Chemistry - Semester Test
1/49
Earn XP
Description and Tags
All of the questions and answers for the semester test in Chemistry.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Which metallic property is explained by the interaction of delocalized electrons with light?
luster
If the substance went straight from A to G, what would be call that phase change?
Sublimation
A scoop of ice cream has an energy value of 775,000 joules, what is the value in Calories?
185 Cal
The electron configuration of K+ is most similar to which noble gas?
Argon (Ar)
Heisenberg, it’s impossible to know both the ________ and __________ of an electron at the same time.
Position and momentum
Where is it freezing? Boiling?
B and E
Which of the orbits are spherical?
2s
A 21.5g piece of iron at 100.0 C was dropped into a calorimeter with 132g of water at 20.0 C. What is the final temperature of the system?
21.4 C
According to whom, each element has a unique spectrum?
Bohr
List one assumption that must be made when using a calorimeter.
All of the above
Where is it melting?
C
Where is the d region of the periodic table?
Groups 3-12
Give an example of a pair of atoms that will form a covalent bond.
N, O
Le Chatelier’s principle, an increase in the concentration of a reactant will cause a reaction to…
consume the added reactant
Where is it a liquid?
D
Charle’s law, at constant pressure and number of molecules, volume is directly proportional to…
temperature
Give a general physical property of covalent compounds.
Low melting point.
The diagram ^v shows two electrons with…
opposite spins
List one thing that determines the temperature increase in a substance.
specific heat of the substance and mass of that substance
What type of bond forms from the transfer of electrons?
ionic
Where is it solid?
A
If the decomposition 1.00 mole of sodium hydroxide (molar mass of 40.0g/mol) absorbs 60.0kJ of energy, how much energy would be absorbed
30
Light is said to have the properties of both particles and…
waves
Boyle, at a constant temperature and number of molecules, the pressure of a gas is inversely proportional to it’s…
volume
What is a substance that conducts heat well, is hard, and is malleable?
metal
When the particles of a solid gain enough kinetic energy to break their ordered arrangement, and slip past one another, the solid…
melts
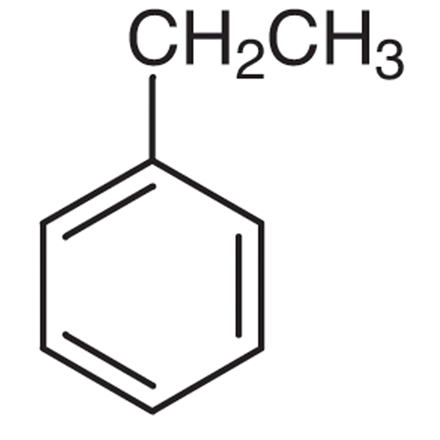
What is the name of the pictured compound?
ethylbenzene
Give 2 examples of units that can measure heat.
all of the following except watts
What is H2SO4?
sulfuric acid
What are the products that would be expected in the neutralization reaction involving nitric acid and barium hydroxide?
Ba(NO3)2 and H2O
Why do road crews put brine on the road in the winter?
all of the above
Give an example of alternative energy.
all of the following except natural gas
What is the substance who's aqueous solution conducts an electric current?
electrolyte
The fifth member of the alkane series has the formula…
C5H12
What is concentration measured in?
molarity
List one thing that happens in a neutralization reaction.
all of the above
Give an example of a polar molecule.
all of the above
An organic compound must contain what?
carbon
Draw the correct Lewis dot diagram for hydrogen chloride.

What is energy transferred as a result of temperature difference?
heat
What is the pH of an acidic solution?
less than 7
What is the conjugate acid in HCl(aq) + KOH(aq) = KCl(aq) + H2O(l)?
H2O
Give an example of a buffered solution.
blood
What is a laboratory technique that uses a solution of known concentration to determine the concentration of another solution?
titration
Name one thing that is happening in a reversible reaction.
all of the above
CH3CH2CH2CH=CH2 is an example of what kind of hydrocarbon?
alkene
What property of a cola soft drink would limit your ability to test it’s pH using an indicator?
it is dark brown in color
What is the heat needed to raise the temperature of 1g of a substance by 1 C
specific heat
Give an example of an amphoteric substance.
water
In a triple bond, how many electron pairs are shared?
3