Nuclear + Space physics
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
Household fire alarms - uses of radiation
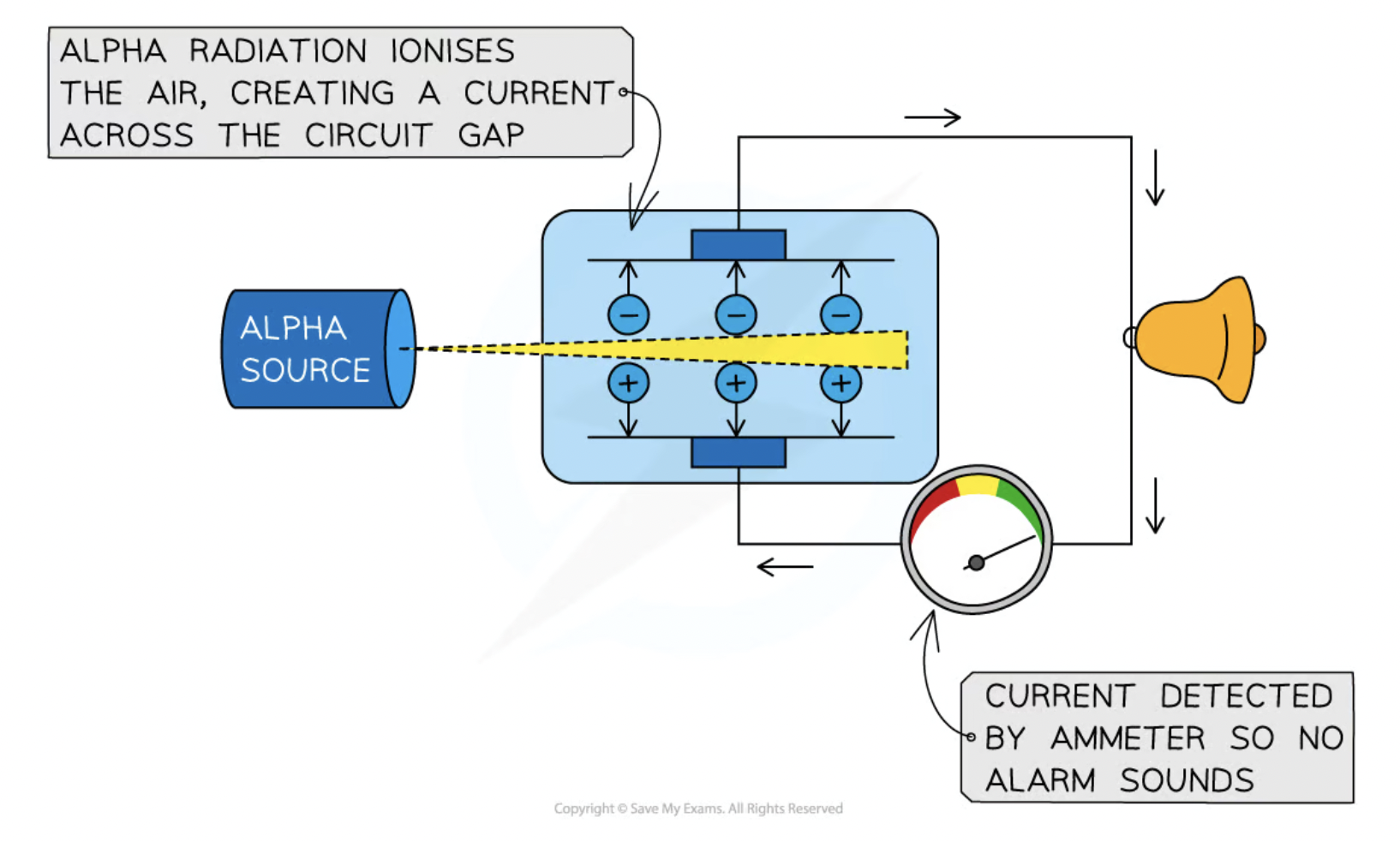
alpha particles are used in smoke detectors
alpha radiation ionises the air within the detector creating a current
Alpha emitter is blocked when smoke enters the detector
Alarm is triggered by microchip when the sensor no longer detects the particles
sterilization of equipment - uses of radiation
gamma radiation
kills bacteria by breaking down bacterial DNA, inhibiting bacterial division
Irradiating food to kill bacteria - uses of radiation
gamma rays can irradiate food to kill any microorganisms that are present on it
this makes food last longer + reduces risk of food-borne infections
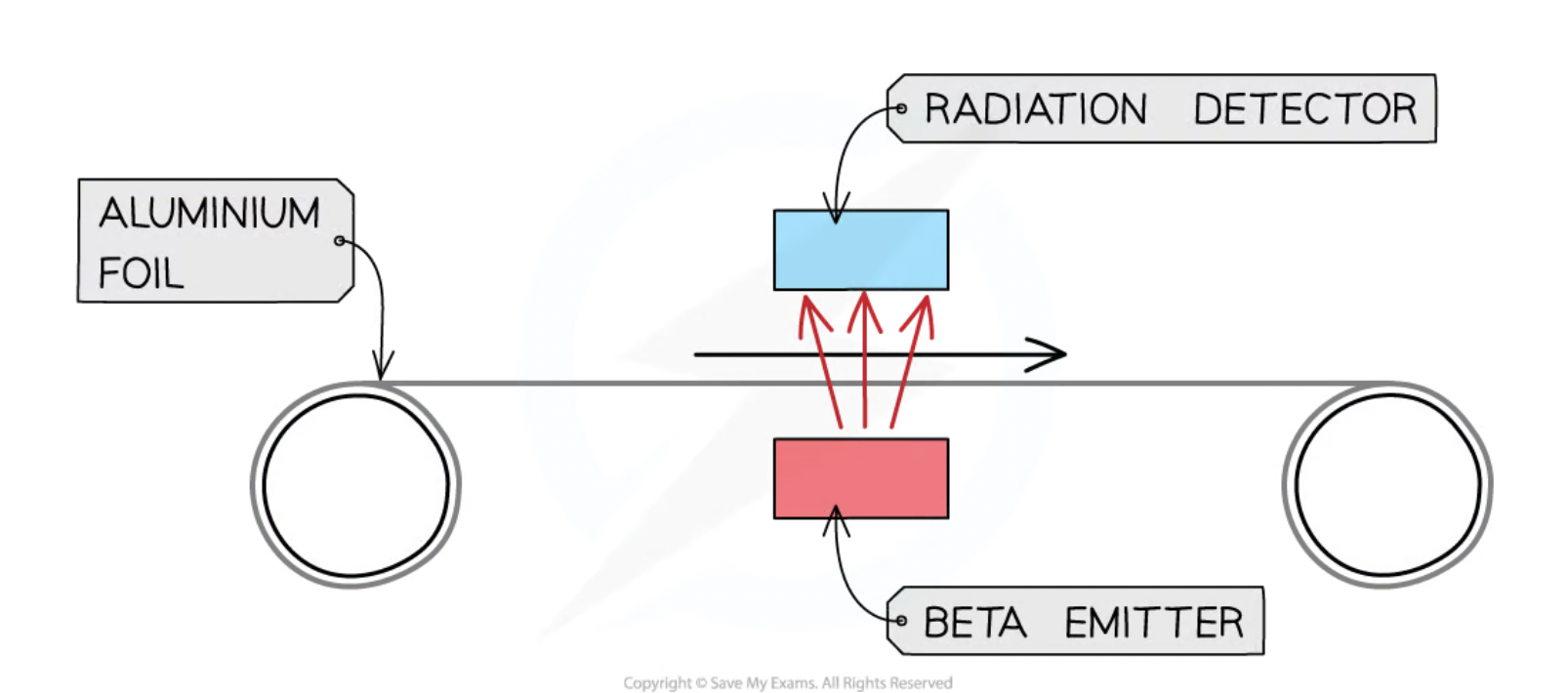
Measuring the thickness of materials - uses of radiation
Beta radiation is most commonly used
1 - material moves across radiation source and the particles that go through it are monitored using a detector above
2 - the thickness of the material is monitored. ( thicker → less particles are detected by detector due to them being absorbed by the material)
