Calculating moles of gases
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
10 Terms
How do you calculate the number of moles of a gas present?
Moles of gas= volume of gas (in dm³) / 24
Moles of gas= Volume of gas (in cm³) / 24,000
What 2 conditions affect the molar volume of a gas?
Temperature
Pressure
State the equation used to calculate the volume of a gas under changed conditions.
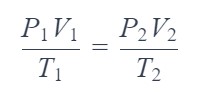
State the ideal gas equation.
pV= nRT
p is the pressure (in Pascals).
V is the volume (in m³).
n is the number of moles.
R is the gas constant.
T is the temperature (in K).
How do you convert from kilopascals to pascals?
x 1,000.
How many pascals are in 1 atmosphere?
1 atmosphere= 101,325 Pa.
How many dm³ are in 1 m³?
1 m³= 1,000 dm³.
How many cm³ are in 1 m³?
1 m³= 1,000,000 cm³.
What is the value for the gas constant?
8.314 J K−1 mol−1.
It is provided in the data sheet.
How do you convert from celcius to kelvin?
Add 273.