Lectures 7-10
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
Solvent Extraction
Separation based on differences in solubility and polarity ("like dissolves like")
Liquid-Liquid Extraction (LLE)
Separation using two immiscible liquids (don’t mix) where solutes partition between phases
Batch extractions - to increase extraction efficiency (recovery)
Solid-Liquid Extraction
Extraction of compounds from a solid using a solvent (e.g. tea/water, beets/water, caffeine/methanol)
Immiscible Solvents
Liquids that do not mix and form two layers (e.g.
Organic Layer
Layer containing organic solvent (often less dense - on top)
Aqueous Layer
Water-based layer in extraction system
Partition Coefficient (Kᴅ)
Ratio of solute concentration in organic phase to aqueous phase at equilibrium
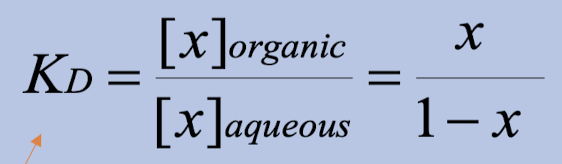
Kᴅ > 1
Compound prefers organic phase (hydrophobic)
Kᴅ < 1
Compound prefers aqueous phase (hydrophilic)
Equilibrium in Extraction
Solute distributes between phases based on relative solubility
Separatory Funnel
Apparatus used to mix and separate immiscible liquids
Green Solvents
Safer
Selective Extraction
Using solvent polarity to isolate specific compounds
Distillation
Separation by vaporization followed by condensation
General distillation - involves heating a liquid mixture to form vapor and condensing the vapor to collect separated components
Components with higher vapor pressure (lower boiling point) vaporize more readily
Volatility
Tendency of a substance to enter the gas phase
Vapor Pressure (Vp)
Pressure exerted by vapor in equilibrium with liquid