the machine makes adjustments to keep the thickness of the material constant
diagnosis of cancer
tracer - a radioactive isotope that can be used to track the movement of substances like blood around the body
PET scan can detect the emissions from a tracer to diagnose cancer and determine the location of a tumor
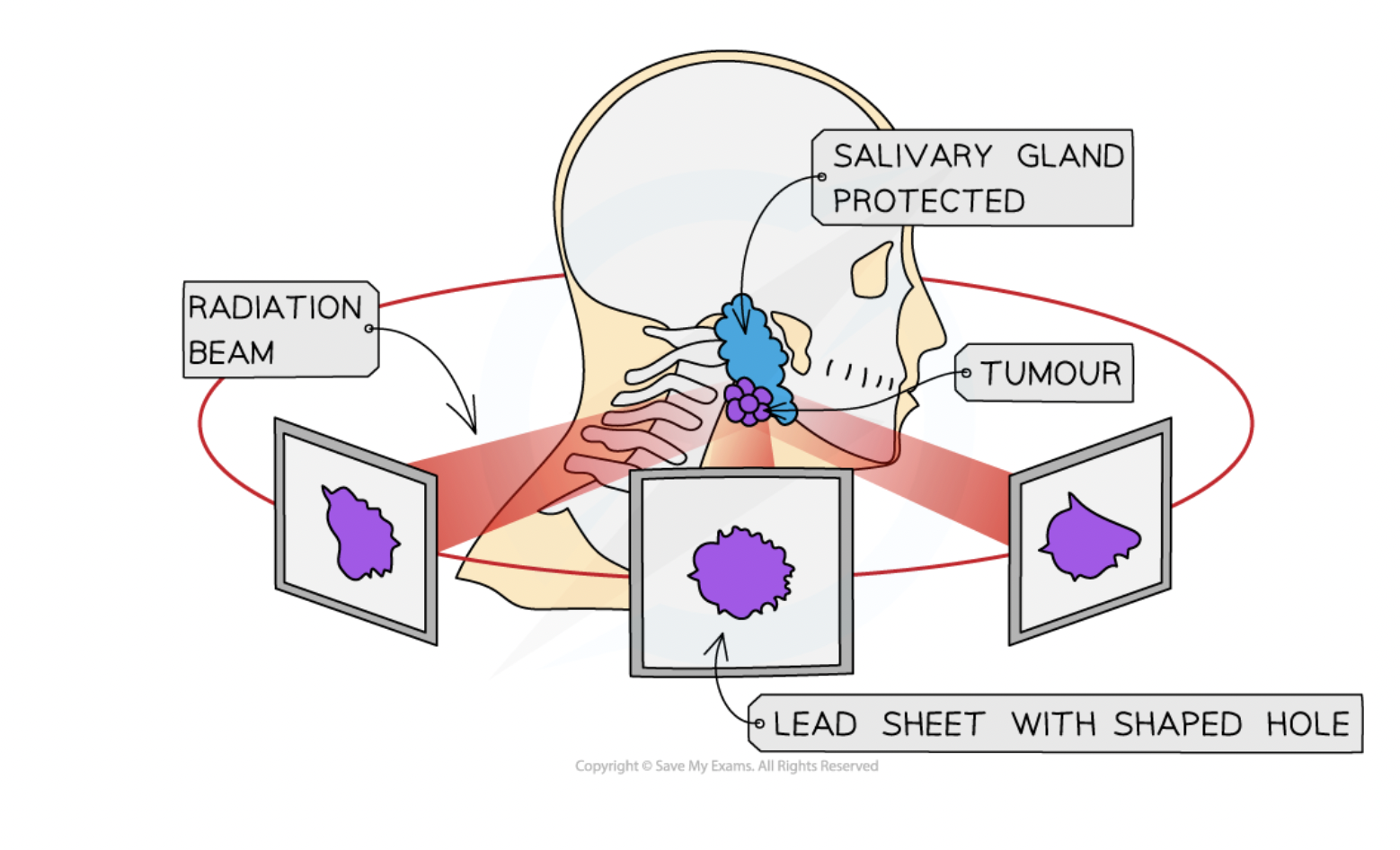
treatment of cancer - uses of radiation
radiotherapy (chemotherapy is with chemicals)
beams of gamma rays are directed on to the cancerous tumor (gamma rays can go through the body)
beams are moved around to minimize harm to healthy tissue whilst being aimed at tumor
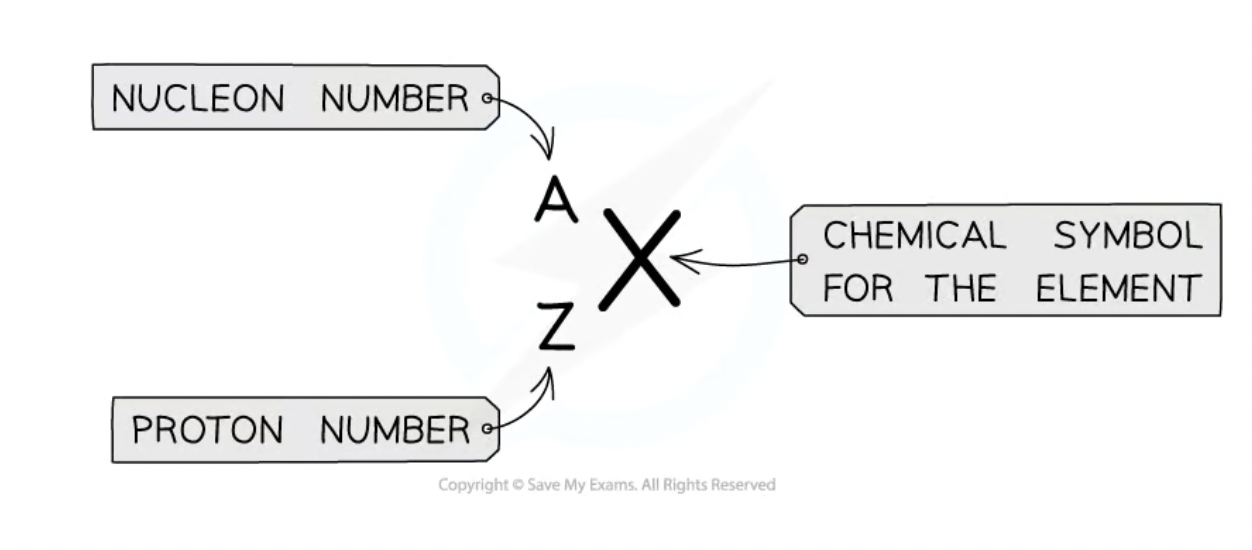
Atomic/proton number (Z)
determines what element it is
= number of protons
= number of electrons
Nucleon number (A)
total number of particles in the nucleus of an atom
= protons + neutrons
Nuclear charge
= relative charge of nucleus
determined by the proton number of the atom
nuclear charge = number of protons in nucleus x relative charge of a proton
ionisation definition
