female repro phys
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
25 Terms
what ovarian structures produce estrogen?
estrogen production requires coordination of both theca and granulosa cells
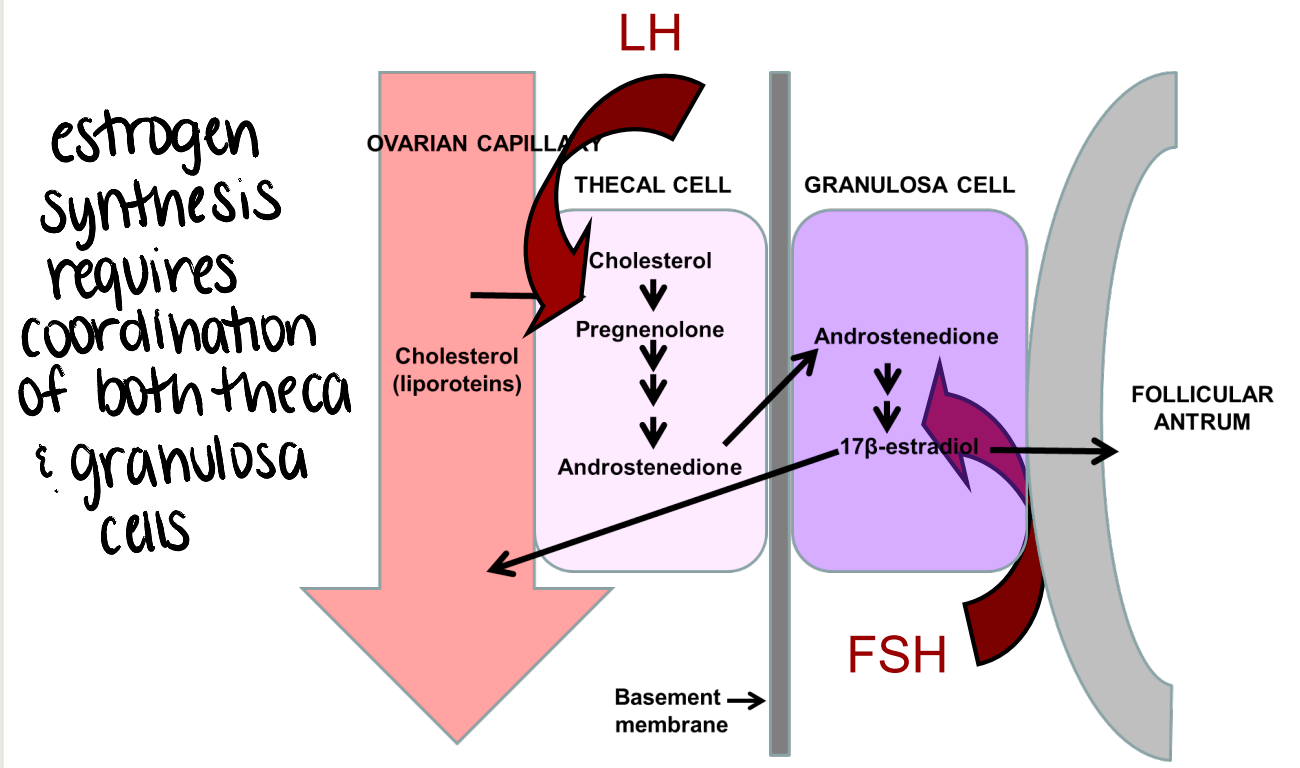
what ovarian structure produces progesterone?
luteal cells (corpus luteum)
gonadotropin-independent follicular development
from primordial follicle up to secondary follicle
can occur at any time, including before birth
if insufficient gonadotropin signaling is present, follicles will undergo atresia
no gonadotropin before puberty → secondary follicle = most developed follicle possible pre-puberty
cohort/wave of primordial follicles is recruited in a cycle
gonadotropin-dependent follicular development
secondary follicle → mature/graafian follicle
follicular phase of the estrous cycle
only possible after puberty, during specific periods of the estrous cycle
what controls development during the gonadotropin-independent stage?