Boiling point (BP)
Temperature where vapor pressure equals atmospheric pressure
lower pressure = higher BP = longer cooking
higher pressure = lower BP = faster cooking
High vapor pressure
More volatile, lower BP
Low vapor pressure
Less volatile, higher boiling point
Intermolecular Forces (IMFs)
Forces between molecules affecting volatility
Stronger IMFs
Lower volatility, higher BP
London Dispersion Forces
Weak forces from temporary dipoles (present in all molecules) - stickyness
Shapes of molecules affect the magnitude of the LD forces
Size: molecules with more surface area (longer hydrocarbon chains) tend to have more LD forces and higher BPs
Dipole-dipole interactions
Attractions between polar molecules
Are additive
Partial negative of one molecule interacts with the partial positive of another molecule
Hydrogen bonding
Strong IMF when H is bonded to N, O, or F
More H bonds = more difficult to get to enter the vapor phase
Ion-dipole forces
Interactions between ions and polar molecules (cation and anion)
Simple distillation
Used when boiling points are very different
Fractional distillation
Used when boiling points are close; uses multiple vaporization steps
Repeated distillations
Temperature gradient as you move up the column (hottest at the bottom)
Each time the vapor condenses and vaporizes, the composition of the more volatile liquid in the vapor increases
Fractioning column is placed between the boiling flask and the condenser
The fractioning column has added surfaces area for “refluxing”