when an atom becomes negatively or positively charged (by gaining or losing electrons)
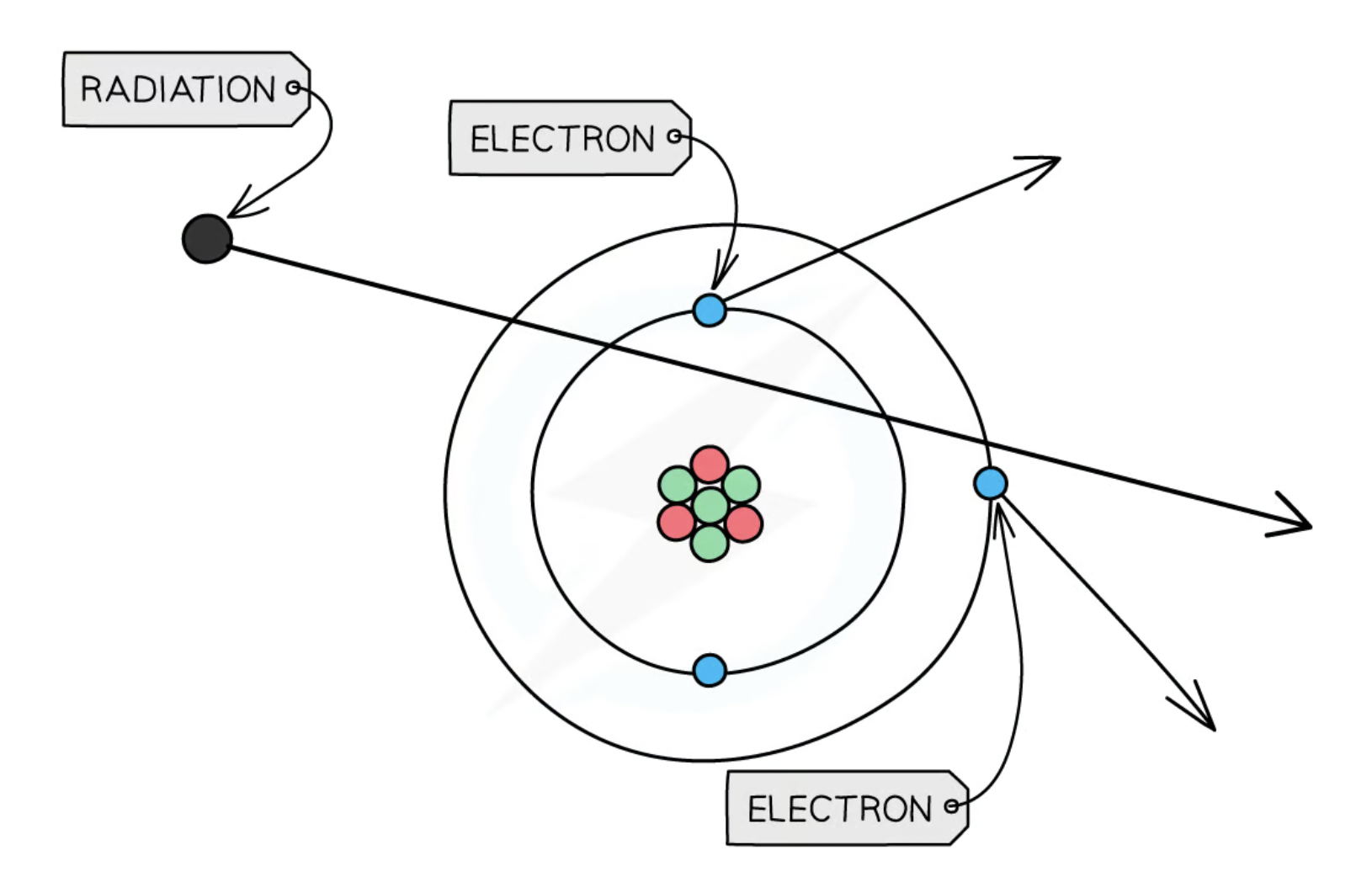
Nuclear radiation ionizing an atom
Nuclear radiation can ionise the atoms that it hits
this is mostly done by removing an electron so the atom looses a negative charge → left with an overall positive charge
Background radiation
the radiation that exists around us all the time
2 types :
Natural sources - from radioactive elements that have always existed on earth + outer space
Man-made sources - from human activity
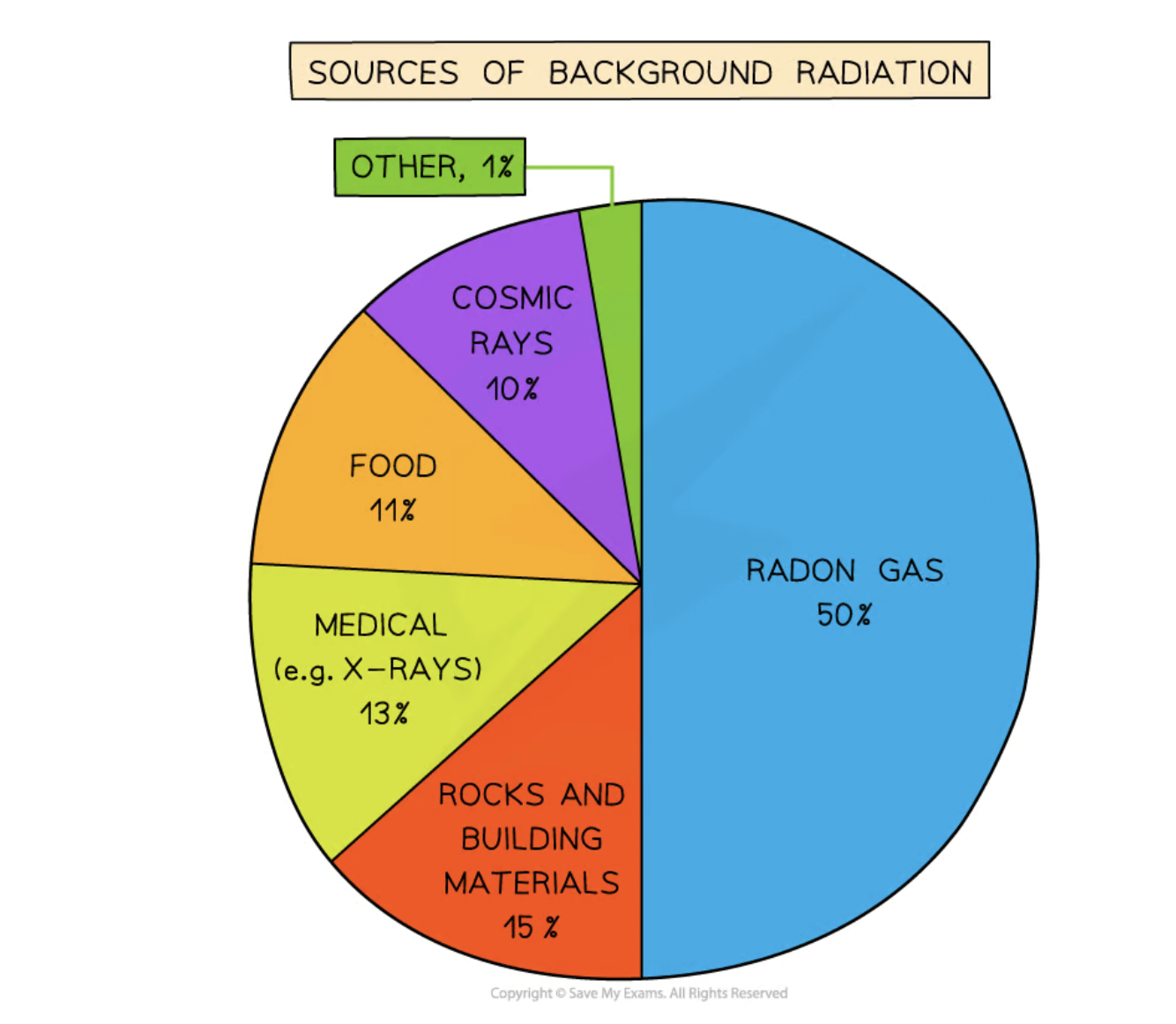
sources of background radiation
radon gas
rocks and buildings
food and drinks
cosmic rays
detecting radiation
Ionising nuclear radiation can be measured using a detector connected to a counter
the detector uses count rate measured in counts/s or counts/minute
count rate = number of decays per second
Geiger–Müller tube
Each time it absorbs radiation, it transmits an electrical pulse to a counting machine
This makes a clicking sound and it displays the count rate on a screen
Higher count rate = tube is absorbing more radiation