local growth factors
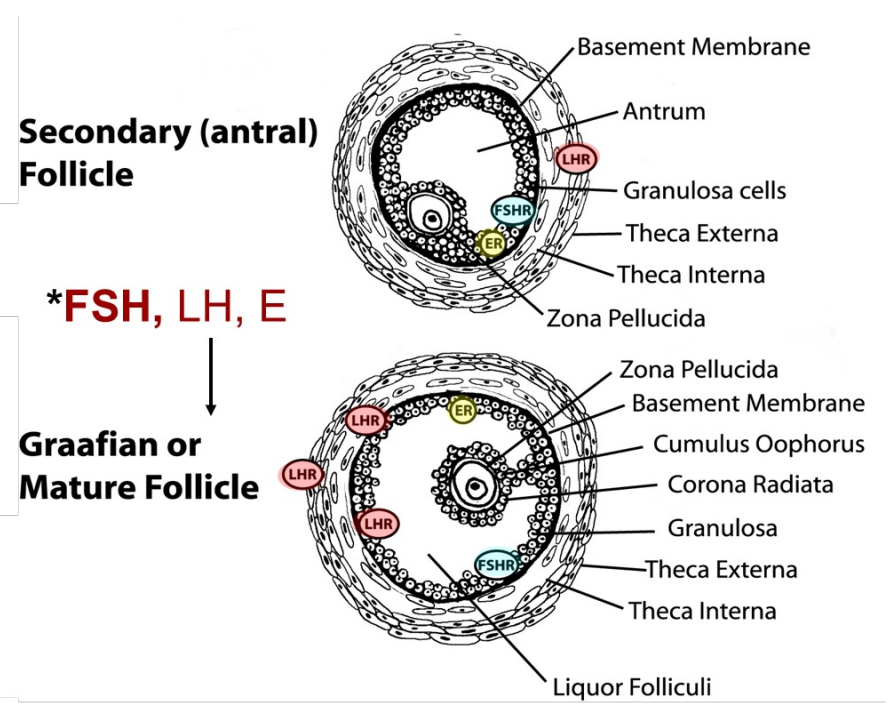
receptors in secondary vs. mature follicle
secondary: contains LH, FSH, and estrogen receptors
LH receptors only on theca cells (more external cells)
mature follicle: contains LH, FSH, and estrogen receptors
LH receptors on both theca and granulosa cells (allow mature follicle to respond to LH surge)
FSH and estrogen receptors on granulosa cells (inner cells) in both stages
what are the general effects of estrogen during the follicular phase?
preparing for mating & fertilization
follicle/gamete maturation
induce ovulation
open cervix
contraction of oviduct to move fertilized eggs along
environmental changes in uterus and vagina to increase survival and transport of sperm
ex. cornification of vaginal cells,
behavior: estrus (receptive for mating)
-/+ feedback on HPG aixs
how does the dominant follicle suppress growth of the rest of the cohort?
dominant follicle develops faster and becomes independent of FSH → secretes inhibin and estrogen → negative feedback suppresses FSH and deprives other follicles of FSH → other follicles undergo atresia
(“kick down the ladder”)
superovulation
rescue the rest of the follicles in the cohort before they undergo atresia at the gonadotropin-dependent stage by providing FSH
allows multiple “dominant” follicles for LH surge
what effects does the LH surge have on mature follicle(s)?
permits continued oocyte maturation
resumes meiosis, then arrests again → formation of secondary oocyte
cytoplasmic maturation
triggers ovulation
initiates formation of corpus luteum
what is LH’s effect on corpus luteum?
LH works on both small and large luteal cells to increase progesterone production; some LH required for function of corpus luteum
luteotrophic hormones
LH (& prolactin — in rodents and dogs)
relative importance of luteotrophic hormones varies with species
regulate function of corpus luteum
note: progesterone is a product of CL → not a luteotrophic hormone
metestrus
stage of the luteal phase in which the corpus luteum is not fully functioning (corpus hemorrhagicum)
diestrus
stage of the luteal phase in which the corpus luteum is fully functional
what hormones are produced by the corpus luteum?
progesterone (dominant)
estrogens
relaxin
what are the general effects of progesterone during the luteal phase?
coordinating physiology for potential pregnancy
reduce contraction of oviduct
uterus: increase nutrition secretion, suppress immune system
close cervix
inhibit estrogen actions in vagina (make inhospitable for sperm)
behavior: stop estrus behaviors
HPG axis: only negative feedback
why must luteolysis occur before the next wave of follicles can mature?
CL produces progesterone (P4) → suppresses HPG axis
apoptosis of luteal cells → rapid loss of P4 → HPG axis negative feedback removed → FSH/LH increases → maturation of next wave of follicles
what triggers luteolysis?
PGF-2a (most species)
two different sources:
uterus (ruminants; other species)
corpus luteum due to decreased LH (primates)
programmed demise (dog)
CL dies of old age
how is ovulation induced in spontaneous ovulators?
high levels of estrogen in late follicular phase cause GnRH/LH surge, which triggers ovulation
induced/reflex ovulators
no positive feedback of estrogen
GnRH/LH surge triggered by cervical stimulation
what advantages do induced/reflex ovulators have?
no luteal phase if not mated → do not waste time with luteal phase; can start developing new follicles sooner
ovulation timed with arrival of sperm
control of corpus luteum formation
option 1: most spontaneous ovulators → fully functional CL
luteotropic support (i.e. LH) is adequate regardless of a mating stimulus (e.g. ruminants, primates, equids)
option 2: rodents → poorly functioning CL if not mated (need LH + mating-induced PRL)
what is prolactin’s (PRL) role in corpus luteum formation (rodents)?
cervical stimulation causes reflex release of PRL
PRL inhibits conversion of progesterone to a weaker form
in the absence of mating, corpus luteum produces weak progesterone
PGF2a
prostaglandin produced by the uterus or corpus luteum responsible for destruction of corpus luteum (luteolysis)
control of luteolysis
uterus secretes PGF2a
ruminants: special relationship between uterine vein and ovarian artery
horse: release into systemic circulation
decreased LH (primates)
uterus not involved; CL produces PGF2a
functional decrease in LH availability → increases PGF2a in CL (self-destruct)
programmed demise (in canids, CL dies of old age)
lifespan of CL in non-pregnant and pregnant females is almost the same