The process in which a liquid is boiled, its vapors are condensed, and the condensate returns to the reaction vessel, allowing prolonged heating at the solvent spoiling point without loss of material
Theoretical plate
Measure of separation efficiency in distillation
Reflux
Continuous vaporization-condensation cycle to improve separation
Color in foods
Result of light interacting with matter and being reflected
White light
Contains all visible wavelengths
Color perception
Brain’s interpretation of reflected light
reflected wavelengths are perceived as color
Chlorophyll
Pigment responsible for green color
Carotenoids
Pigments responsible for yellow/orange color
Anthocyanins
Pigments responsible for red/blue color (pH dependent)
pH < 2: flavylium cations (RED) → MORE STABLE
More -OCH3 = redder color
pH 2.5-6: quinoidal base (BLUE)
More -OH = deeper blue color
pH > 6: chalcone (NO COLOR)
Stability
Increase with more methoxyl groups on the B-ring
Decreases with increasing hydroxylation of the B ring
Colorimetry
Science of measuring color objectively
Hue
Type of color (red, blue, green)
Value (lightness)
Brightness or darkness of a color
Chroma
Intensity or saturation of color
Subjective color
Perceived differently by individuals
Objective color
Measured numerically using instruments
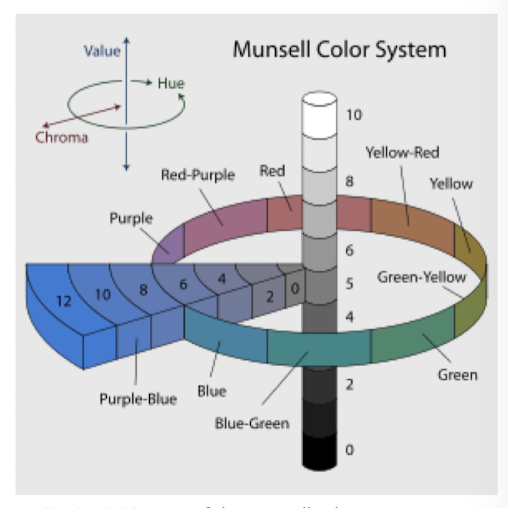
Munsell system
Color system based on hue (R, Y, G, B, P), value (lightness), chroma (color intensity)
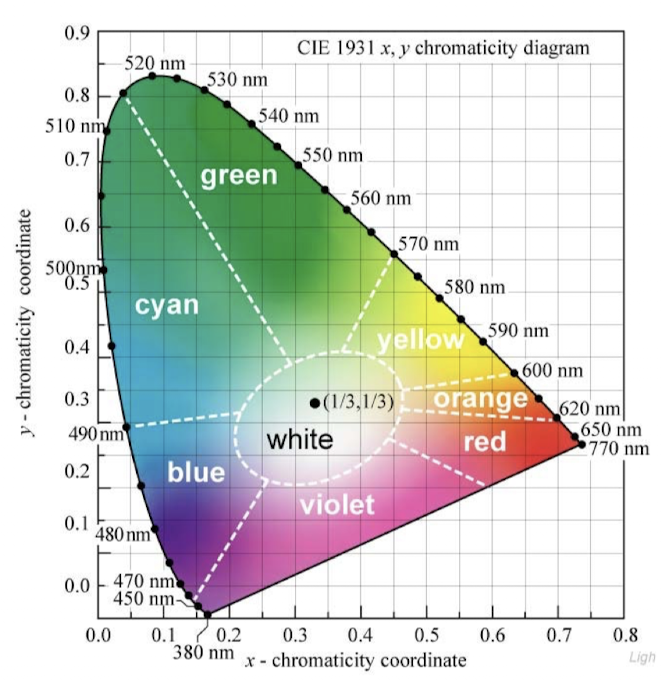
CIE system
Standardized color measurement system
Tri-stimulus CIE X(red)Y(green)Z(blue) color space
Dominant wavelength - white light through the object coordinates to the edge
