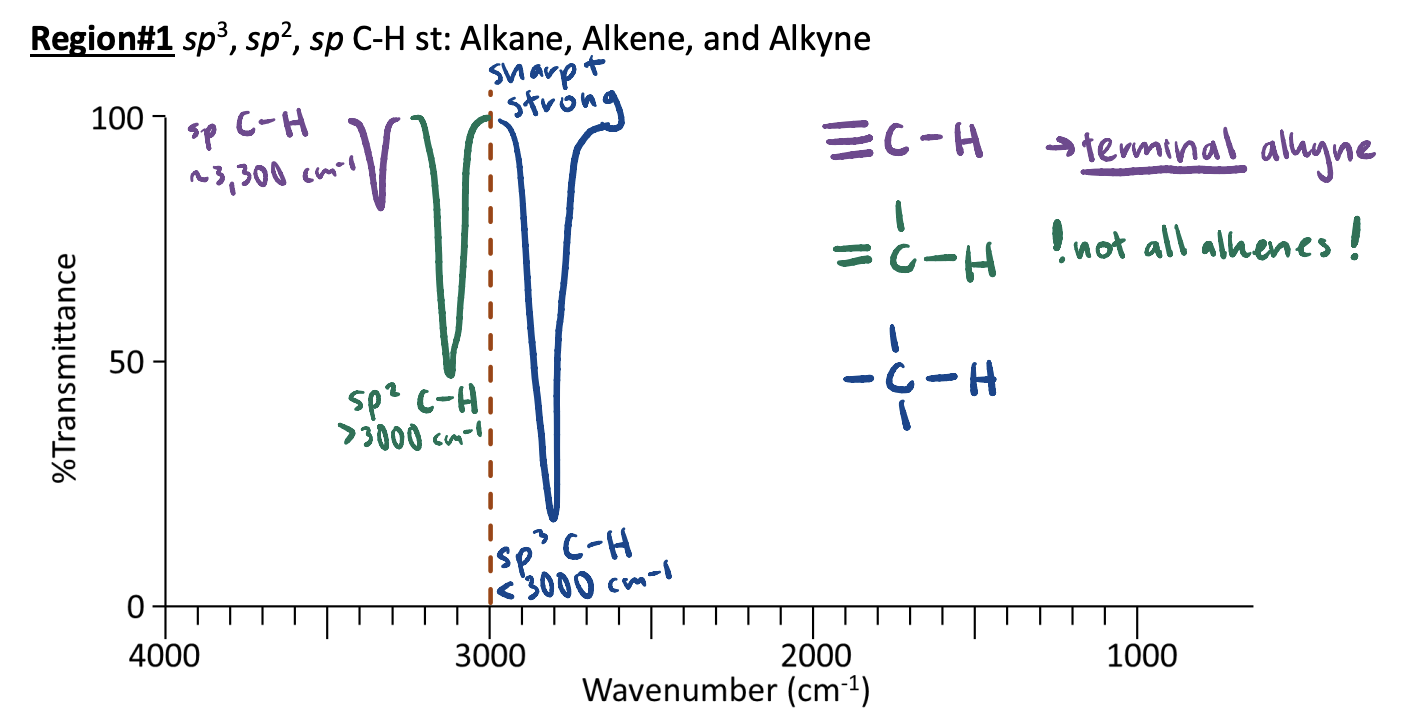
IR Region #2: 3,000 cm-1 O-H and N-H Bonds
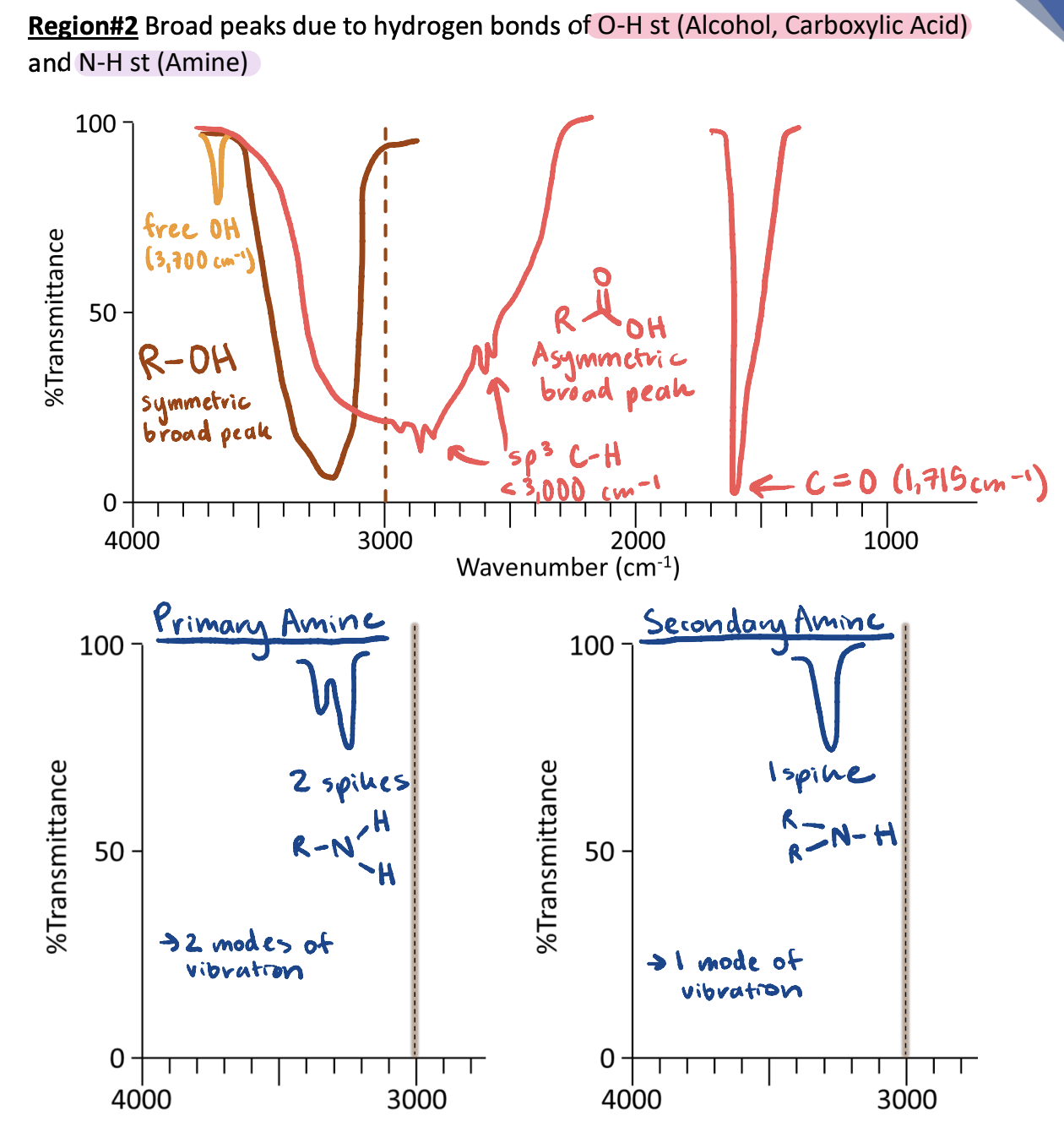
IR Region #3: 1,700 cm-1 Carbonyl C=O and Alkene C=C Bonds