tube higher away from source = lower count rate
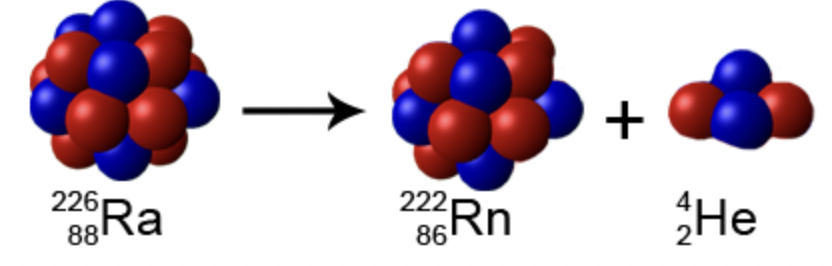
alpha decay (α)
alpha particle = helium nucleus = 2 neutrons + 2 protons
unstable isotope → helium nucleus + stable isotope
Range in air = few cm
High ionising ability
can be stopped by a sheet of paper (low penetrating power)
affected by an electric + magnetic field as it has a 2+ charge, towards negative/deflected away
looses 2 atomic numbers + 4 mass numbers
beta decay (β)
neutron in nucleus spontaneously loses an electron and becomes a proton
can be affected by electrical + magnetic field due to having a charge of -1, towards positive
Beta particle = high energy electron
Range in air → a few 10s of cm
moderate penetrating power → stopped by a few mm of aluminum/perspex
moderate ionising ability
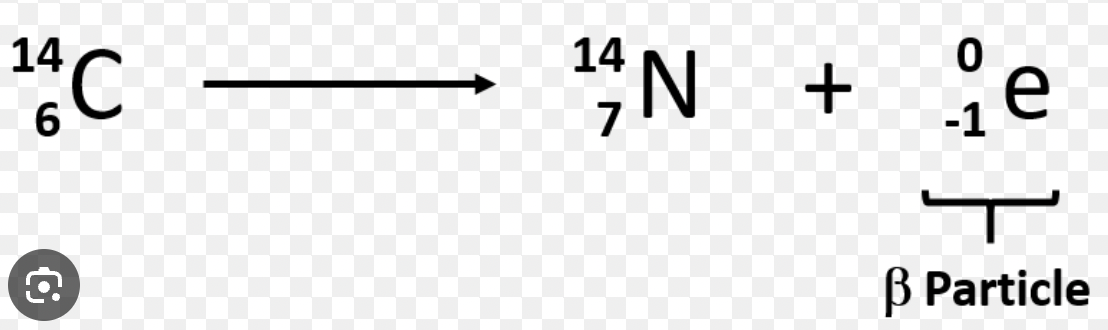
gamma decay (γ)
no charge
electromagnetic wave
infinite range in air
high penetrating power (reduced by few cm of lead)
low ionising ability
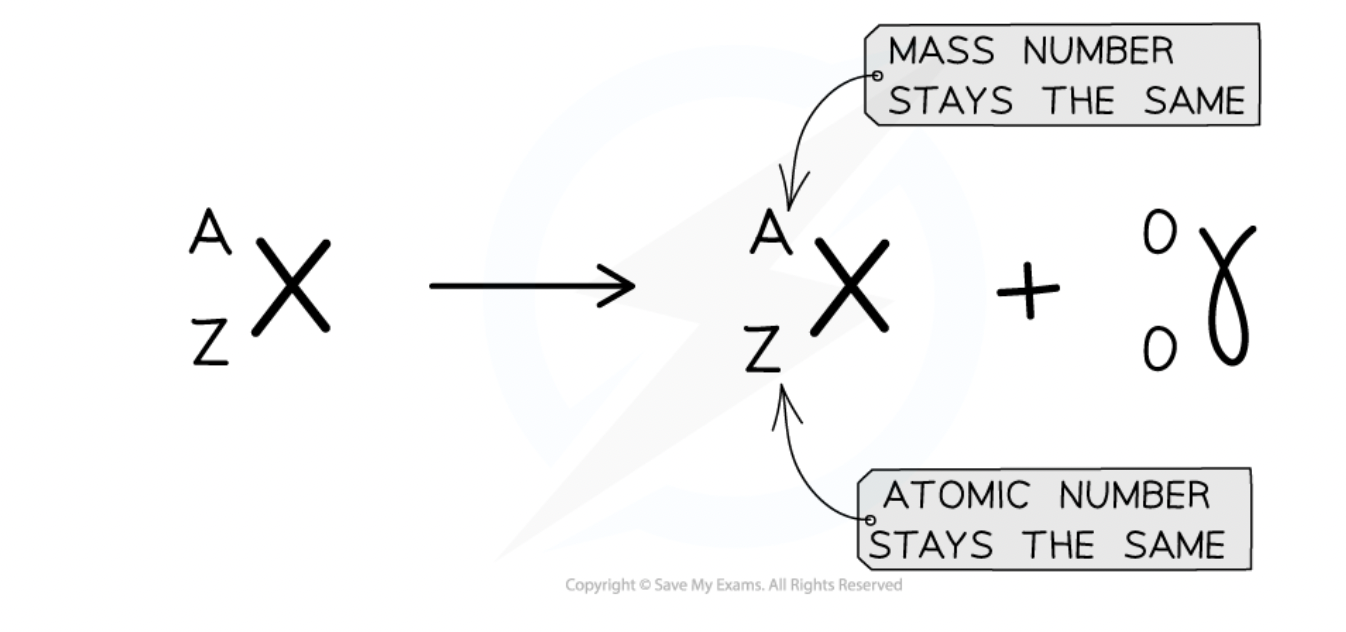
radioactive decay definition