Distance from white light to the object’s coordinates is % purity
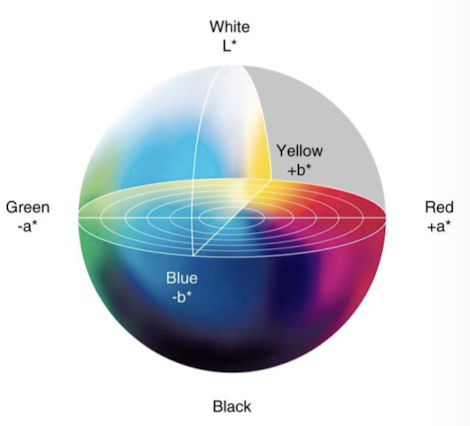
Lab Color system
Uses L* (lightness), a* (red-green), b* (yellow-blue)
Hunter lab values
Factors affecting color
Light source, observer, background, and sample size
Phenolic compounds
Molecules with aromatic ring + hydroxyl group(s)
Especially abundant in plants
Many are pigments
Important for quality, stability, and as antioxidants
Involved for browning reactions (converted to quinones)
Aroma
Quinone + free amino acids = aromas (aldehydes)
Polyphenols vs phenolics
“Phenolic compounds” is more accurate term
Antioxidant function
Donate electrons to neutralize free radicals
Phenolics in food
Affect color, flavor, stability, and astringency
Enzymatic browning
Phenolics oxidized to quinones by PPO
Tissue damage (cutting, bruising) brings PPO into contact with phenolics
PPO catalyzes oxidation of phenolics to quinones
Quinones condense and polymerize to form brown pigments (melanins)
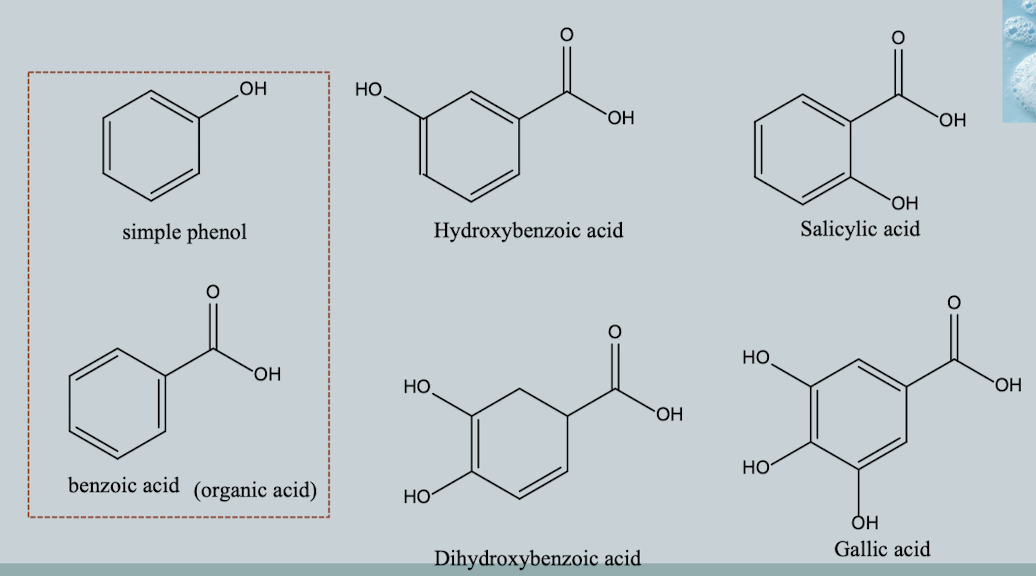
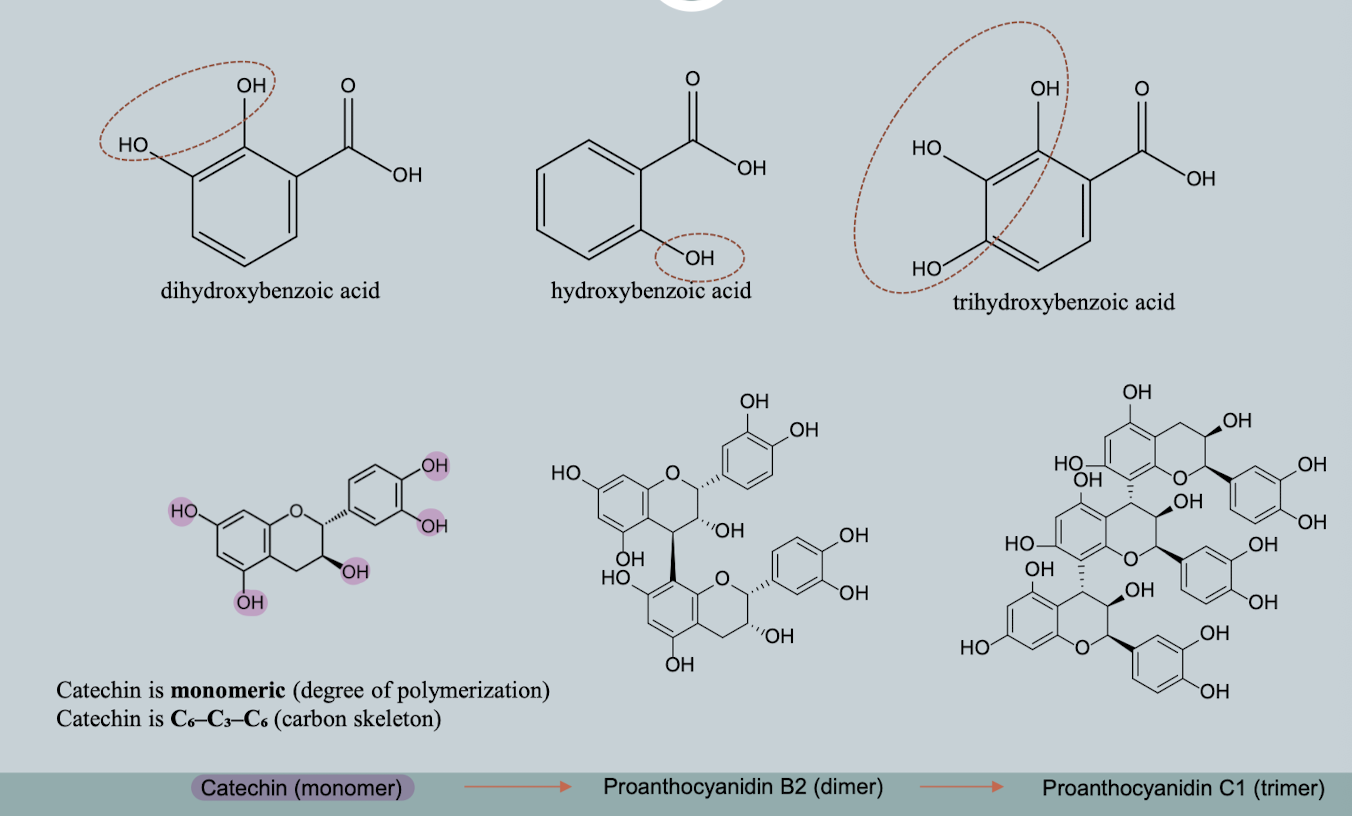
Hydroxybenzoic Acids (C6–C1)
Phenol + carboxylic acid structure
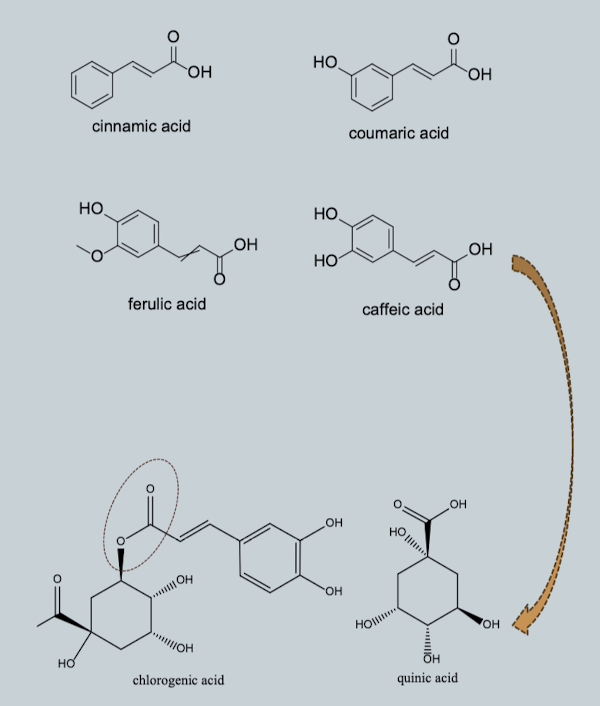
Hydroxycinnamic Acids (C6–C3)
Aromatic ring + propionic acid side chain
Predominantly present as esters
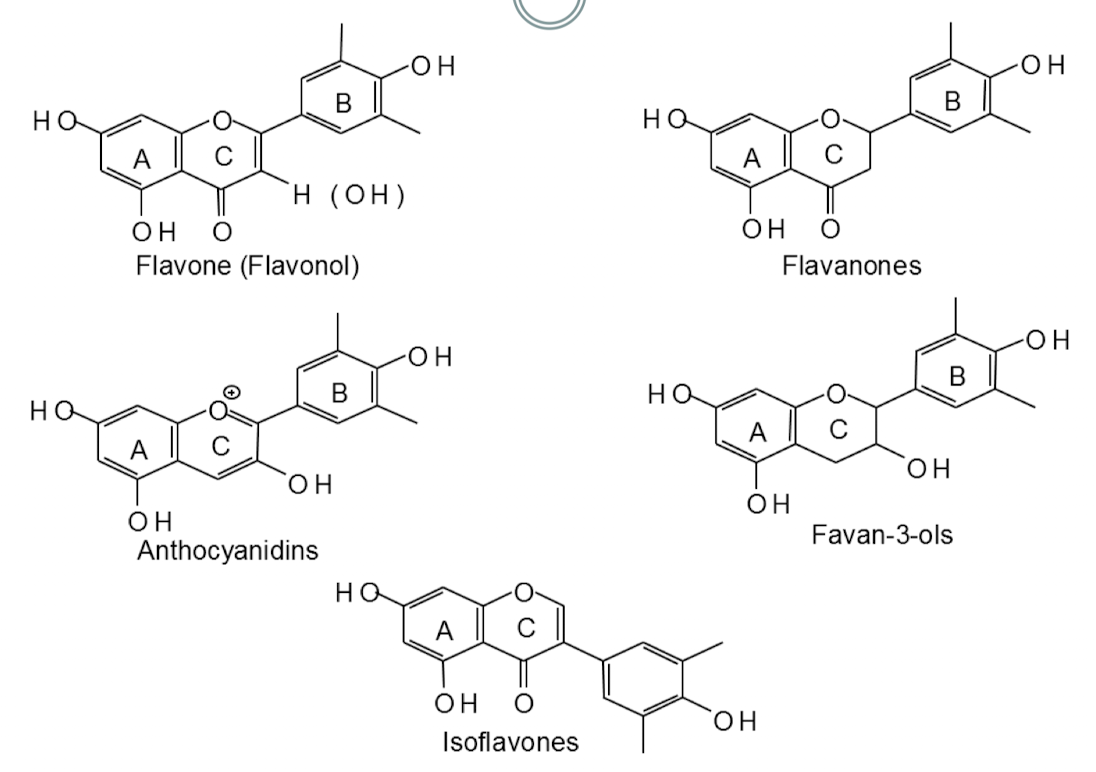
Flavonoids (C6–C3–C6)
Major class of plant phenolics with 3-ring structure
Color properties are related to the conjugated systems
Double bonds are conjugated when they are separated by one single bond (C=C-C=C)
