Basic concepts- core organic chemistry
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
What is isomerism?
Compounds which have same molecular formula but be different
what goes first
carbonyl group is the highest priority
Remember: There is no need to number an aldehyde as it is automatically carbon-1
—
What are the different type of isomers
Structural isomerism
Stereoisomerism
What is structural isomerism?
Compounds with same molecular formula but w. diff structural formula
What are the different types of structural isomerism?
Chain isomerism
Positional isomerism
Functional group isomers
Chain e.g.
Pentane and 2-methlybutane
diff branching
What is positional isomerism?
Contain same functional group but in different positions on carbon chain
What is functional group isomerism?
Compounds which have same molecular formula but different functional groups
e.g, aldehydes and ketones
molecular formulae
shows actual numbers of each atom in the molecule
empirical formulae
shows numbers of each atom in simplest whole number ration
general formulae
simplest formulae for a member of a homologous series
displayed formulae
shows every atom and every bond
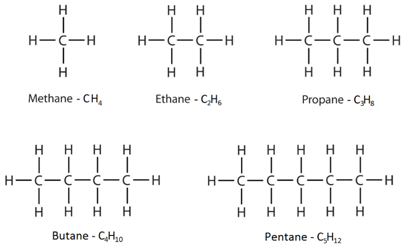
structural formulae
Shows how atoms are joined together
CH3-CH2-CH2-CH3
hydrocarbon definition
compound of only hydrogen and carbon
saturated definition
containing single bonds only
unsaturated definition
containing multiple carbon-carbon bonds
homologous series definition
a series of organic compounds with same functional group therefore similar chemical properties but with each successive member differing by CH2
functional groups
part of organic molecule largely responsible for its chemical reactions
aliphatic
carbon atoms are joined to each other in unbranched (straight) or branched chains or non-aromatic rings (alkenes, alkanes, alkynes)(not ring
alkynes
containing at least one triple carbon-to-carbon bond
alicyclic
containing carbon atoms joined together in a ring that is not aromatic, with or without branches
aromatic
some or all the carbon atoms are found in a benzene ring
stem =
n. of carbons in longest continuous chain
what n. of carbons = stem
1- meth
2-eth
3-prop
4-but
5-pent
6-hex
7-hept
8-oct
9-non
10-dec
prefix=
added before stem to indicate side chain or functional group
naming aliphatic alkanes (straight carbon + branched)
Identify longest continuous chain - corresponding alkane
Identify side chains (alkyl group)- count n. of carbon atoms from end
Add n. before alkyl groups to show position on parent alkane + dash + name of branch
If multiple chains put names in alphabetical order. if on same branch use, di, tri
e.g.
3-methlyhexane
4-ethyl-2,2-dimethylheptance
If two or more possible chains of same length how do you choose a chain
chain with most branched is considered the longest
Separate words and numbers with ____
Separate numbers with a ____
dash
comma
How to find name of alicyclic alkanes?
1- Identify longest continuous chain of carbon atoms
2- Add prefix cyclo-
e.g. cyclopentane
naming alkenes
Use smallest number between double bonds
eg. but-1-ene
but-2-ene
4-methyl,pent-2-ene
alcohol
functional group: -OH
prefix: hydroxy-
haloalkane
Functional group: -Cl, -Br, -I
Prefix: chloro-, bromo-, iodo-
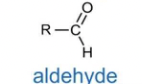
aldehyde
Functional group: -CHO
suffix: -al
e.g. propanol
ketone
Functional group: -C(C0)C-
suffix: one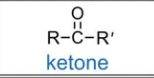
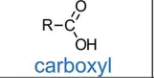
Carboxylic acid
Functional group: -COOH
Suffix: -oic acid
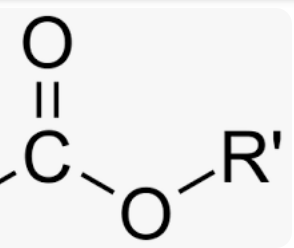
ester
functional group: -COOC-
Suffix: -oate
amine
functional group: -NH2
prefix: amino-
suffix: -amine
which goes first
alphabetical order
ol should get smallest number
When naming branched chain alcohols, be careful to number the longest possible carbon chain first.
what should have smallest number
ketone
e.g. of structural formula
CH3CH2CH3
skeletal for benzene + molecular
C6H6
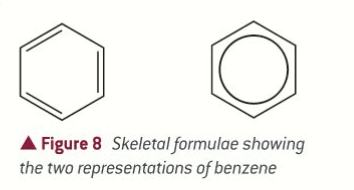
How to draw double bond in skeletal form
2 parallel lines
formation of a sigma bonds
Covalent bond is sharing pair of electrons
Orbitals overlap, they form covalent bond
end-on overlap of orbitals results in sigma bond
each carbon in an alkane forms four sigma bonds
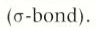
What is shape of alkanes and why
tetrahedral arrangement around each carbon atom and bond angle of 109.5°
beacuse eahc carbon is surrounded by four electron pairs in four sigma-bonds
What does sigma bond in alkanes act as?
axes around whcih atom can rotate freely, so shapes are not rigid
As chain length of alkanes increases of alkanes what happens to b.p?
increases
Why does chain length lead to increased b.p
n. of electrons increases which increases chain length
leads to more induced dipole-dipole interactions
greater london forces, more energy required to separate molecules
In straight chain molecules what happens to surface area and why?
greater surface area in contact possible between molecules
as chain increases in length
therefore more energy needed to overcome it
What does greater surface area in contact mean for b.p?
increases and greater in
branching if carbon chain _____ b.p
deacreases
why does increased branching increase b.p.