a change in an unstable nucleus that can result in the emission of α-particles or β-particles and/or γ-radiation. (changes are spontaneous + random)
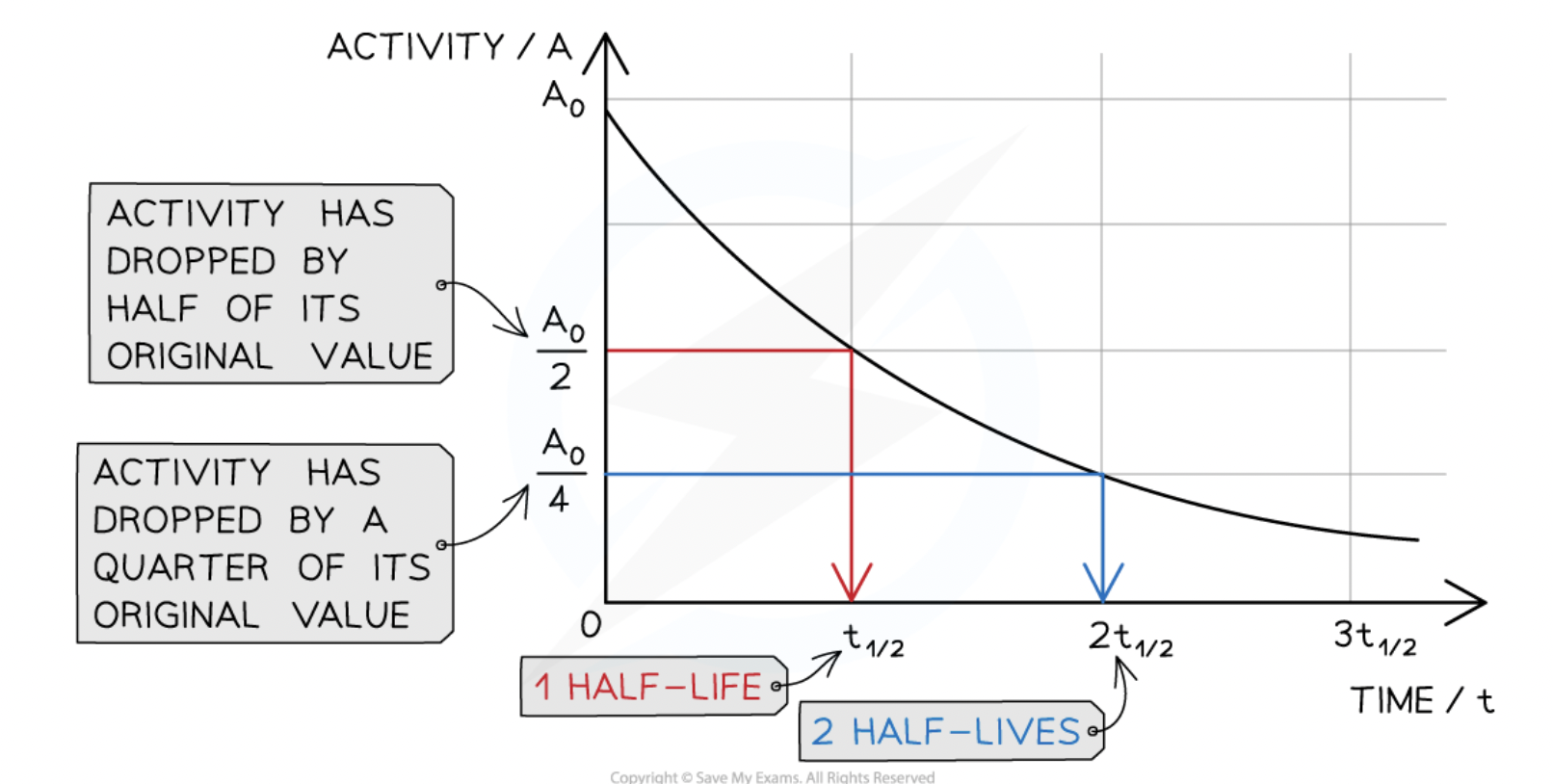
half-life definition
the time taken for half the nuclei of that isotope in any sample to decay
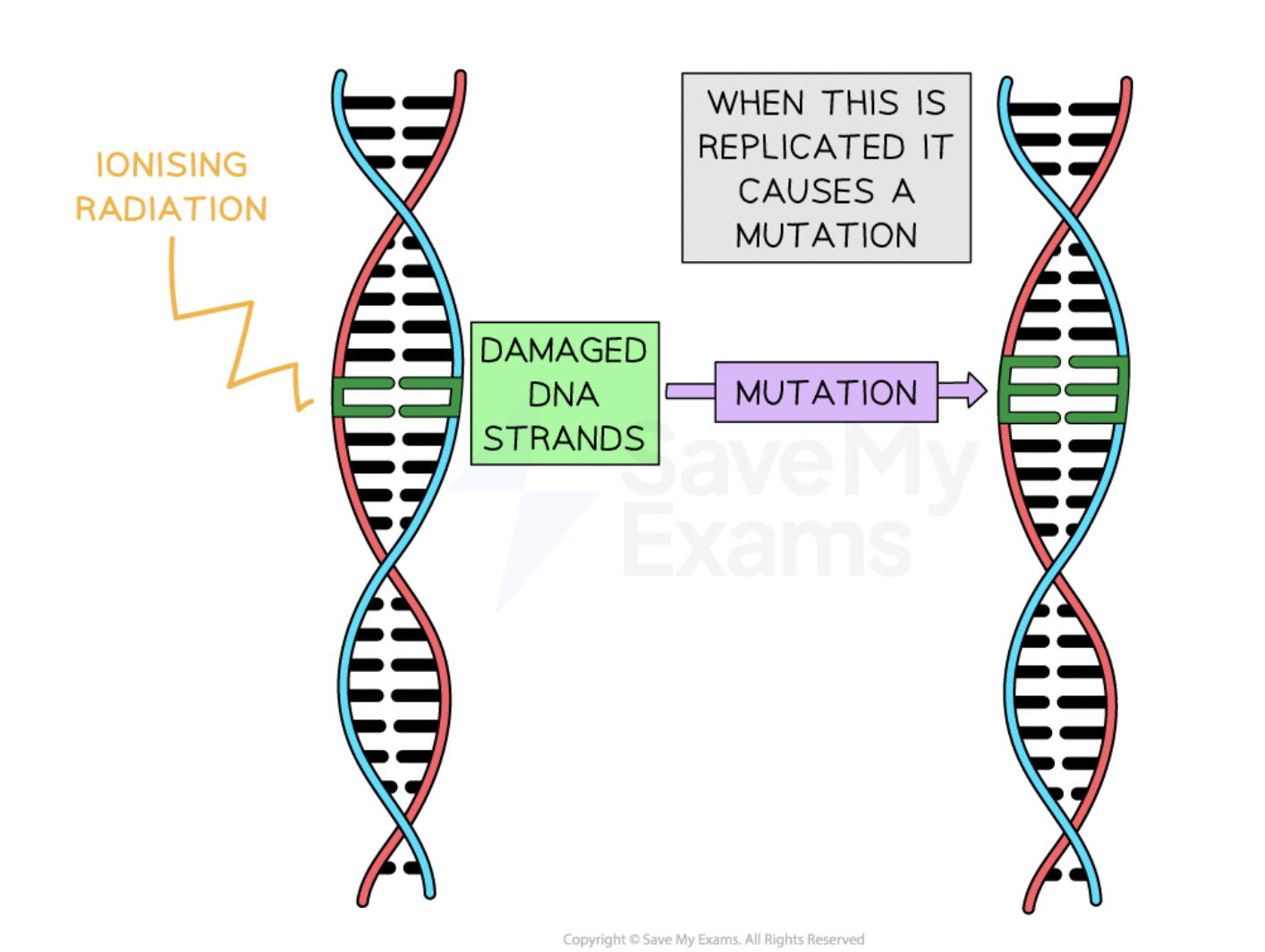
dangers of ionising nuclear radiation
cell deaths
mutations
cancer
safe storage of radioactive materials
reducing exposure time
increasing distance between source and living tissue (e.g using tongs, building power plants in remote places)
using shielding to absorb radiation (e.g made out of lead, water or concrete)
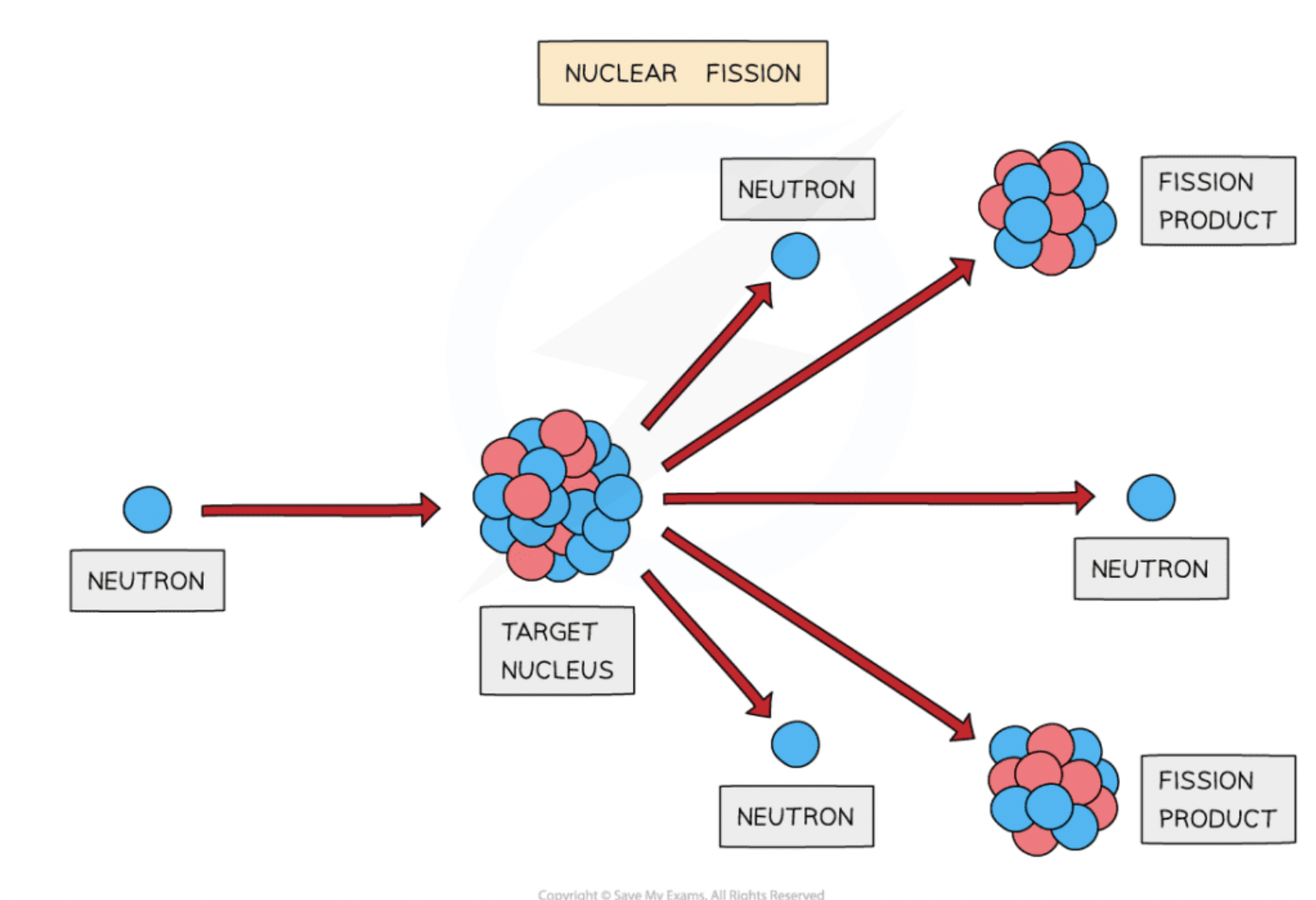
Nuclear fission
splitting a large unstable nucleus into 2 stable nuclei
neutron collides with unstable nucleus (reactants) → 2 smaller nuclei + 2/3 neutrons + gamma
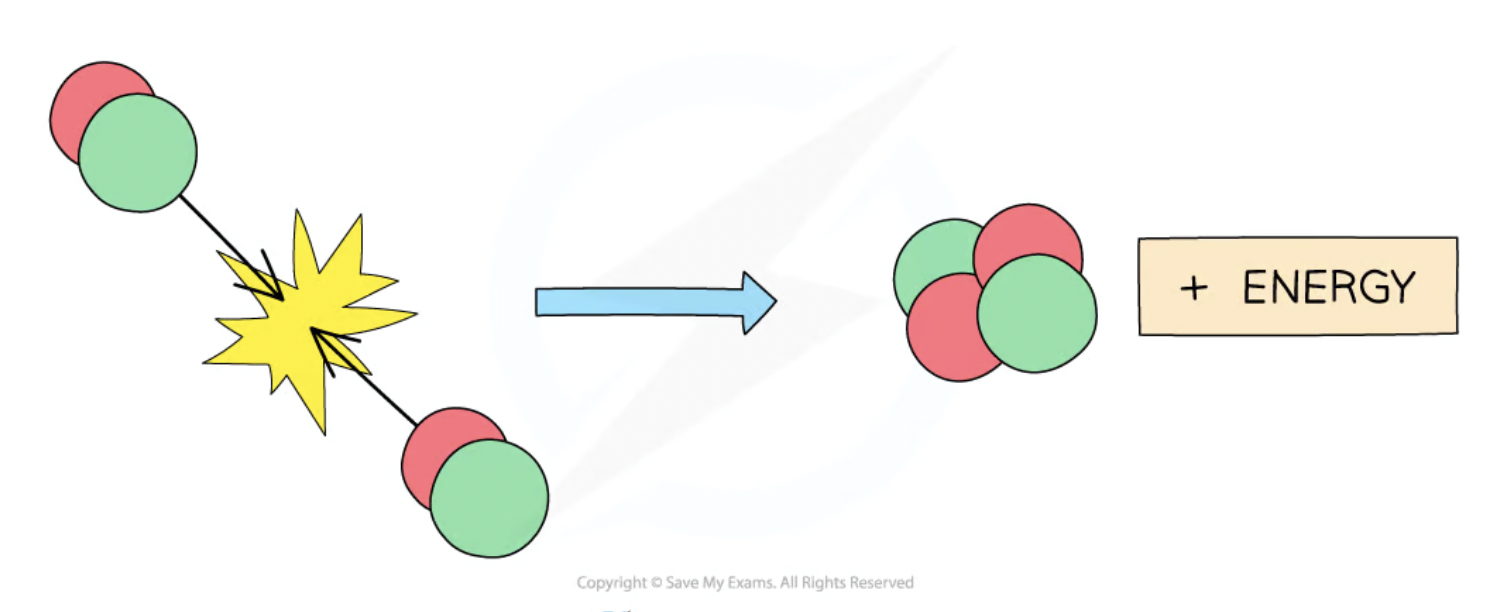
nuclear fusion
when 2 light nuclei join together to form a heavier nucleus
requires high pressure + temperature
what is a light year
the distance travelled in (the vacuum of) space by light in one year
order of planets
mercury, venus, earth, mars, jupiter, saturn, uranus, neptune
