Electronic Configurations
1/9
Earn XP
Description and Tags
An electronic configuration is a description of which orbitals in an atom contain electrons and how many electrons are in each orbital
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
10 Terms
Aufbau Principal
As the atomic number increases, electrons are added to the available orbitals. Electrons are added to the orbitals having the lowest energy first.
Hund’s Principle
Orbitals of equal energy are each occupied by one electron before any pairing of electrons occur
Pauli Exclusion Principle
Each orbital can contain only a massive amount of two electrons with opposite spins
2 electrons in an s-type subshell
6 electrons in a p-type subshell
10 electrons in a d-type subshell
14 electrons in an f-type subshell
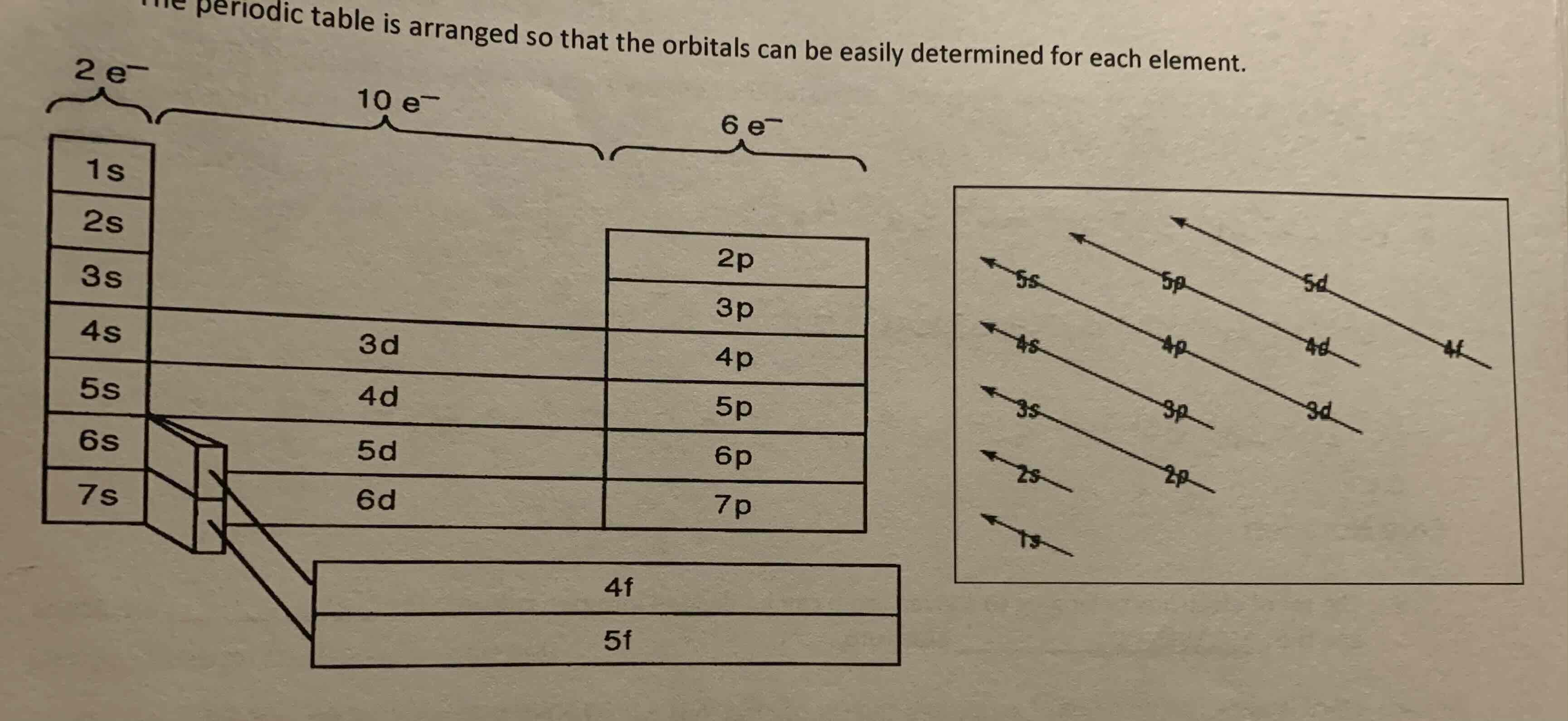
Orbitals
Core Notation
The core of an atom is the set of electrons with the configuration of the previous noble gas, having an atomic number less than that of the atom being considered
Rules for Core Notation
1. Locate the atom and note the noble gas at the end of the row above the element.
2. Start to write the electron configuration as usual replace a part of the electron configuration corresponding The configuration of the noble gas with the symbol for the noble gas in square brackets. Followed the Core symbol with the electronic configurations of the remaining outer electrons.
Ex. [Noble Gas] + Extra Orbitals
Exceptions
Chromo and copper, because half filled and filled orbitals are more favourable
Electron Configurations for Anions
Add electrons equal to a charge to the last unfilled subshell, starting with the Neutral atom left off
Electron configurations for Cations
1. starting with the neutral configuration in core notation, removes electrons from the outermost shell the largest energy level first
2. If there are electrons in both the S and P orbitals of the outermost shell, the electrons in the P orbitals are removed first
Summary: P than S than D than F
Valence electrons
Valence electrons are all the electrons in the atom EXCEPT those in the
- [core]
- filled D and F sub shells
* if it’s unfilled, ‘F + D’ then it’s part of the valance
* If you can make something helpful or filled, do it
*S+P UNLESS P is filled