chem135-exam2
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
23 Terms
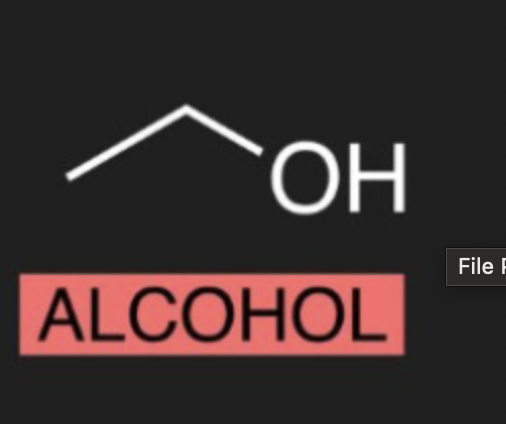
what is the organic molecule of Alcohol
what is the organic molecule of Ether

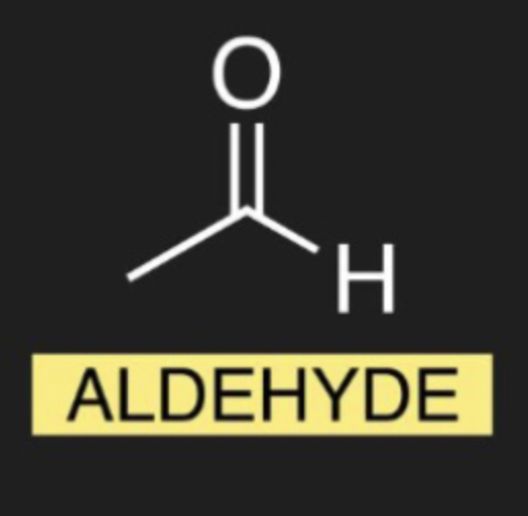
what is the organic molecule for Aldehyde
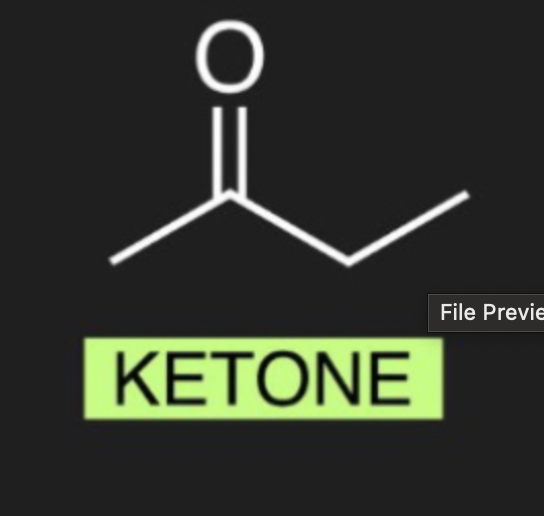
what is the organic molecule for Ketone
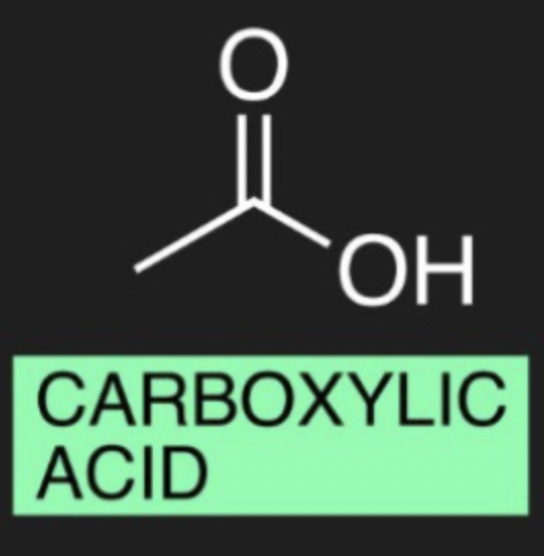
what is the organic molecule for carboxylic acid
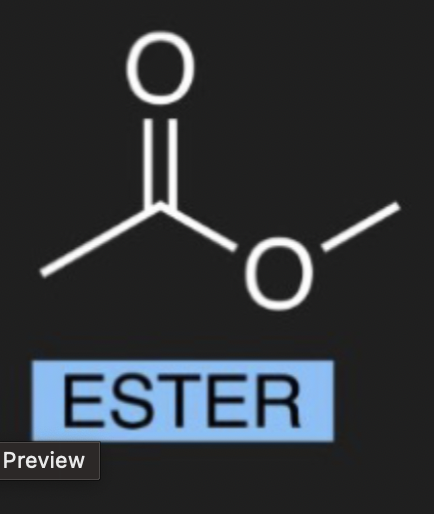
what is the organic molecule for Ester
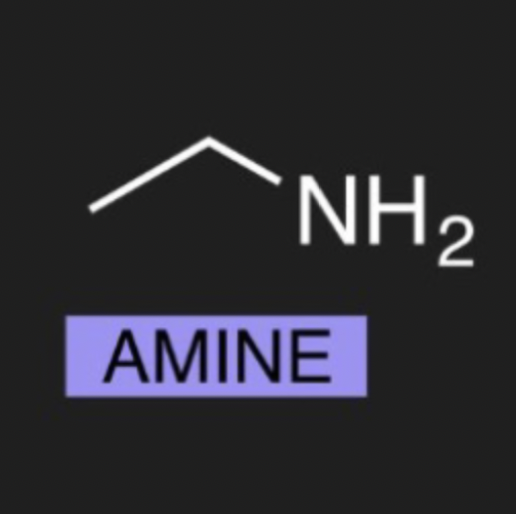
what is the organic molecule for Amine
What is the organic molecule for Amide

What are the exceptions for expanded octets