less SA of contact between molecules therefore weaker london forces
branches can get in way and prevent branched molecules getting as close together as straight chained molecules, decreasing IMF further
Reasons for lack of reactivity of alkanes:
C-C and C-H sigma-bonds are strong
C-C bonds are non-polar
Electronegativity of carbon and hydrogen is so similar the C-H bond can be considered non-polar
Complete combustion- exces oxygen:
alkane + oxygen —> carbon dioxide + water
equation for balancing complete combustion of alkanes
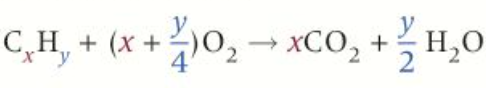
combustion of alkanes is highly ____ useful beacuse
exothermic
release a relativly large amount of energy per gram of fuel
Incomplete combustion- oxygen is limited:
alkane + oxygen —→
carbon + water
or
carbon monoxide + water
alkanes with chain lengths of ___ carbon atoms are used as fuels in internal combustion engines
5-10
2 types of bond fission (breaking cov bonds in reactions)
-Homolytic
-Heterolytic
What is homolytic fission?
each of atoms in covalent bond takes one of the shared pair of electrons
H3C-CH3 —> H3C· + ·CH3
What does homolytic fission form?
2 radicals
What are radicals
an atom/group of atoms with an unpaired electron. they are v reactive
What is heterolytic fission?
One of atoms in cov bond takes both electrons from shared pair
H3C-Cl —> H3C+ + Cl-
What does heterolytic fission form?
2 ions are formed
atoms that took the pair of electrons forms a neg ion
atom withut pair of electrons forms a pos ion
In heterolytic fission is arrow double headed or single header
double- shows pair of electrons moving
In homolytic fission is a single headed arrow or double headed arrow used
single- show movement of one electron
What is a substitution reaction?
Atom or group of atoms is replaced by another atom or group
How do alkanes react with halogens?
UV radiation
CH4 +Br2 —> CH2Br + HBr
What happens in substitution reactions?
replacement of hydrogen atoms with halogen to form hydrogen chloride and mixture of haloalkanes
What are the three steps of radical substitution?
1- Inititation- forming a radical
2- Propagation - new radical is made
3- Termination- two radicals comb
1: Initiation
Reaction is started when cov bond in bromine molecule is broken by homolytic fission.
UV splits chlorine molecule into two chlorine radicals
Cl-Cl —> Cl· + Cl·
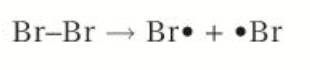
2: Propagation
Radicals are v. reactive. When Cl radicals collide with methane molecule with sufficient energy, a hydrogen atom is removed to form methyl radical and a molecule of hydrogen chloride.
Cl· + CH4 —> HCl + ·CH3 (methyl radical)
.Reaction propagates through 2 steps, a chain reaction:
Bromine radical, Br·, reacts with C-H bond in the methane, forms a methyl radical, ·CH3, and a molecule of hydrogen bromine, HBr
Each methyl radical reacts with another bromine molecule, forming organic product, bromomethane, CH3Br, together with bromine radical.
New bromine radical then reacts with another CH4 molecule as in first propagation step, and the two steps can continue to cycle through in a chain reaction
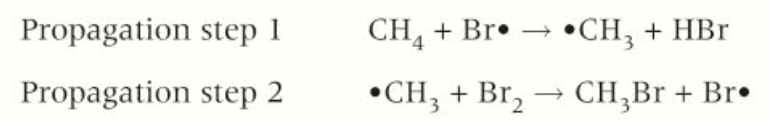
Another propagation reaction
Methyl radicals are also v reactive and will remove a chlorine atom from a chlorine molecule forming another radical
·CH3 + Cl-Cl —> H3C-Cl (chloro methane) + Cl·
3: Termination
2 radicals collide, forming molecule with all electrons paired. There are a number of possible termination steps with different radicals in a reaction mixture
When 2 radicals collide and react, both radicals are removed from reaction mixture, stopping reaction
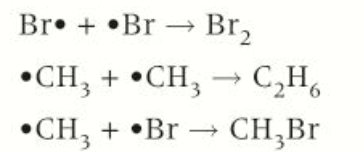
When do propagation steps stop
When two radicals collide and the overall reaction stops when there are no radicals left
Cl· +Cl· —> Cl2
·CH3 + ·Cl —> CH3Cl
·CH3 + ·CH3 —> C2H6
Products of reaction
Mixture of products is made during radical substitution reaction due to reactive radicals in propagation step
As conc of CH3Cl builds an alternative propagation step becomes increasingly likely as a chlorine radical can collide with CH3Cl molecule:
CH3Cl + ·Cl —> ·CH2Cl + HCl
The ·CH2Cl collides with chlorine molecule:
·CH2Cl + Cl2 —> CH2Cl2 + ·Cl
Leads to di-, tri-, tetra- substituted methane
CH4 + Cl2 —> CH3Cl, CH2Cl2, CHCl3, C2H6 + HCl
Produces mixture of mono and poly-substituted chloromethanes, hydrogen chlorine and some ethane
Presence of ethane proves the reaction proceeds by this mechanism
In strong sunlight, reaction between chlorine and methane is explosive. Reaction with Br is lower
·CH3 + ·CH3 —> C3H6
Free radical substitution is of limited use in synthesising haloalkanes since a mixture since a mixture of products is made. If carbon chain is longer this can also lead to halogenation in diff positions
why does Radical substitution produce a mixture of organic products?
further substitution OR produces different termination products OR More than one termination step
substitution at different positions along chain