Color is only present when the C ring is conjugated with both the A and B rings
Classes:
Flavonols
Isoflavones
Flavan-3-ols
Flavonones
Anthocyanidins
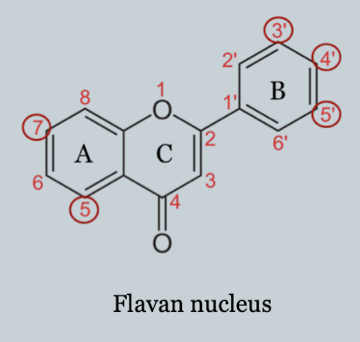
Flavan Nucleus
Two aromatic rings + heterocyclic ring
Anthocyanidins
Flavonoids responsible for red/blue colors
Tannins
Phenolics that bind proteins and cause astringency
Phenolic compounds in plant extracts that can convert animal skin into leather (tanning)
Hydrolysable Tannins
Breakable by hydrolysis (ester bonds)
Bind protein
Condensed Tannins
Polymers of flavan-3-ols (non-hydrolyzable)
Proanthocyanidins
Bind protein
Astringency
Dry, puckering sensation from tannins binding proteins
Polyphenol Oxidase (PPO)
Enzyme that catalyzes browning reactions
% Recovery & Yield
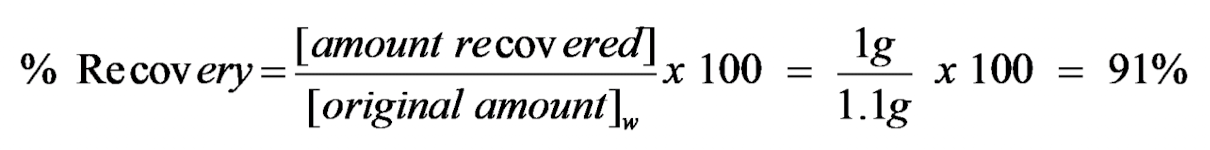
Drying agents
Water removal from solvents
Wash the organic layer with anhydrous salt
Add a drying agent (salt) to the organic solvent
Salt pulls the water from the organic layer
Techniques for extracting volatiles - Purge and Trap
Used to isolate volatile organic compounds (VOCs) from liquids or solids, especially for flavor and aroma analysis
How purge and trap works
An inert gas (usually helium or nitrogen) is bubbled through the sample
Volatile compounds are stripped (purged) from the sample into the gas phase phase
The gas carrying the volatiles passes through a sorbent trap
Volatile compounds are captured and concentrated on the trap
Distortion and analysis
Trap is rapidly heated
Trapped compounds are released (desorbed) and transferred to an instrument, typically GC or GC-MS
