Units 1 & 2 AP BIO
1/15
Earn XP
Description and Tags
Cumulative Exam, May 15
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
Cohesion
Occurs when molecules of the same kind tend to stick together. In water, this is due to hydrogen bond cohesion between water molecules.
Adhesion
on the other hand, is the tendency of dissimilar molecules to be attracted to each other. Adhesive forces can be strong between water and charged molecules and are responsible for… (see next card)
Capillary Action!
Which is the movement of liquid through spaces on its own, sometimes in opposition to gravity. Capillary action is the result of adhesive forces between water and the surface it is touching, which draws the liquid towards it.
Due to cohesive forces, the water also pulls more water molecules behind it. These properties of water are essential to all life on Earth. For example, in plants, capillary action is responsible for moving water from the roots up through the rest of the plant.
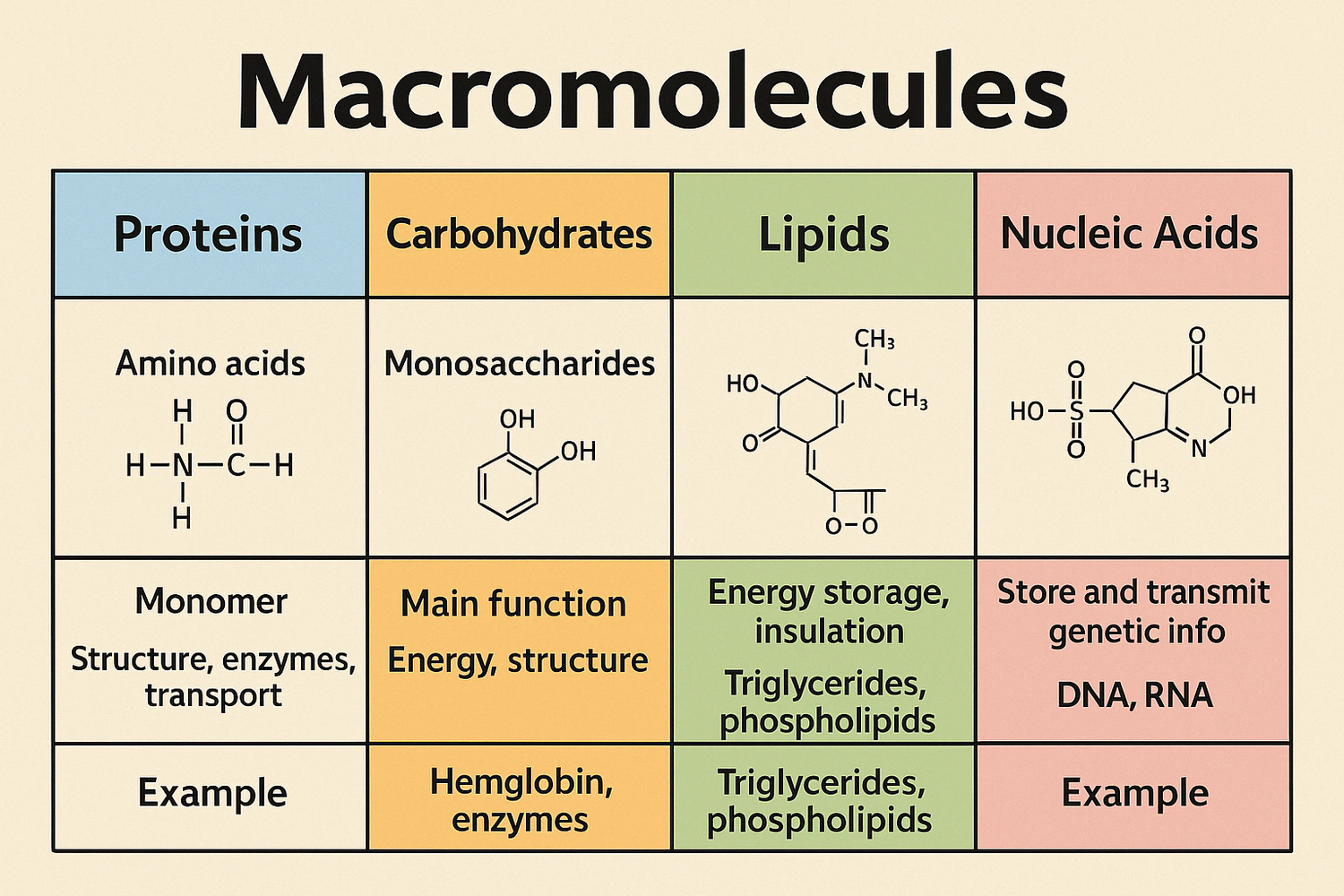
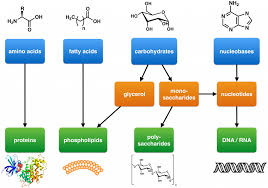
Organic molecules
Carbon, hydrogen, nitrogen, and oxygen, basis of all life on Earth.
