Section D: Earth's Atmosphere
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
what is the atmosphere
a gaseous envelope surround earth, a series of concentric layers
how is the atmosphere held to the earth
by gravity
where is most of the atmosphere’s mass
near the surface
what is weather related to the atmosphere
fluid properties that are constantly changing spatially and temporally
how is constant (permanent) gas distributed in the atmosphere
more or less same proportion in the lower atmosphere
what is the constant (permanent) gas made up of
78% nitrogen, 21% oxygen, 1% argon
how does variable (trace) gases differ
differ in proportion over time and space
what are variable (trace) gases made up of and how much
they make up less than 1%: water vapour, co2, ozone, helium, neon, methane
why is water vapour important and how does it affect temperature
it determines the humidity and is a source of clouds/precipitation
what is the most important greenhouse gas and why
water vapour because it absorbs and stores heat energy (movement of energy)
how much does the percentage of water vapour vary
<1% to 4% by volume (avg 2%)
what does the variation of water vapour depend on
air temperature (warmer air holds more water vapour than cooler air) and proximity to large bodies of water
what is global distribution and variation
average amount of water vapour in a column of atmosphere in a given month (cm)
what are the global distribution and variation patterns
seasonal (summer/winter) and land/water contrasts (especially in winter)
greenhouse effect
trapping of longwave radiation (warms the atmosphere which warms the earth)
what is earth’s temperature
15°
photosynthesis
plants absorb co2 and release oxygen and water as a by-product
what is carbon dioxide’s climatic influence
0.04%
what would happen without water vapour in the atmosphere
the earth would be -15°C because there wouldn’t be anything to trap the heat
what are co2 sources examples
volcanic eruptions, decay of living matter, respiration, burning of fossil fuels (coal, oil, natural gas)
what are co2 sinks examples
chemical reactions with rocks, oceans, plants, soil
by how much do co2 concentrations increase every year
2 parts per million every year
particulates/aerosols
minute suspended particles in the atmosphere, existing in both solid and liquid form
examples of solid particulates