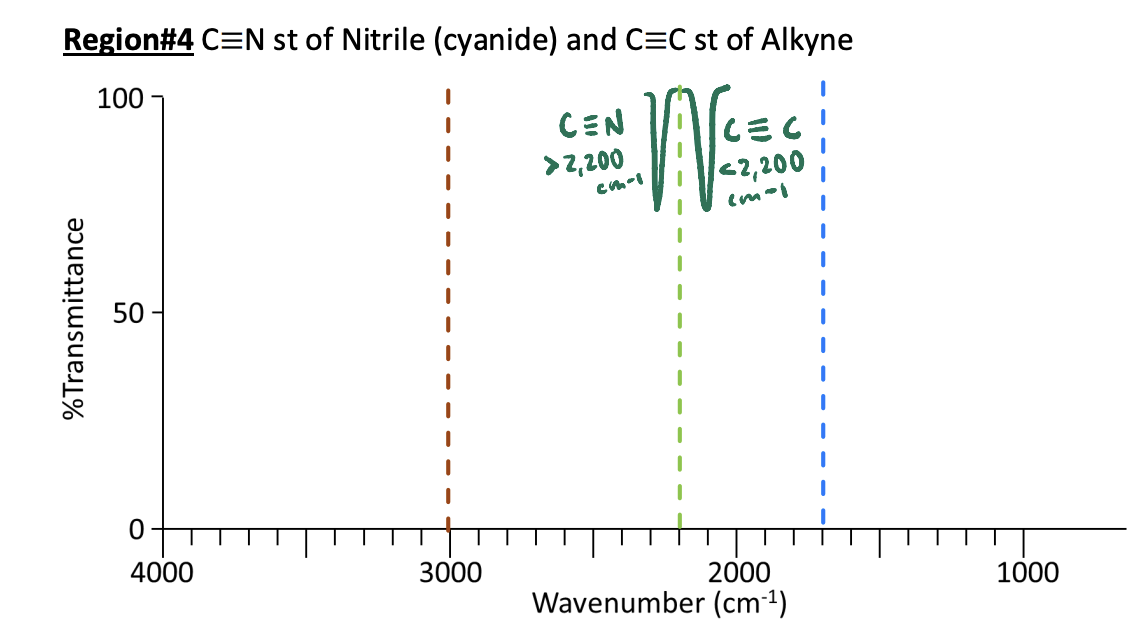
IR Region #4: 2,200 cm-1 Nitrile C≡N and Alkyne C≡C Bonds
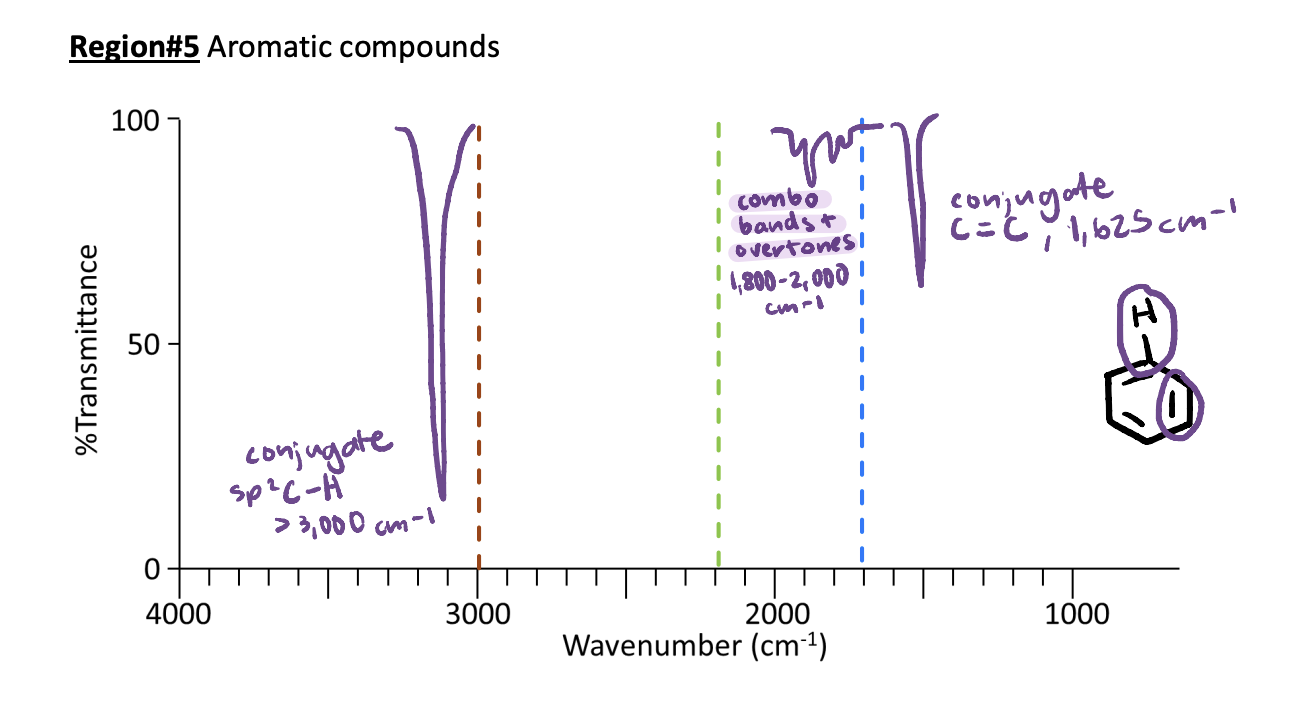
IR Region #5: Aromatic Compounds