minor planets
dwarf planets - object similar to planet but much smaller - pluto
asteroids of asteroid belt
moons
natural satellite
orbit planets
Light
electromagnetic wave
3×10 to the power of 8
how to calculate the time it takes for light to travel a distance
time = distance / speed of light
orbital motion
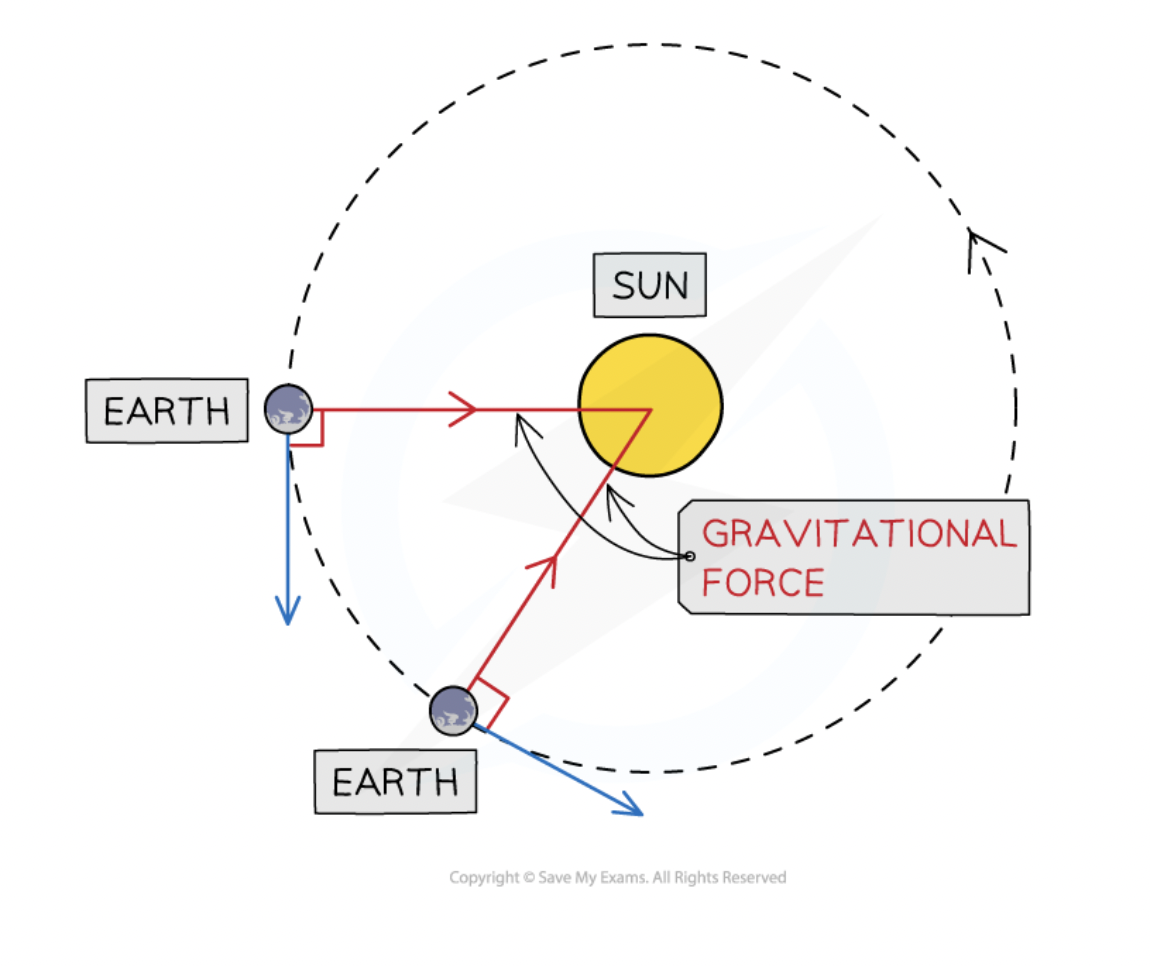
a result of the gravitational force of attraction acting between two bodies
always acts towards the centre of the larger body
causes the orbiting body to move in a circular path
gravitational attraction of the sun
sun contains most of the mass of the solar system
objects orbiting the sun stay in orbit due to the gravitational attraction of the sun
the force is directed from the orbiting object to the center of the sun
The Sun
a medium sized star
consists mostly of hydrogen and helium
radiates most of its energy in the infrared, visible and ultraviolet regions of the electromagnetic spectrum
orbital speed equation
v = 2πr/T
T = orbital period (time it takes to complete one cycle in seconds)
r = radius of the orbit in meters
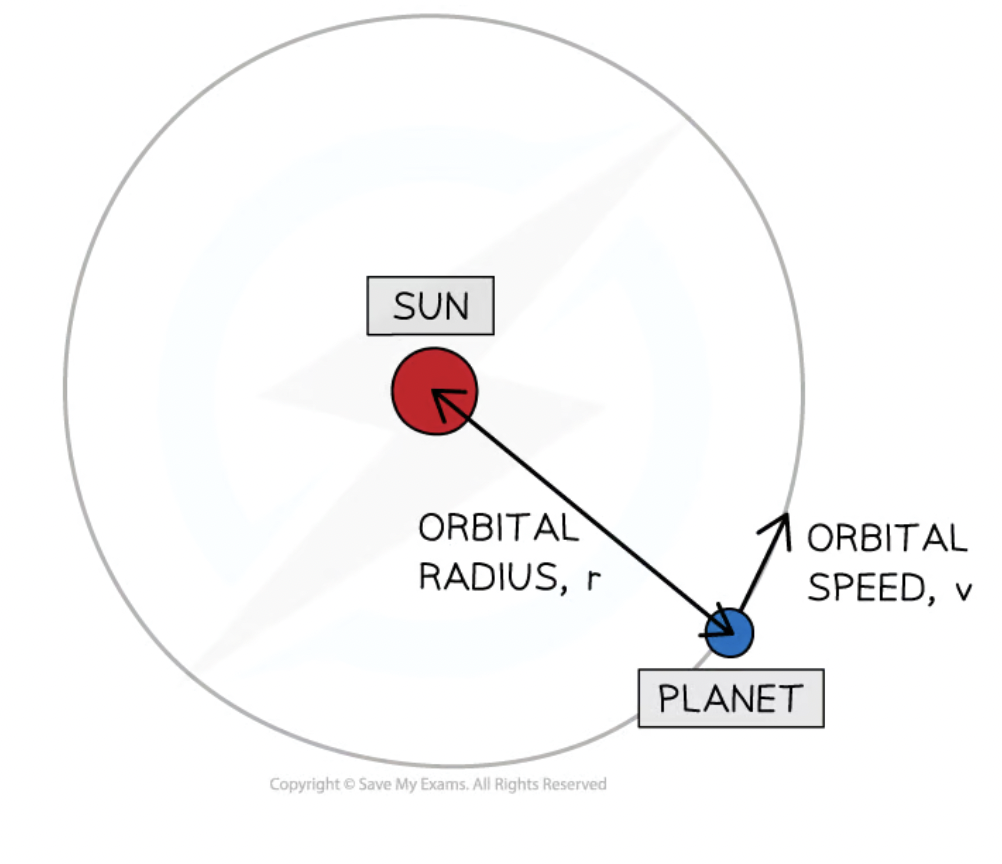