Head space analysis
Technique used to measure volatile compounds present in the gas phase (headspace) above a sample
Sample is sealed in a closed vial
Volatile compounds partition from the sample into the headspace
After equilibrium is reached, a portion of the headspace gas is sampled
Fractional Distillation Columns
Open tubular column
Low surface area
Low efficiency (plates)
Vigreux column
Moderate surface area
Moderate efficiency
Packed column
Increased surface area
High efficiency
Cones
Color
Blue: 420-440 nm
Green: 530-540 nm
Red: 560-580 nm
Rods
Sensitive to brightness
Monochromatic
Light
EMR (electromagnetic receptors) which stimulate the retina of the eye
What influences color?
Light source differences
Observer differences
Size difference
Background differences
CIE Standardized Geometrics for Reflectance Instruments: 45/0 and 0/45
45 illumination/0 measure
Use to minimize specular (gloss) reflection (apples, tomatoes)
When surface texture or shine could distort color readings
0 illumination/45 measure
When illumination needs to be uniform and normal to the surface
When sample shape or orientation varies (grains, flat matte foods)
Examples of Polyhydroxylated Phenols
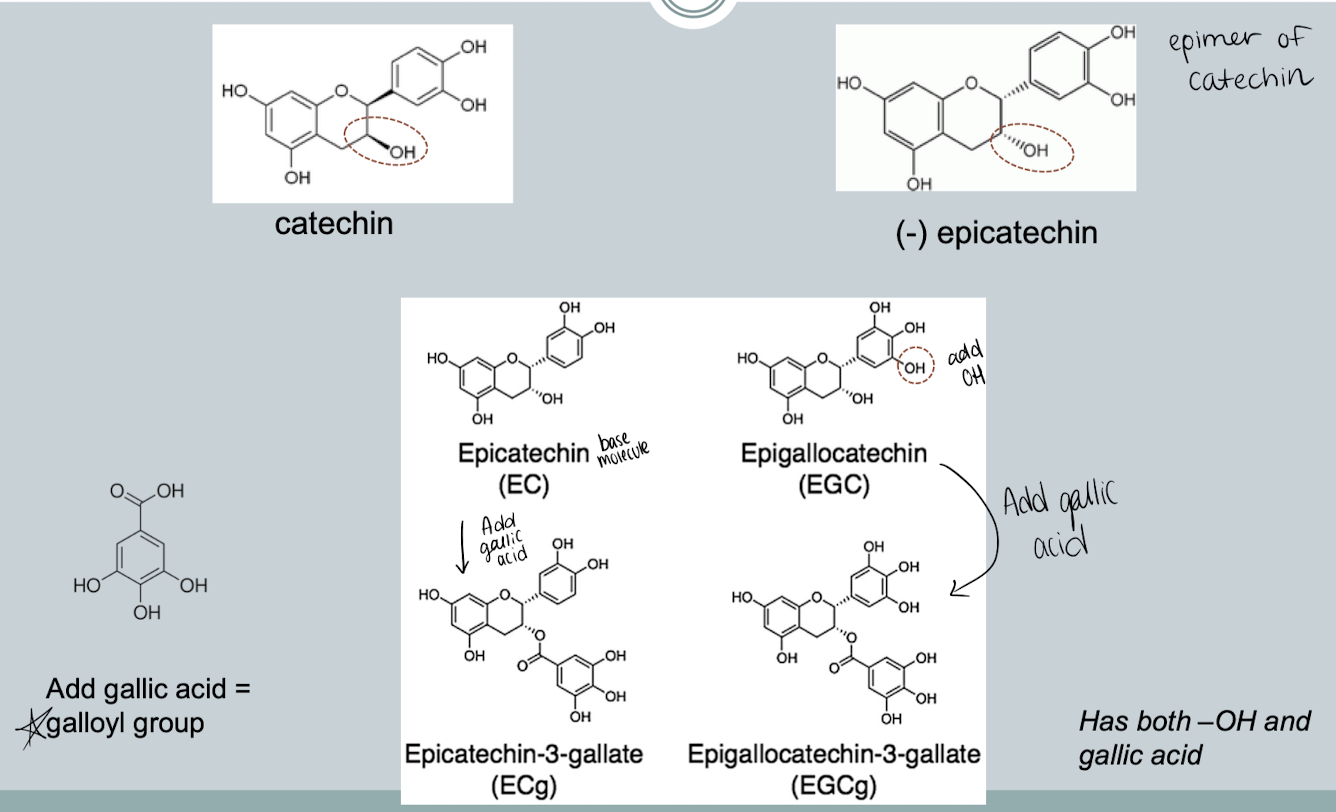
Phenolic compounds in tea
Lignin
Part of the plant cell wall, non-digestible material
Defined as dietary fiber
Glycosylated Phenolic Compounds
Glycosylated forms
Phenolics are bound to one or more sugars (mono-, and oligosaccharides)
Aglycone form - not bound to a sugar
Antioxidant properties of phenolic compounds
Many exhibit antioxidant properties
