Enthalpy
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Thermodynamics
The study of change in energy/heat
State function
Difference between initial and final condition is independent from the path taken
Endothermic
Takes heat in, reactant
Exothermic
Heat oes out, product
Enthalpy is expression
Delta H
Paths to enthalpy
Calorimetry, Hess’ Law, Heat of formation
Calorimetry equation(s)
0 = q(rxn) + q(cal) + q(sol)
q = mC(delta T)
Hess law
Reactions used to make new reaction
Heat of formation
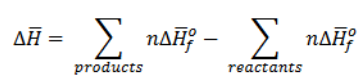
System
Area you are studying
Surroundings
Everything outisde the system
Universe
The system plus the surroundings
Open system
Exchange matter and energy
Closed system
Exchange energy but not matter
Isolated system
Can’t exchange energy or matter
Internal energy equation
Delta U = q + w
Work equation (non reversible)
w = -p(delta V)
Work equation reversible
w = nRTln(V2/V1)
negative q
-delta h, exothermic
positive q
positive delta H, endothermic
Solution cal
deltaH = q(p)
Bomb cal
delta E = q(v)
Calorimetry
heat absorbed/lost in reaction
Heat capactiy
ability to sustain heat raised to temp of 1 k or c
What is work
Work is motion through a distance against some opposing force.