Carbon has the unique chemical property of being able to form four bonds with other elements, making it an ideal element to form the backbone of complicated biological molecules. Carbon based molecules are able to take on many configurations, as carbon can form single, double, or triple bonds with other elements. These molecules can take on many shapes: rings, branches, or long chains. Thus, carbon is the elemental basis of the major biological macromolecules: carbohydrates, proteins, lipids, and nucleic acids. In addition to carbon, nucleic acids and proteins rely on nitrogen and phosphorus to build their structure, which we will discuss in more detail below.
Monomers
A molecule that can be bonded to other identical molecules to form a polymer.
Polymers
Large biological molecules like carbohydrates, proteins, and nucleic acids.
Name for large molecules
Can also be called macromolecules!
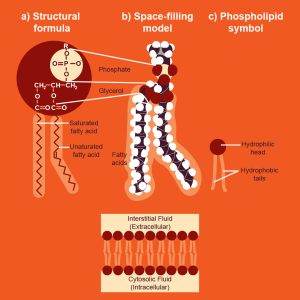
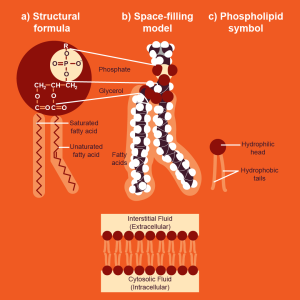
Lipids, on the other hand, are not generally polymers, thus are NOT ALWAYS CONSIDERED MACROMOLECULES!!! Why? Flip!
