Functional Groups
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
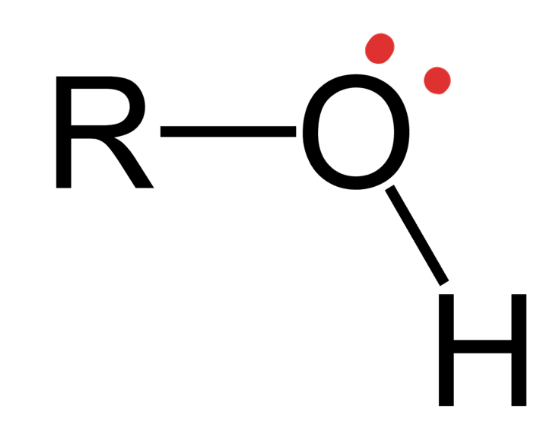
Hydroxyl
Structural Formula: an oxygen atom bonded to a hydrogen atom (−OH)
Properties: Hydrophilic and polar —> increasing solubility in water Examples: alcohols (ex. ethanol), carbohydrates (ex. sugars)
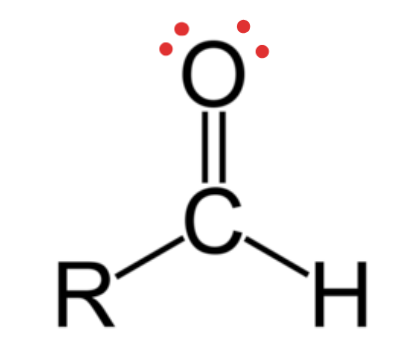
Carbonyl (Aledhyde)
Structural Formula: a carbon atom double-bonded to an oxygen atom (C=O) and bonded to a hydrogen (found at the end of a carbon chain)
Properties: polar —> boiling points and solubility.
Examples: Formaldehyde (methanal), vanillin, sugars
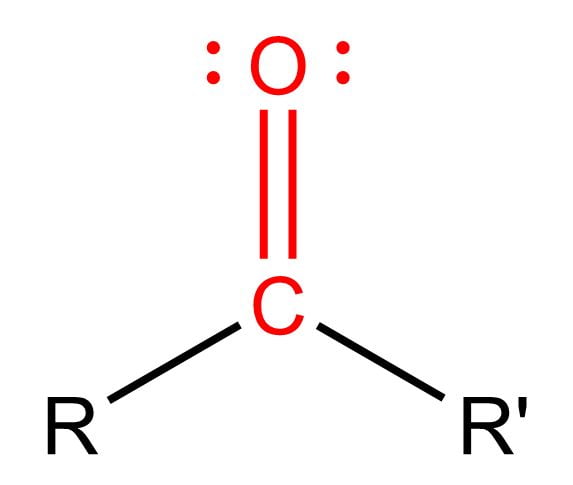
Carbonyl (Ketone)
Structural Formula: a carbon atom double-bonded to an oxygen atom (C=O), with 2 available bonds to connect in a carbon chain (found in the middle of a carbon chain)
Properties: polar
Examples: acetone, methylethyl ketone, cyclohexanone
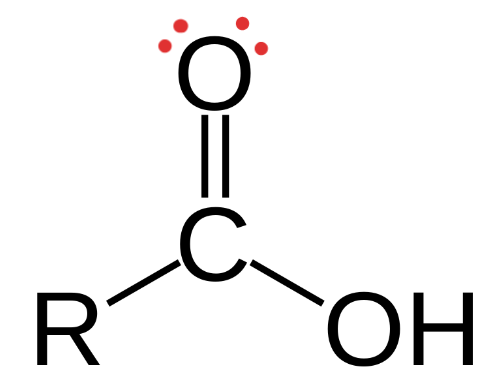
Carboxyl
Structural Formula: a carbon atom double-bonded to an oxygen atom and also bonded to a hydroxyl group (−COOH).
Properties: polar, (acts as an acid by donating protons, contributing to acidity in solutions.
Examples: carboxylic acids (ex. acetic acid (vinegar), citric acid (vitamins), fatty acids)
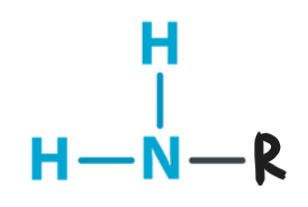
Amino
Structural Formula: a nitrogen atom bonded to two hydrogen atoms (−NH2).
Properties: polar, basic (can accept protons), forming ammonium ions.
Examples: amino acids (ex. glycine), proteins, ammonia
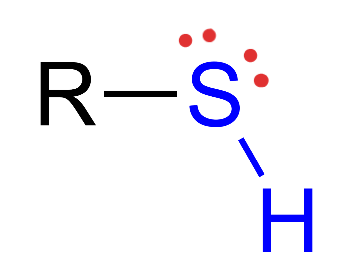
Sulfhydryl
Structural Formula: sulfur atom bonded to a hydrogen atom (−SH). Properties: polar, contributes to the structure of proteins through disulfide bonds, influencing protein folding.
Examples: proteins, rubber
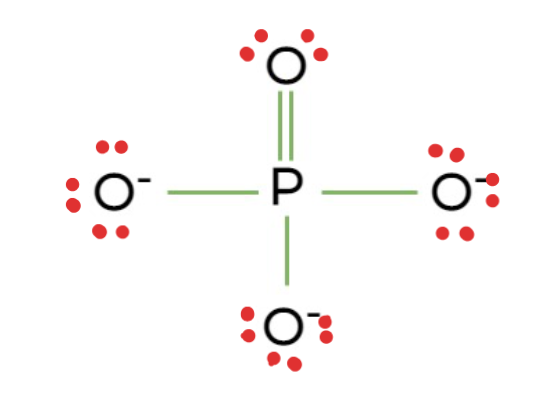
Phosphate
Structural Formula: a phosphorus atom bonded to four oxygen atoms (−PO4).
Properties: Highly polar and negatively charged, affecting molecular interactions.
Examples: nucleic acids (DNA and RNA), ATP (adenosine triphosphate), phospholipids
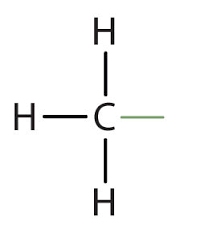
Methyl
Structural Formula: a carbon atom bonded to three hydrogen atoms (−CH3).
Properties: non polar (hydrophobic), often preventing hydrogen bonding.
Examples: methane, many fatty acids.