S3.2 organic chem
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
same class mean what
same fictional group
homologous series means what
same functional group and general formula that differ by a CH2.
as homologous series goes up what is shown
increase in physical properties such as boiling point and melting point
Structural isomers
same molecular formula but a different arrangement of atoms
Structural isomers can be further divided into what
chain and functional group isomers.
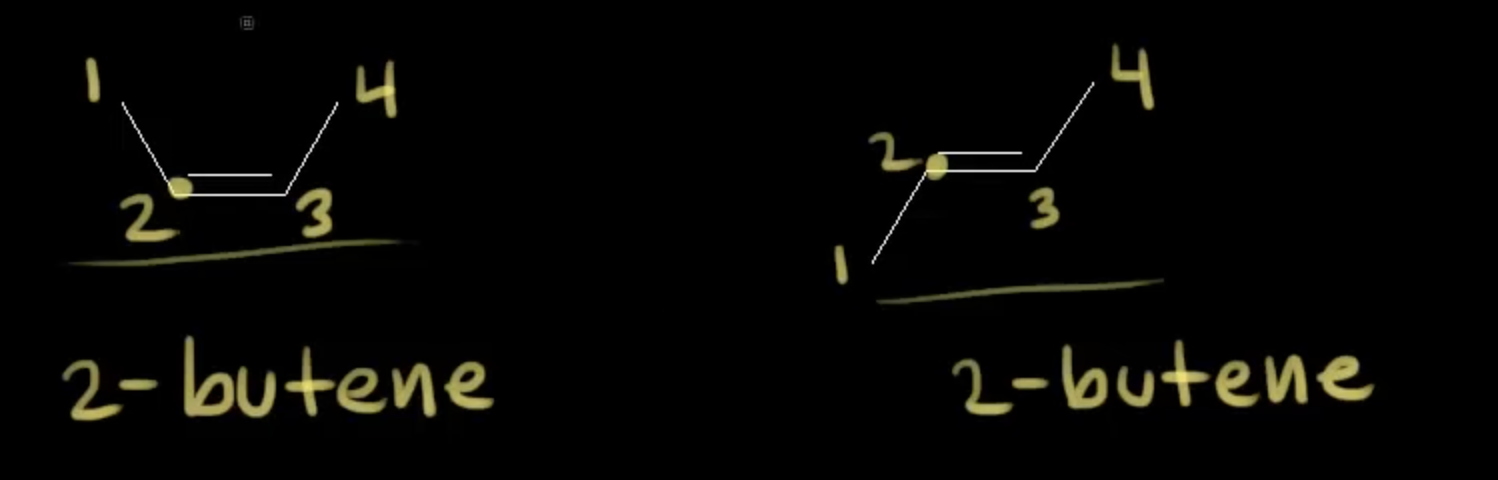
these are what isomers
Cis-trans
Cis-trans isomers form due to restricted rotation of what
double bond
Cis-trans isomers can only be interconverted by
breaking covalent bonds
Optical isomers have
chiral carbon atom bonded to four different atoms or groups.
enantiomer
non-superimposable mirror images
two enantiomers can be distinguished from each other by
interaction with plane-polarised light.
what kind of bonds does mass spectrometry break
covalent
what causes the GHG absorption of IR
molecular vibrations that cause a change in the molecule's dipole moment,
postion isomers
same formula functional group is in different place
fictional group isomers
same formula , different functional group
chain isomer
same molecular formula but parent chain has different length
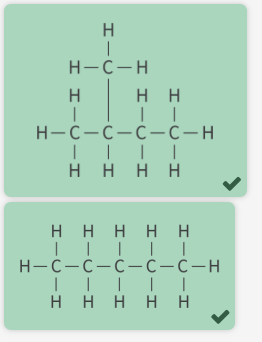
what are these
chain isomers
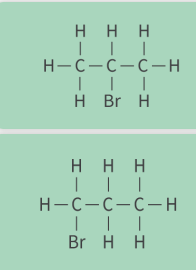
what are these
positional isomers
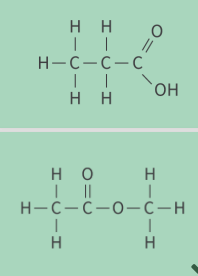
what are these
fictional group isomers
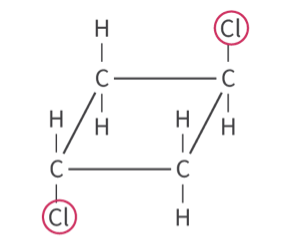
whats this
trans isomer
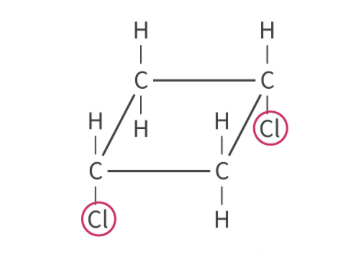
whats this
cis isomer
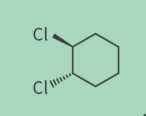
whats this
trans isomer
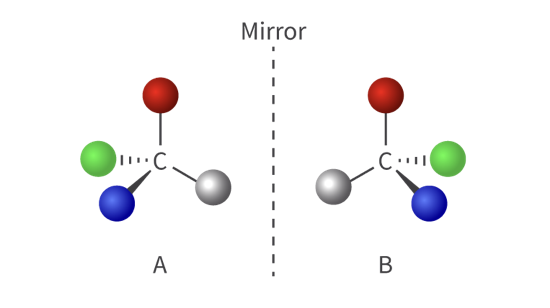
what this
optical isomer
if somthing can form an enatomer its what
optically active
number of signals on HNMR spectrum
number of chemical environments
chemical shift of HNMR signal.
type of protons in each different chemical environmen
HNMR splitting pattern
N=1 where N is the number of hydrogens on the adjacent carbon