snow, hail, pollutants, soil (dust), smoke, ash, pollen grains, sea salt, bacteria
examples of liquid particulates
clouds and rain
how do particulates help precipitation
they form by acting as condensation nuclei, they are tiny surfaces for water vapour to condense around
hygroscopic
absorb water, water loving
how do particulates affect temperature
they absorb and reflect energy
how do particulates influence air quality
they affect public health and those with copd or asthma
how do dust storms play a role in southern alaska
they supply nutrients (iron) into the gulf of alaska and provide fuel for phytoplankton blooms which are the foundation of the aquatic food web
what are the thermal properties of the atmosphere
troposphere, stratosphere, mesosphere, thermosphere
what are the compositions of the atmosphere
homosphere, heterosphere
what are the function properties of the atmosphere
ozone layer
how are the thermal layers divided
by temperature changes and gases, in density and composition, and sphere and pause
when we say “sphere” what is that including
the entire layer
when we say “pause” what is that including
the boundary between 2 layers
what is the lowest thermal layer
the troposphere
what is the most active zone and why
the troposphere because there is lots of movement from weather and vertical mixing in and out of the atmosphere
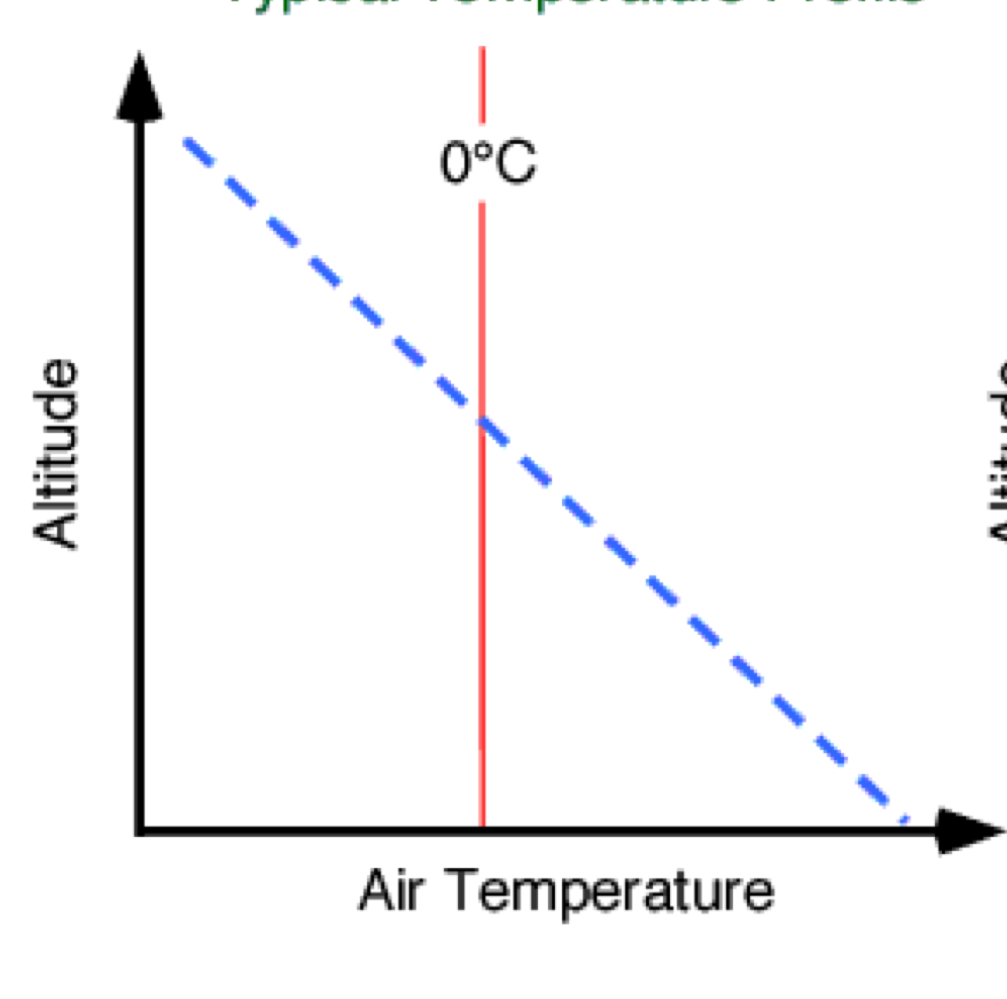
what happens to the temperature in the troposphere
the temperature decreases with increasing altitude
what is the environmental lapse rate
the temperature decreases at an average rate of 6.5°C per 1000m or 1km
what is the tropopause and what is it’s thickness
the upper limit of the troposphere, 8-18km
why does the thickness of the tropopause vary
it’s higher at the equator because the hotter air rises and expands and causes the equatorial bulging
what is temperature inversion
the reversal of normal temperature pattern so that air temperature increases with altitude in the troposphere
normal or temperature inversion
normal
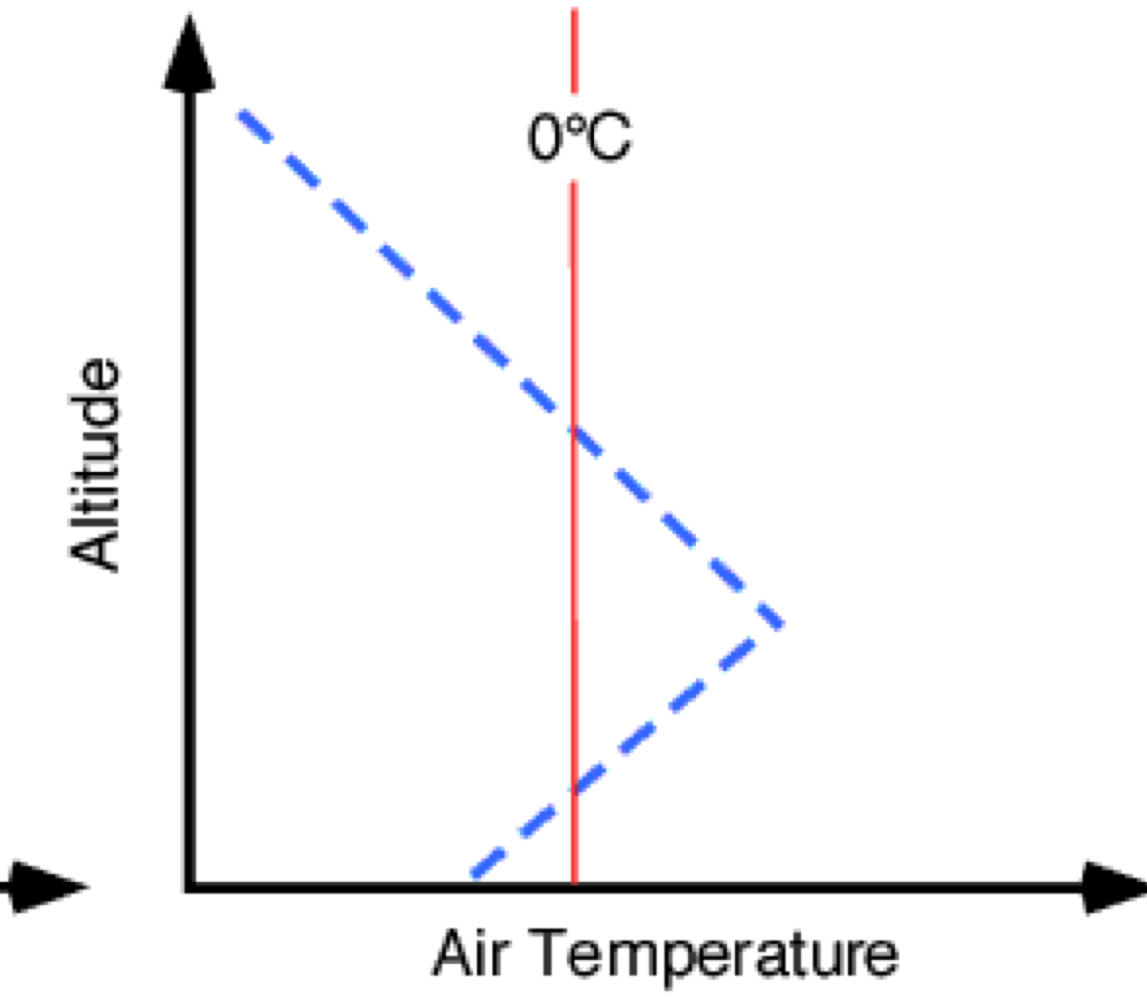
normal or temperature inversion
temperature inversion
where does the stratosphere reside
immediately above the tropopause
what does the stratosphere contain
the ozone layer
what is the general range of the stratosphere
12-50km altitude
what is the ozone in the stratosphere compared to in the troposphere
10 ppmv vs 0.04
how does the temperature change in the stratosphere and why