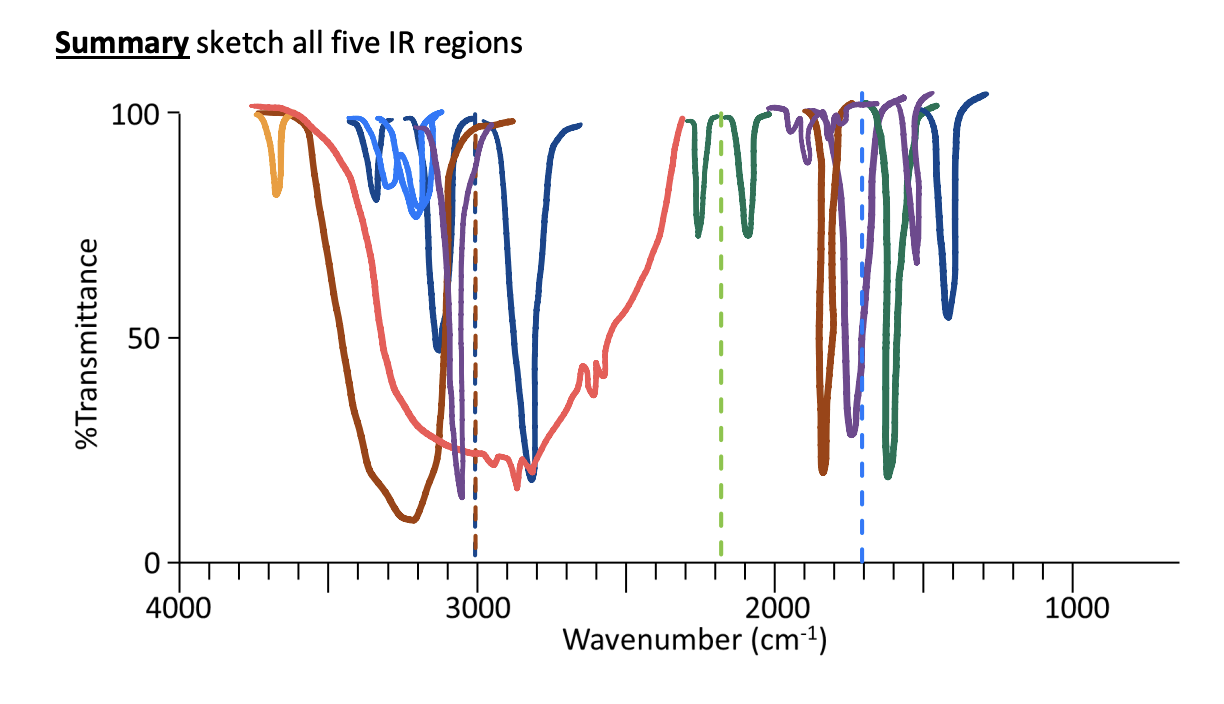
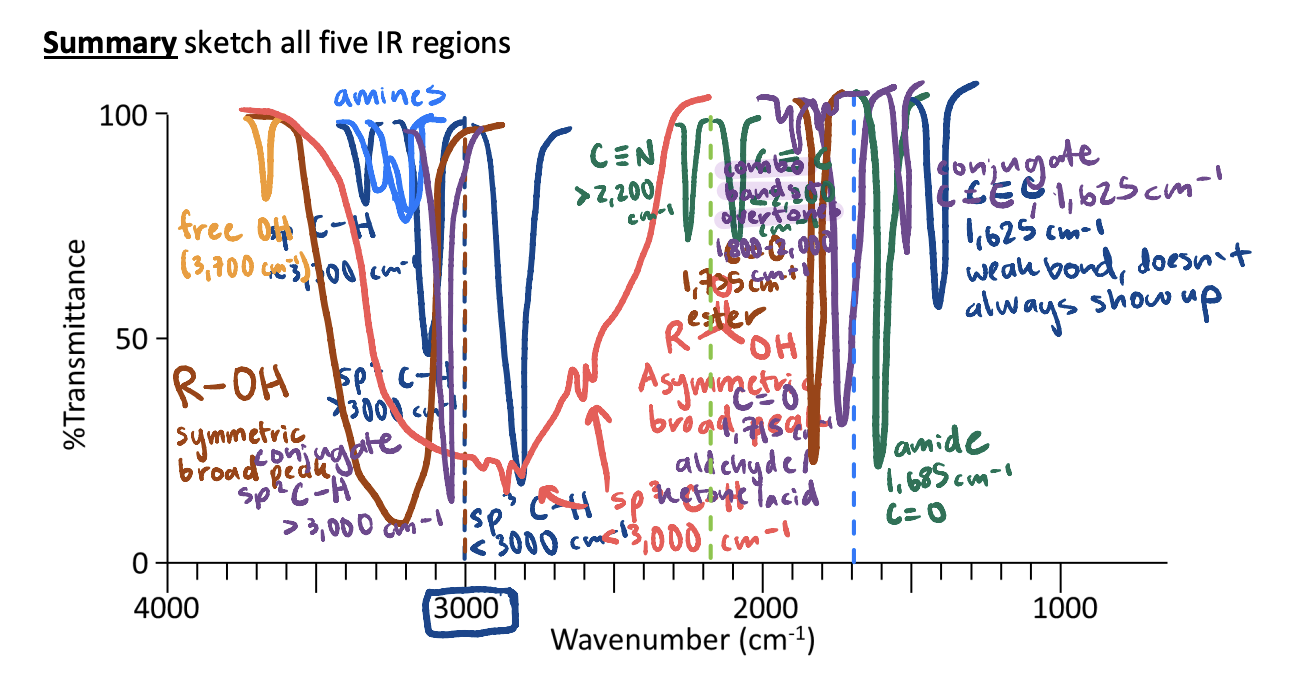
All IR Regions
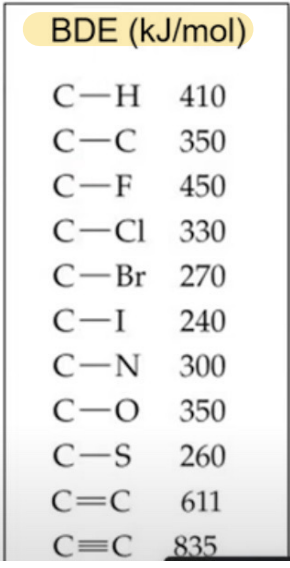
Common Bond Energy
Molecular Ion (M+)
