Understanding pH in Water Quality
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
23 Terms
pH
Measure of acidity or alkalinity in water.
Acids
Substances that donate protons (H+) in solutions.
Bases
Substances that accept protons (H+) in solutions.
Arrhenius Acids
Produce H+ ions in aqueous solutions.
Arrhenius Bases
Produce OH- ions in aqueous solutions.
Brønsted Acids
Proton donors in acid-base reactions.
Brønsted Bases
Proton acceptors in acid-base reactions.
Lewis Acids
Electron pair acceptors in chemical reactions.
Lewis Bases
Electron pair donors in chemical reactions.
Strong Acids
Ionize completely in solution, e.g., HCl.
Weak Acids
Partially ionize in solution, e.g., CH3COOH.
Multiprotic Acids
Can donate more than one proton.
Water Dissociation
Equilibrium between water, H+, and OH-.
pH Calculation
pH = -log[H+], determines acidity.
Phenolphthalein
Common pH indicator for alkalinity.
Litmus Paper
Paper that changes color with pH.
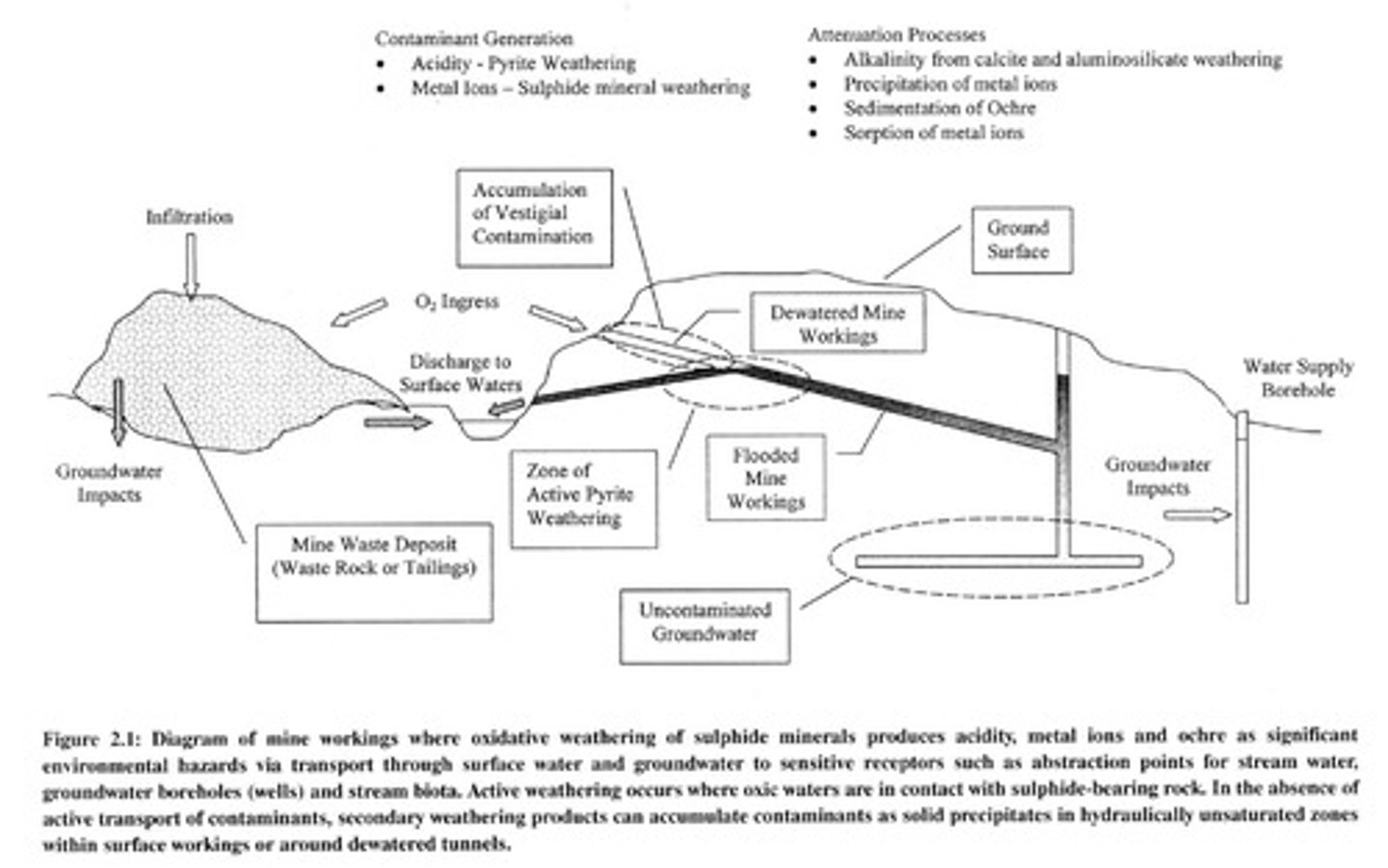
Acid Mine Drainage
Acidic water from mining activities.
Flint Water Crisis
Lead leaching due to low pH water.
Orthophosphate
Used to inhibit corrosion in drinking water.
Disinfection Byproducts
Chemicals formed during water disinfection.
pH Water Quality Criteria
Standards for acceptable pH levels in water.
Activity vs Concentration
Activity accounts for interactions in solution.
Chemical Speciation
Distribution of chemical species in solution.