what happens when the distance from the sun increases
Sun’s gravitational field strength decreases
orbital speed of planets decreases
what are stars powered by
nuclear reactions that release energy
in stable stars nuclear reactions involve the fusion of hydrogen into helium
first stages of a stars life cycle that every star experiences
stellar nebula → protostar → stable star
how is a stable star formed
formed as prostars from interstellar clouds of gas and dust(stellar nebula) due to gravitational attraction
life cycle of a small mass star
about the same mass as the sun
red giant → white dwarf + planetary nebula
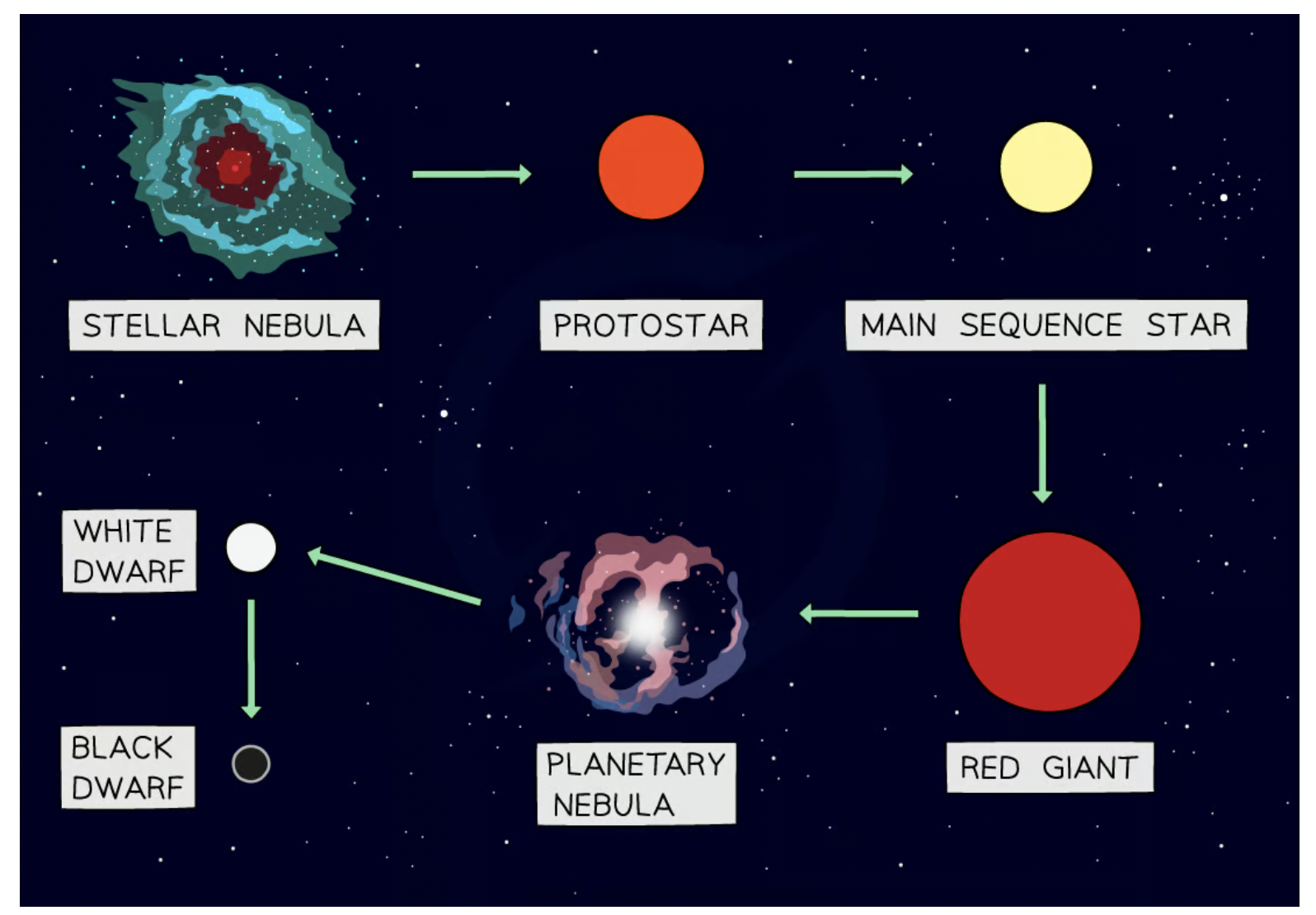
what is a Red giant
Formed when a small mass star reaches the end of its life
The outer layers of the star expand and cool
planetary + supernova nebula
planetary nebula - Formed when the outer layers of the star are pushed away
supernova nebula - may form new stars with orbiting planets
what is a white dwarf
Formed when the core of the star collapses
Found at the centre of a planetary nebula
life cycle of a large mass star