Heaviest peak whose radical cation corresponds to weight of intact molecule. NOT always base peak.
Base Peak and Relative Abundance
Peak with highest relative abundance - reference measurement for % abundance (base peak is 100% relative abundance).
Favors stable radical species.
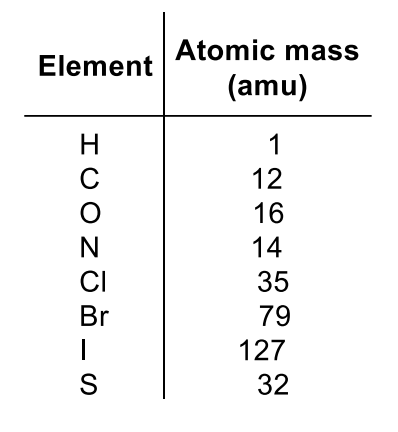
Isotope Split Peaks
Br: M+ and M+2 (~50%)
Cl: M+ and M+2 (~25%)
C: M+ and M+1 (~1%)
S: M+, M+1 (~1%) and M+2 (~4%)
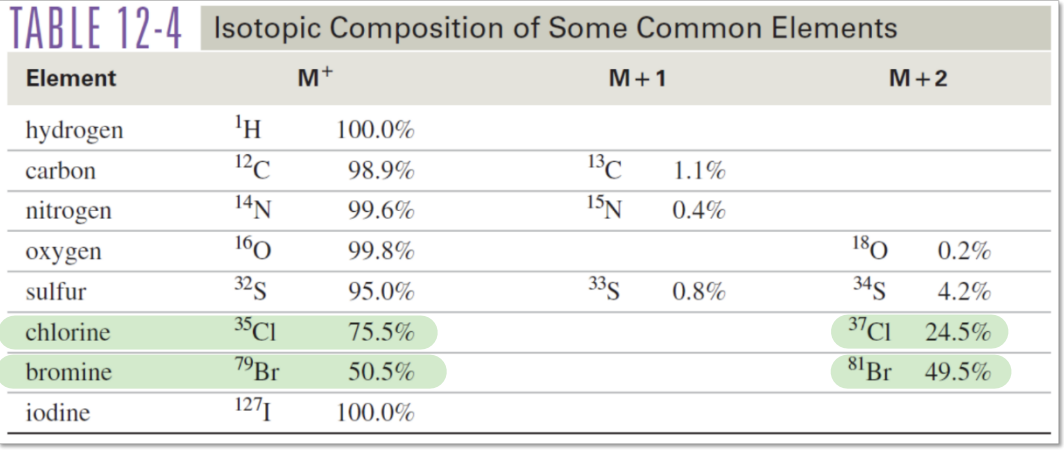
Common Atomic Masses
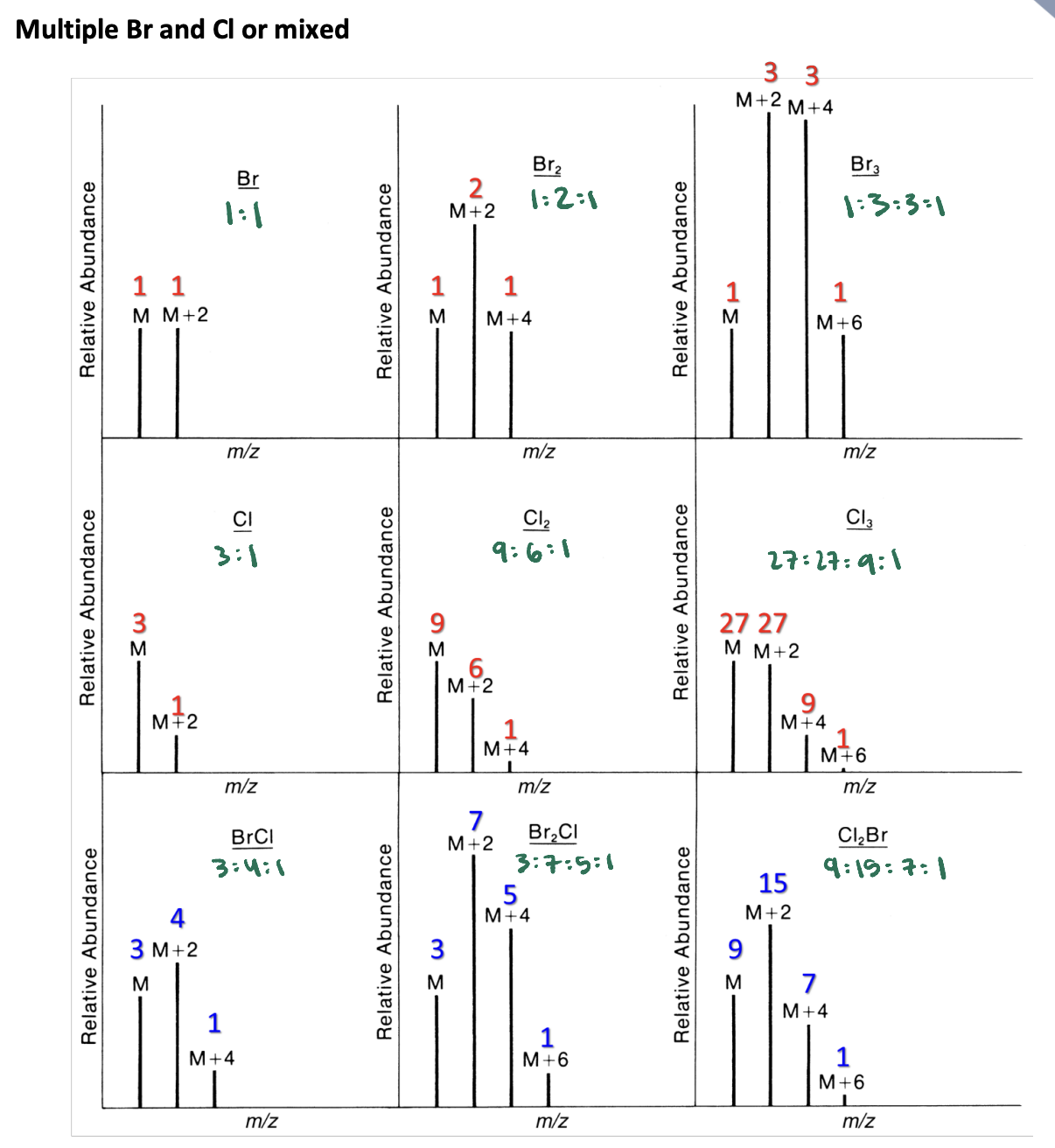