Phenolic compounds act as reducing agents (donate H+ and e-)
Phenolics donate the H+/e- from the -OH groups on the aromatic ring
Phenolic becomes a free radical that is stabilized through resonance
Controlling enzymatic oxidation reactions
Eliminate oxygen (PPO requires oxygen)
Addition of metal complexing compounds (PPO is a metal low enzyme therefore, substances that complex metal ions will inactivate the enzyme)
Reduce the pH (most PPO enzymes have a pH optimum between pH five through seven, reducing pH, add adding acid will limit the rate of the enzyme
Heat-denature the enzyme (denatures above 40°C)
Add ascorbic acid (reduces the Keynote back to the o-diphenol state)
Add sulfite (binds with amino acids in the active site of PPO and deactivates it)
Final compounds and taste
Some phenolic compounds can be bitter (citrus fruit)
Glycoylated flavones/flavanones
Glycosyl bond is responsible for the bitterness
Total phenolic compounds assay (Folin-Ciocalteu)
Measuring the reduction of reagent and alkaline conditions by phenolic compound → blue color
Not specific to phenolics:
Also detect other reducing agents
Report reported as total phenolics
2,2-Diphenyl-1-picryhydrazyl radical assat (DPPH)
Radical scavenging acids
DPPH is a stable free radical with an unpaired electrons (blue/purple)
When an antioxidant is added to the assay, the antioxidant donates a hydrogen atom and an electron to the DPPH
This neutralizes (quenches) the DPPH radical
Absorbance decrease is measured at 517 nm
Measures radical scavenging (antioxidant) activity
More specific to antioxidants than FC