red supergiant → supernova → neutron star
what is a red supergiant
when a star reaches the end of its life
the outer layers of the star expand and cool
what is a supernova
an exploding red supergiant
what is a neutron star
formed when the core of a large super star collapses
very dense (not as much as dark holes)
life cycle of a very large mass star
red supergiant → supernova → black hole
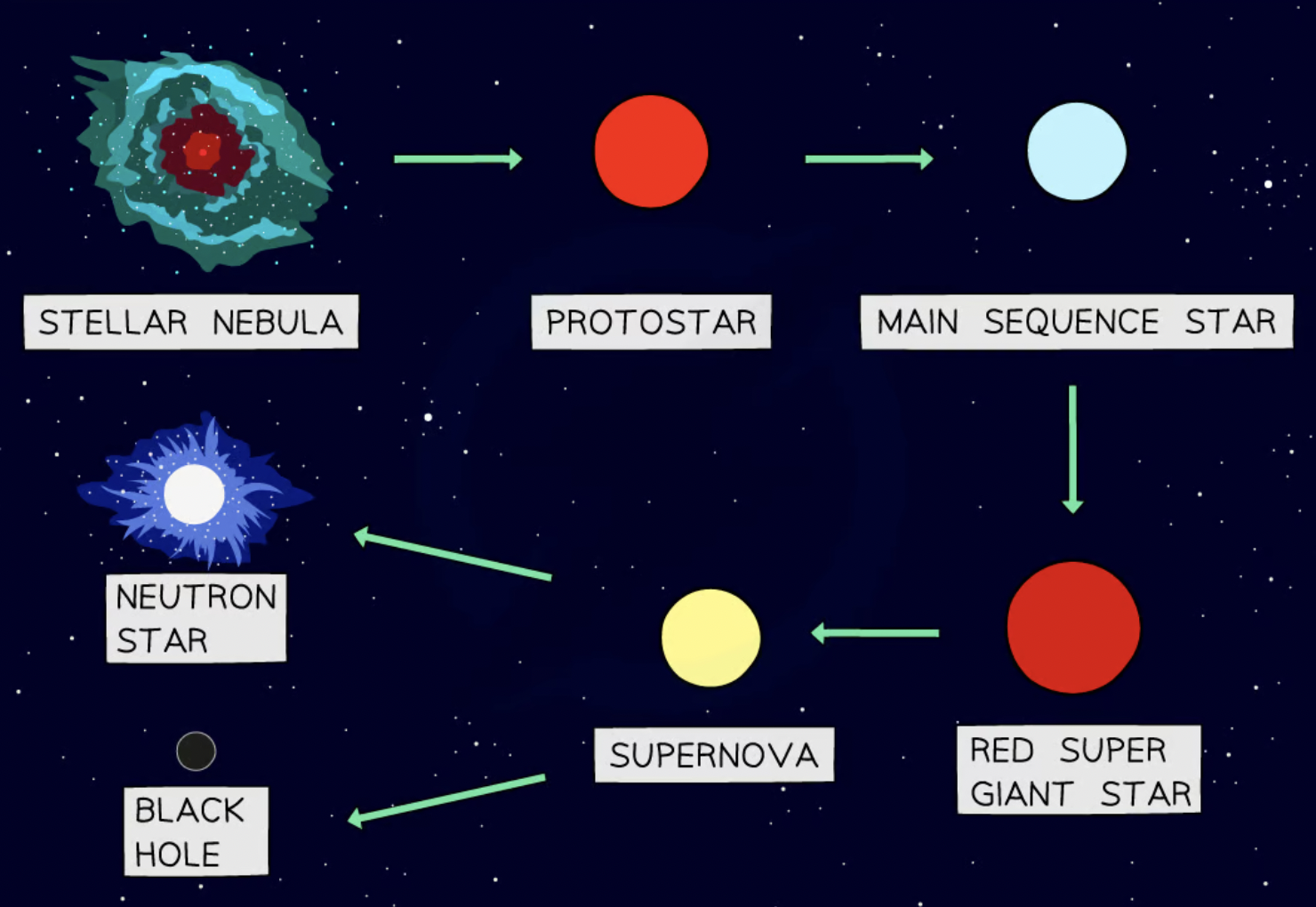
black hole
Formed when the core of a very large star collapses
Extremely dense
how far away are other stars in the milky way
other stars that make up the Milky Way are much further away from the Earth than the Sun is from the Earth
galaxies + milky way
galaxies are made up of billions of stars
Sun is a star in the milky way(a galaxy)
diameter of milky way = approximately 100,000 light-years
Big Bang theory
theory is supported by many astronomical observations
13.8 billion years ago a giant explosion caused the universe to expand from one single point of high density and temperature
The Universe is still expanding ( causes galaxies to be further apart from each other)