Relative Abundance Ratios of Br and Cl
Rule of 13: M+ Weight to Base Formula
#C = (M+)//13
#H = #C + (M+)%13
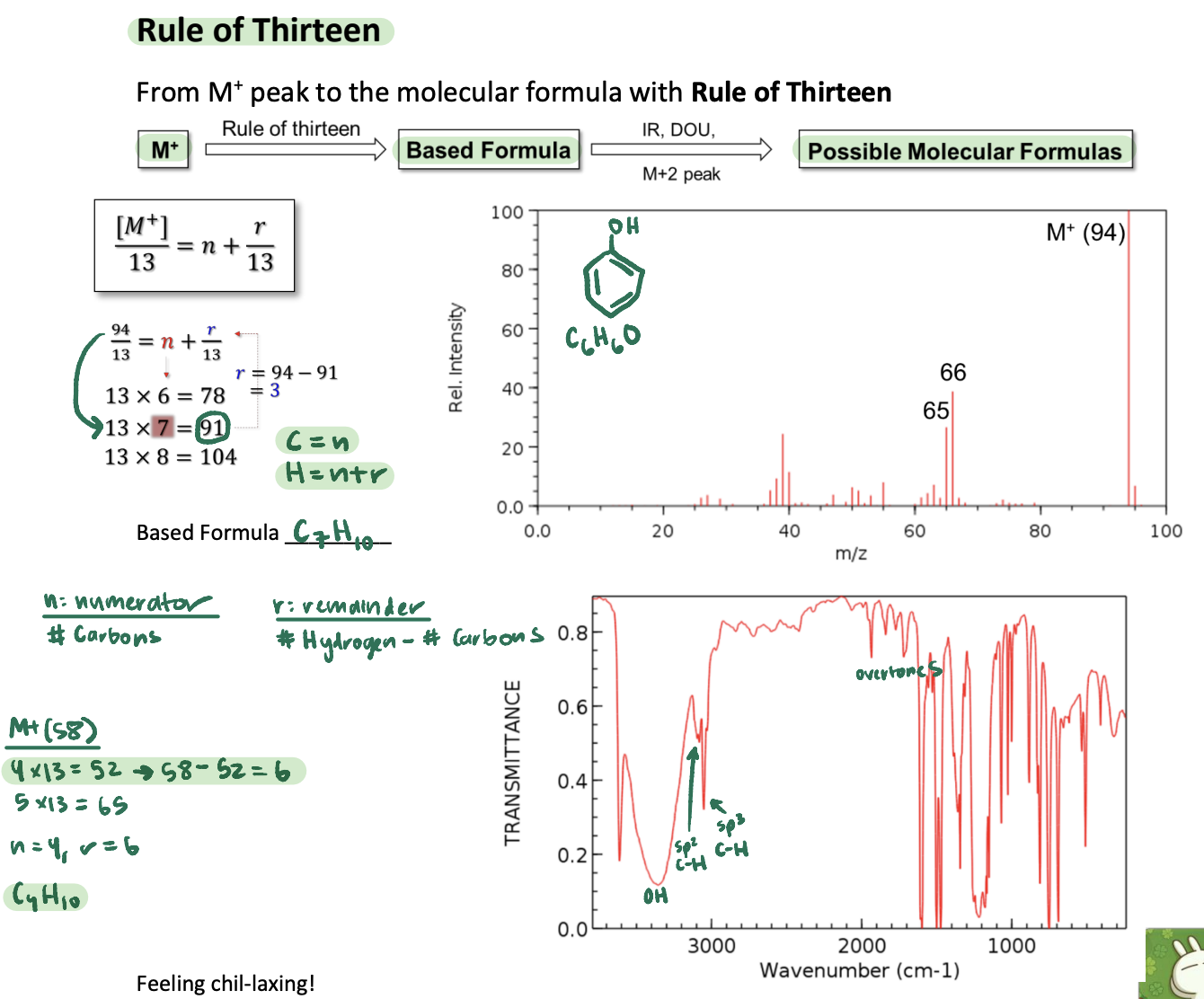
Base Formula to Alternate Formulas - Substituents