the temperature increases with increasing altitude because of the ozone absorbing UV rays
what is the upper limit of the stratosphere
the stratopause
where do commercial jets fly and why
in the stratosphere where there is less turbulence because of little vertical mixing
where does the mesosphere reside
immediately above the stratosphere
what is the coldest atmospheric layer and how cold can it get
the mesosphere, upper limit = -100°C
what happens with the temperature in the mesosphere
temperature decreases with increasing altitude
what happens in the mesosphere
solar radiation reduces gas molecules to ions (strips an electron to form a positively charged ion) which disrupts some satellite communication
what is the uppermost layer
the thermosphere
what happens to the temperature in the thermosphere
temperature increases with altitude
why is it so hot in the thermosphere
intense solar reactions cause molecules to vibrate at very high speeds which creates kinetic energy
which layer does the international space station orbit in
the thermosphere
how are the gases sorted in the thermosphere
into a variety of sublayers based on molecular mass
what is the density like in the thermosphere
low density
how are the northern and southern lights created and where
high energy particles emitted from solar wind interact with earth’s magnetic field and emit photons of light. found mostly near the poles where earth’s magnetic field concentrates solar wind particles
homosphere
the inner atmosphere where there is a uniform distribution of gases (even mixing)
heterosphere
the outer atmosphere (thermosphere) where the gases are sorted into layers according to molecular weight
where is ozone found and its role
stratosphere - the uv absorbing ozone layer, and the troposphere - in the form of atmospheric pollution (toxic to plants and people)
stratospheric ozone steps
uv radiation is absorbed by O2 which creates free oxygen
free oxygen combines with O2 to form O3
tropospheric ozone
the interaction of sunlight and volatile organic compounds and nitrogen oxides (NOx)
chlorofluorocarbons
anthropogenic pollutants - synthetic chemical
how do CFCs affect ozone
they go into the stratosphere where uv rays break a chlorine atom away from it. it then reacts with O3 and breaks it into O2 and a free oxygen. the free oxygen reacts with the chlorine to form chlorine oxide which damages the ozone layer