In the formation of biological macromolecules, the composition and order of monomers affect their function. Also, Lipids are not considered polymers because they are not built from long, repeating chains of identical or similar monomer subunits, unlike carbohydrates, proteins, and nucleic acids.
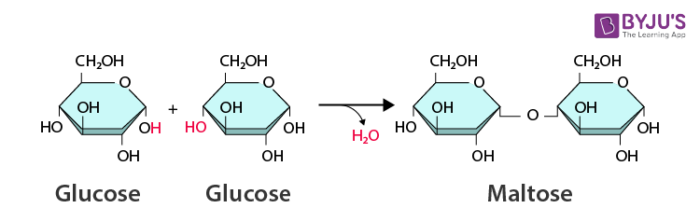
Dehydration Synthesis
The formation of molecules from the action of taking away water.
Hydrolysis
The breaking of molecules with the addition of water! Hiya molecules!
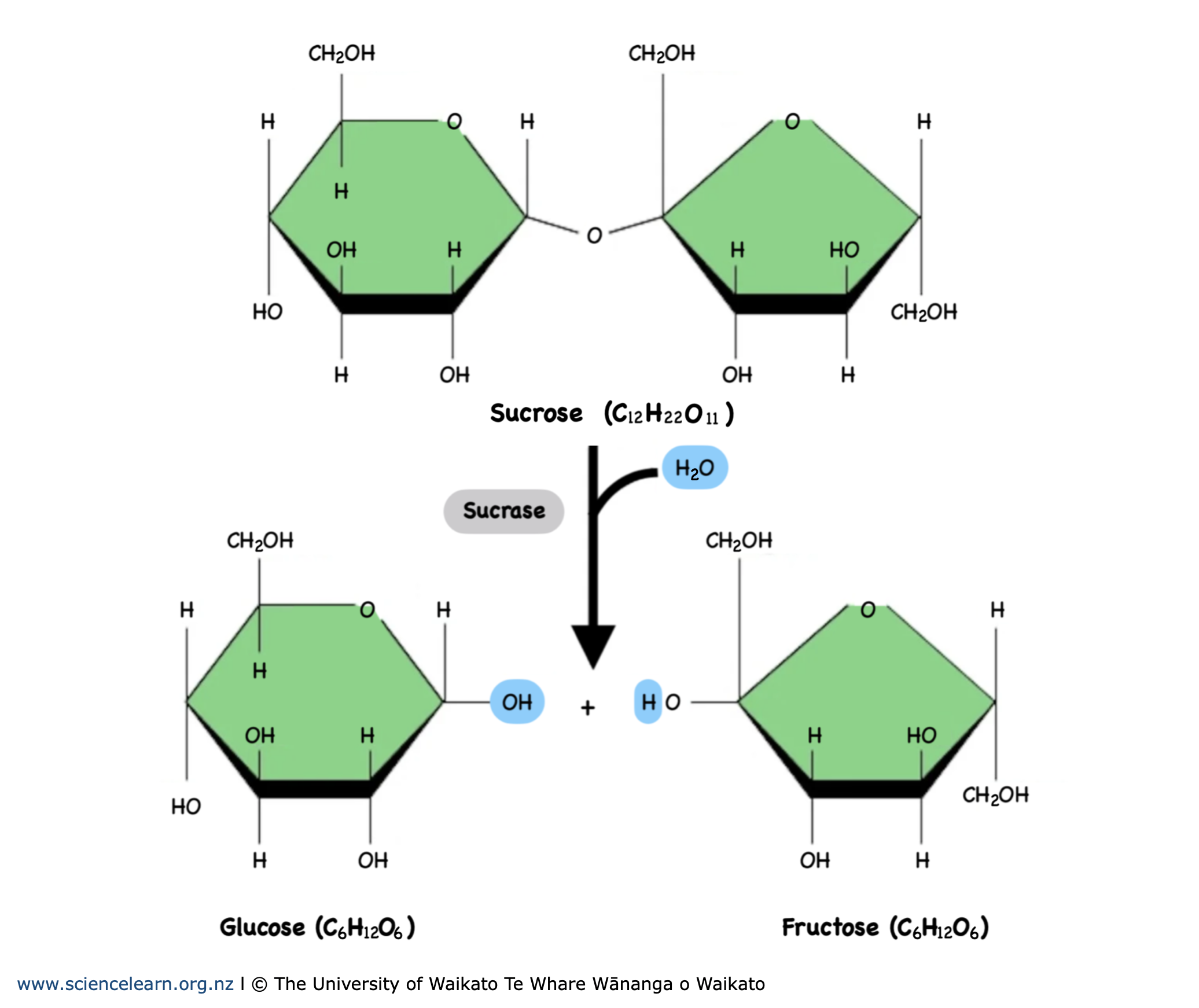
Synthesis reactions generally use energy, which is then stored within the covalent bonds of the macromolecule. When hydrolysis occurs, this energy is released for the cell to use.
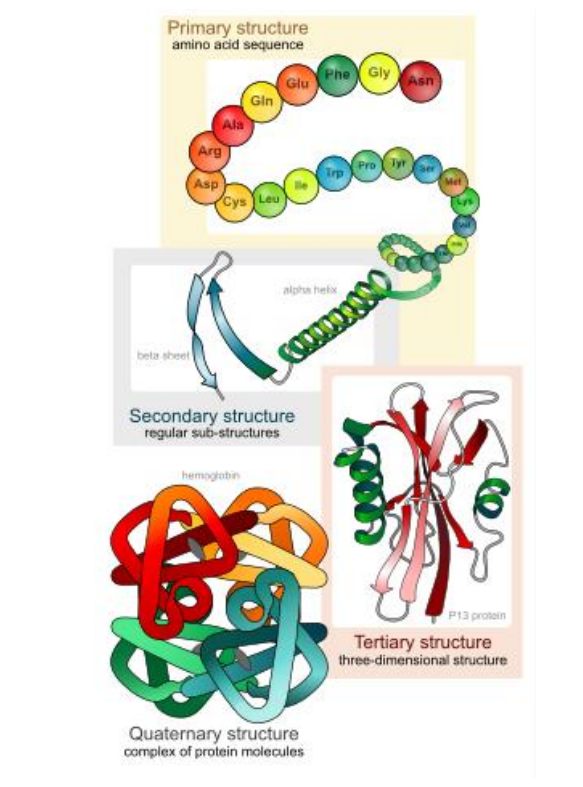
Proteins comprise the majority of organic molecules in organisms and have huge diversity in structures and function. Proteins are made of strings of amino acids connected by covalent bonds. There are 20 types of amino acids in biological organisms, but they all share similar structural features.