combustion and pollutants
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
what can the products of combustion be?
complete: CO2 + H2O
incomplete: CO + H2O or C + H2O
which pollutants does an internal combustion engine produce?
NOx, CO, C and unburned hydrocarbons
what is the function of a catalytic converter?
removes toxic gaseous pollutants from internal combustion engines and converts them into less harmful substances (e.g. CO2, water vapour, N)
how is CO2 formed in the atmosphere? what potential problems may this cause?
complete combustion of C in fuel
greenhouse gas causing global warming
how is CO formed in the atmosphere? what potential problems may this cause?
incomplete combustion of C in fuel
toxic
how is C formed in the atmosphere? what potential problems may this cause?
incomplete combustion of C in fuel
blackens buildings, global dimming
how is SO2 formed in the atmosphere? what potential problems may this cause?
combustion of S in fuel (through fuel impurities)
acid rain, air pollution
how are NO and NO2 formed in the atmosphere? what potential problems may this cause?
reaction of N2 in air w/ O2 in air at very high temperatures (e.g. engines/furnaces)
acid rain
what potential problems may unburned fuel cause?
fuel waste, harmful greenhouse gas
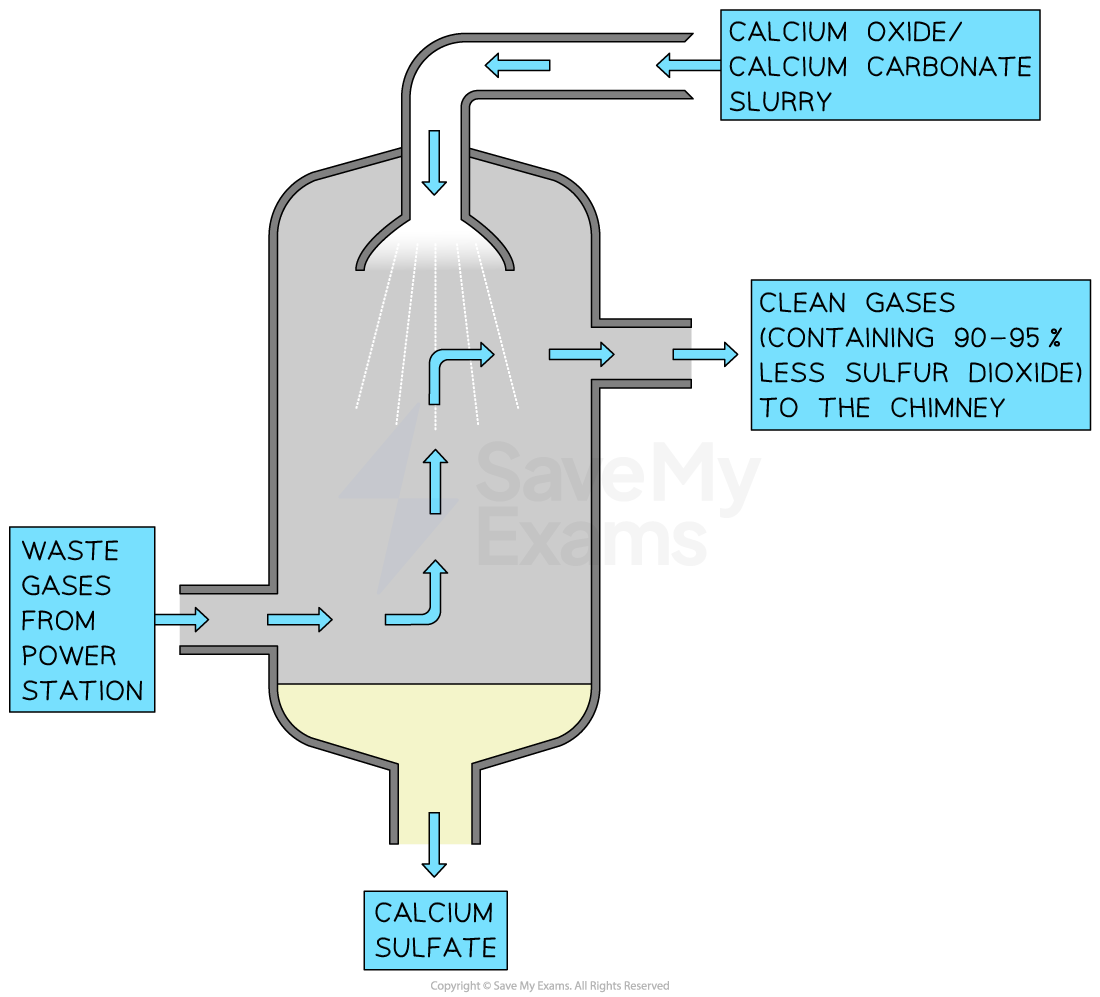
how can we reduce SO2 emissions?
flue gas desulfurisation:
powdered CaO or CaCO3 mixed w/ water to make an alkaline slurry
when flue gases mix w/ alkaline slurry, acidic SO2 gas reacts w/ calcium compounds to form CaSO3, which is harmless