Only elements in the 3rd row of the periodic table and below can have expanded octets (up to 12 – 14 electrons are typical)
Two electron groups in electron geometry form
Linear
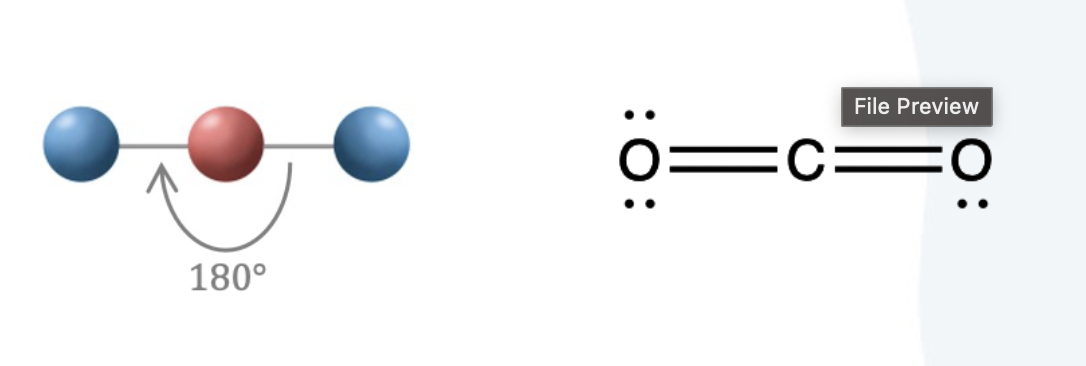
Three electron groups in electron geometry form
trigonal planar

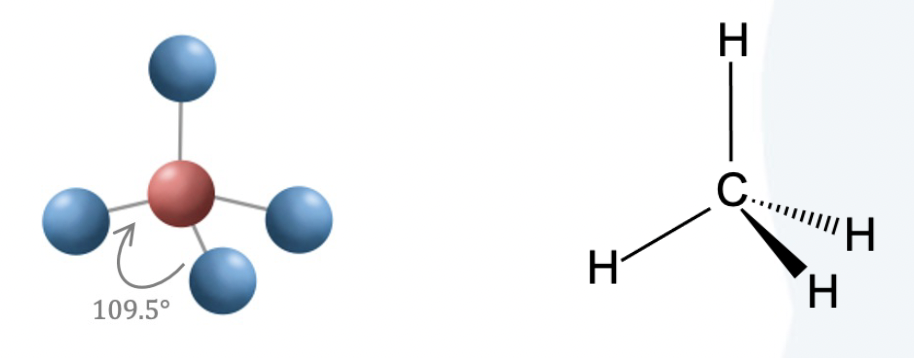
Four electron groups in electron geometry form
tetrahedral
What is the hybridization of two electron groups
sp
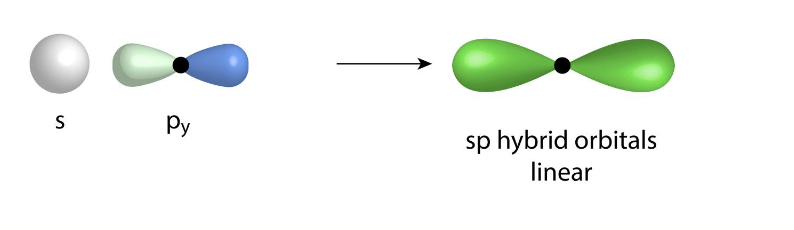
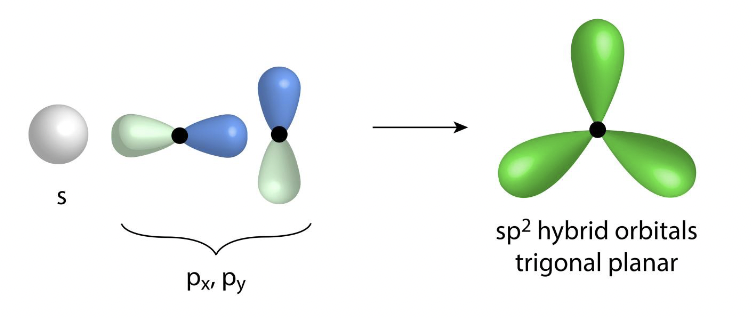
what is the hybridization of three electron groups
sp²
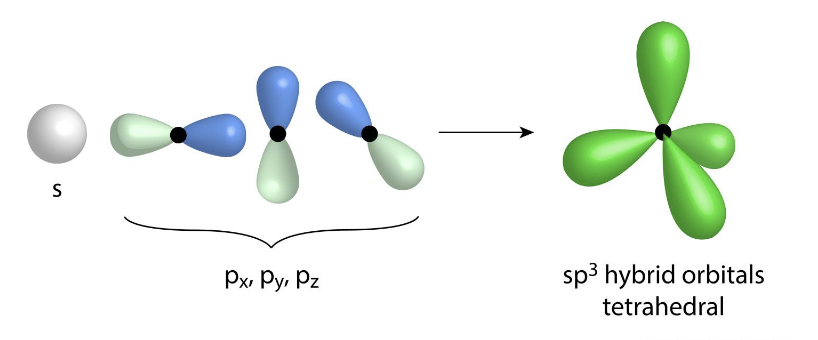
what if the hybridization of four electron groups
sp³
sp² hybridization results in two types of orbitals
sigma and pi orbitals
Dipole-dipole
interactions between two molecules with a permanent dipoles
Dispersion
Present in all atoms/molecules but most important for non-polar ones.
Dipole-induced dipole
interactions between a polar molecule and a nonpolar molecule, where the polar molecule induces a temporary dipole in the nonpolar molecule.
Hydrogen bonding is
The strongest type of dipole-dipole interaction between a hydrogen atom bonded to a highly electronegative atom and another electronegative atom.
the greater the surface area of a molecule (the size and molar mass)
the greater/stronger the dispersion
hydrolysis
adding water to polymer to break them into monomers
What does amphipathic mean?
Amphipathic refers to molecules that possess both polar (hydrophilic) and non-polar (hydrophobic) regions.